Clear Sky Science · en
Universal method for polyvalent ions’ redox state quantification in inorganic materials down to trace concentrations
Why tiny charges inside solids matter
Many of today’s smart materials—those that store energy, glow after the lights go out, or clean up pollution—owe their special powers to atoms that can carry different levels of electrical charge. These “shape‑shifting” metal ions are hidden inside glasses, ceramics, and crystals, and their exact charge balance governs how the whole material behaves. Yet, until now, measuring that balance has often required large, expensive machines or complicated procedures. This article introduces a simple bench‑top chemistry approach that lets researchers accurately count these different charge states, even when they are present only in trace amounts.
A simple idea for a hard measurement
The central challenge is to determine how many of the same kind of ion in a solid are in a more “electron‑rich” (reduced) state and how many are in a more “electron‑poor” (oxidized) state. Traditional tools—such as advanced X‑ray spectroscopy or magnetic measurements—can do this, but they are costly, not always accessible, and sometimes even disturb the delicate charge state they try to probe. The authors revive and generalize an older wet‑chemistry concept: dissolve the material in an acid together with a well‑chosen reacting agent, let the ions exchange electrons in a controlled way, and then carefully measure how much reacting agent has changed. From this small shift, one can work backward to how many ions in the original solid carried each charge state.
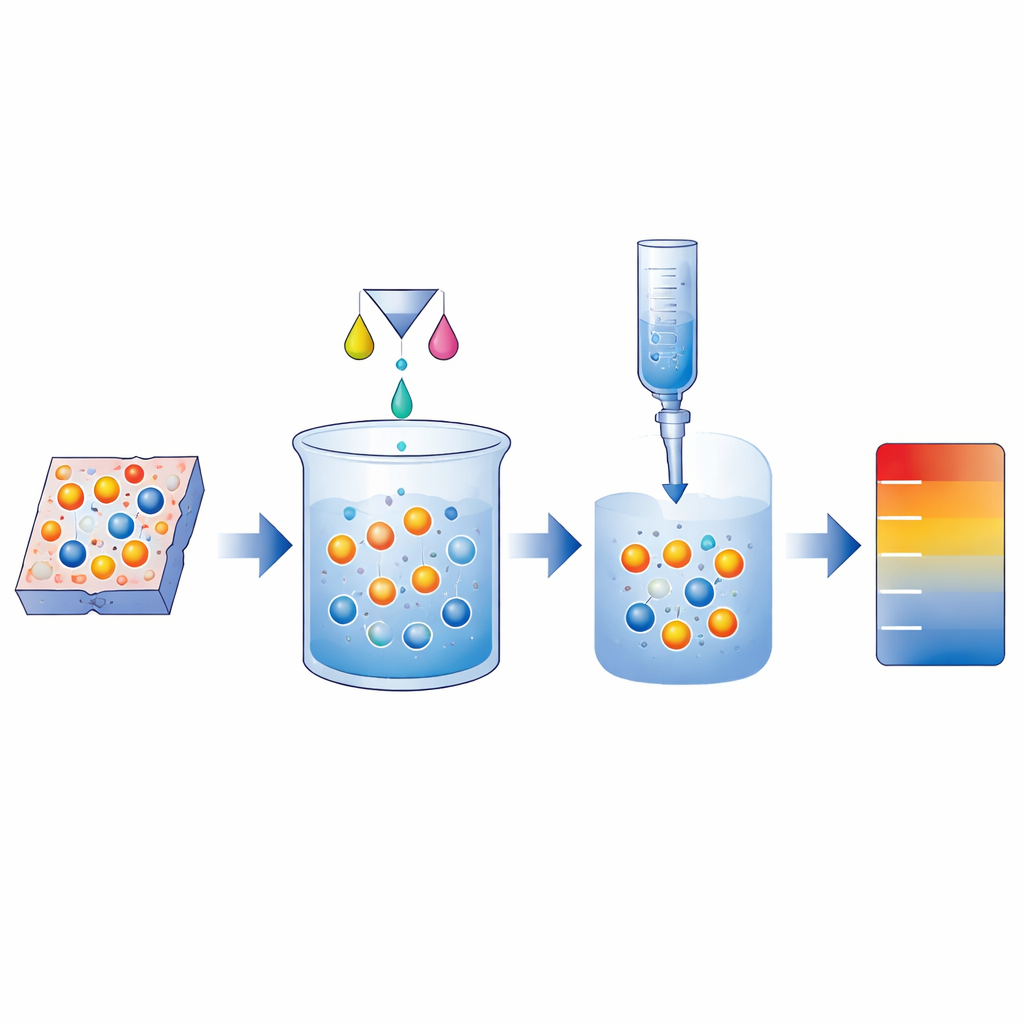
Two twin methods: counting electron takers and givers
The study presents a matched pair of methods with memorable roles. The first, called Quantification of Oxidizing Species (QOS), focuses on ions that like to take electrons—strong “electron grabbers” such as certain forms of cerium, terbium, or chromium. In this case, the dissolved material is mixed with iodide ions. The oxidizing ions steal electrons from iodide, turning it into iodine. That iodine is then titrated—gradually neutralized—with a thiosulfate solution while monitoring the color or electrical potential. The second method, Quantification of Reductive Species (QRS), targets ions that like to give away electrons, such as europium in its bright‑glowing form. Here, the dissolved sample meets excess iron ions that accept electrons, and the newly formed iron species are titrated with a cerium solution. In both cases, the volume of titrant needed to reach the turning point directly reveals how many ions in the solid were in the high or low charge state.
From standard samples to complex real materials
To show that their approach is robust, the authors first test it on well‑defined powders containing known charge states of metals like copper, tin, manganese, and rare‑earth elements. The measured results match theoretical expectations with uncertainties of only a few percent, even when the relevant ions lie outside the normal stability range of water. They then apply the methods to realistic glass and crystal compositions that are important for optics and lighting, including materials hosting unusual high‑charge forms of manganese and efficient light‑emitting europium. The technique reliably quantifies these species even when they are present at levels of a few parts per million, and it works across oxides, nitrides, and fluorides. The authors also explore edge cases such as materials with three different charge states of the same element, or mixtures of several metals that can trade electrons with one another, and outline how to adapt the calculations in these more tangled situations.
Linking simple tests to deeper material design
Because the wet‑chemical routes give absolute numbers for oxidized versus reduced ions, they can serve as a yardstick for more sophisticated but ambiguous probes. The authors demonstrate how their measurements can calibrate optical absorption spectra, turning broad color bands into precise counts of how many ions occupy specific local environments in the glass. They also map how the balance between charge states shifts with the “electron‑donating strength” of the glass itself, its melting temperature, and the available oxygen during processing. These trends provide designers with a practical recipe book: by tuning composition and furnace conditions, they can steer the hidden charge balance, and in turn control properties like color, electrical conductivity, or long‑lasting glow.
What this means for future smart materials
In everyday terms, the work offers a low‑cost, accurate way to check whether a material’s “internal knobs” have been set to the right positions. With only small sample pieces, common glassware, and safe reagents, laboratories can now quantify charge states that were previously accessible only at large facilities or not at all. This enables faster optimization of glasses and ceramics for tasks such as persistent luminescence, photo‑driven reactions, smart windows, and advanced energy storage. By turning a hard measurement into a routine one, the methods open the door to more deliberate and fine‑grained control over the behavior of complex inorganic materials.
Citation: Duval, A., Greiner-Mai, N., Scheffler, F. et al. Universal method for polyvalent ions’ redox state quantification in inorganic materials down to trace concentrations. Commun Mater 7, 87 (2026). https://doi.org/10.1038/s43246-026-01109-w
Keywords: redox state, wet chemical analysis, functional glasses, transition metal ions, luminescent materials