Clear Sky Science · en
Synthesis and application of silk fibroin nanoparticles for drug delivery
Spider Silk’s Cousin as a Smarter Medicine Carrier
Imagine if the same material that makes a silkworm’s cocoon could help medicines find diseased tissue while sparing the rest of the body. This article reviews how silk fibroin — the main protein in silkworm silk — can be turned into tiny particles that ferry drugs directly to tumors, inflamed intestines, and wounds. For readers, it’s a glimpse into how a familiar, natural material might quietly transform cancer treatment, gut health, and tissue repair.
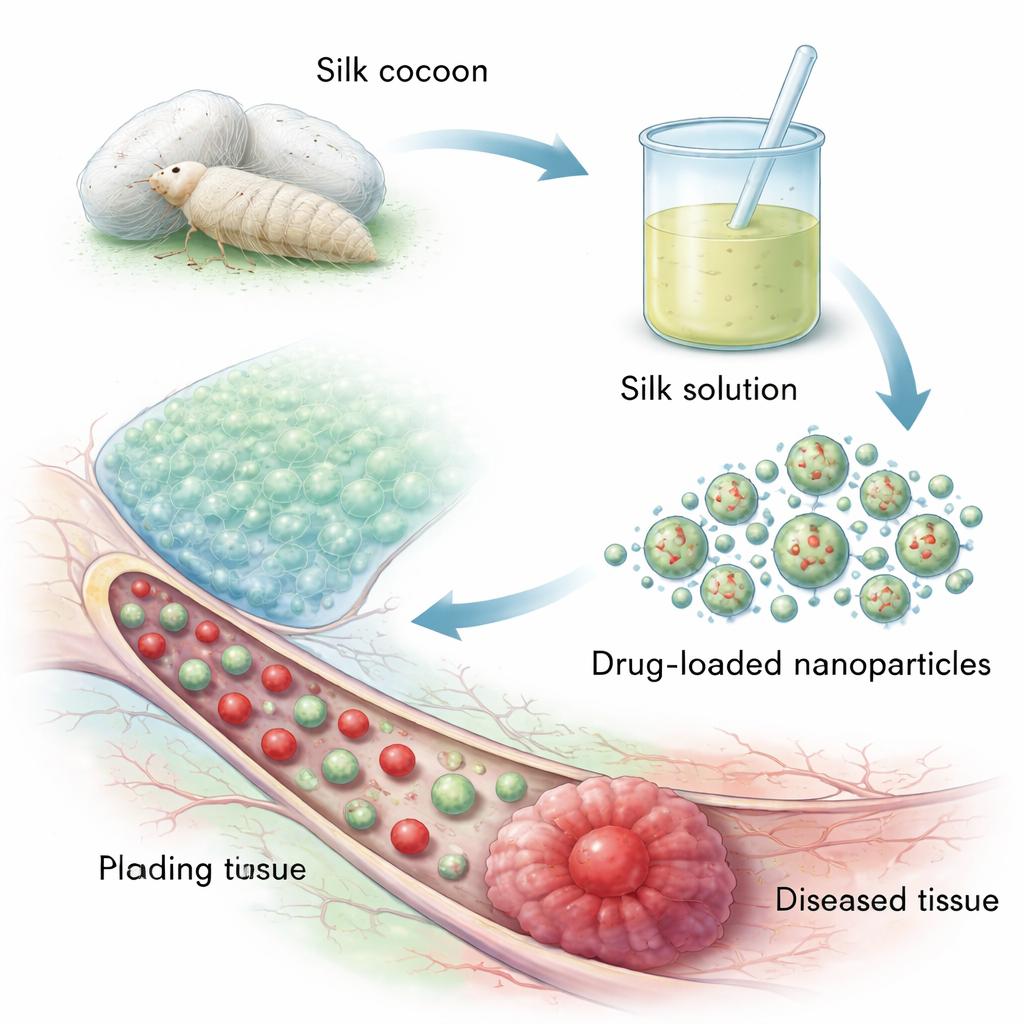
From Cocoon Threads to Tiny Medicine Packages
Silk fibroin comes from the cocoons of the domesticated silkworm, a readily farmed source compared with spider silk. The cocoon has a strong fibroin core wrapped in another protein, sericin, which can trigger immune reactions. To make medical-grade silk, manufacturers first remove sericin in a process called degumming, then dissolve, purify, and dry the fibroin. At the molecular level, silk fibroin is built from long, repeating chains of simple amino acids like glycine and alanine that can fold into tightly packed “beta-sheet” regions. These ordered regions give silk its strength and let scientists tune how fast it dissolves in the body, a key feature for controlling how long a drug is released.
Ways to Shape Silk into Nanoparticles
Researchers have developed several routes to turn liquid silk solutions into nanoparticles only tens to a few hundred billionths of a meter wide. In desolvation methods, silk solution is dripped into a non‑solvent such as ethanol, prompting the protein chains to fold into beta‑sheets and self‑assemble into solid spheres. Emulsion techniques create water‑in‑oil droplets that harden into particles, useful for carrying oily drugs but requiring careful removal of solvent. Electro‑spraying uses high voltage to break a silk solution into fine charged droplets that dry mid‑air into particles, while microfluidic chips mix silk and “antisolvents” in tiny channels to generate very uniform, reproducible nanoparticles. Each approach trades off cost, control over size, and ease of scaling up for mass production.
Fine-Tuning Silk for Targeted and Timed Release
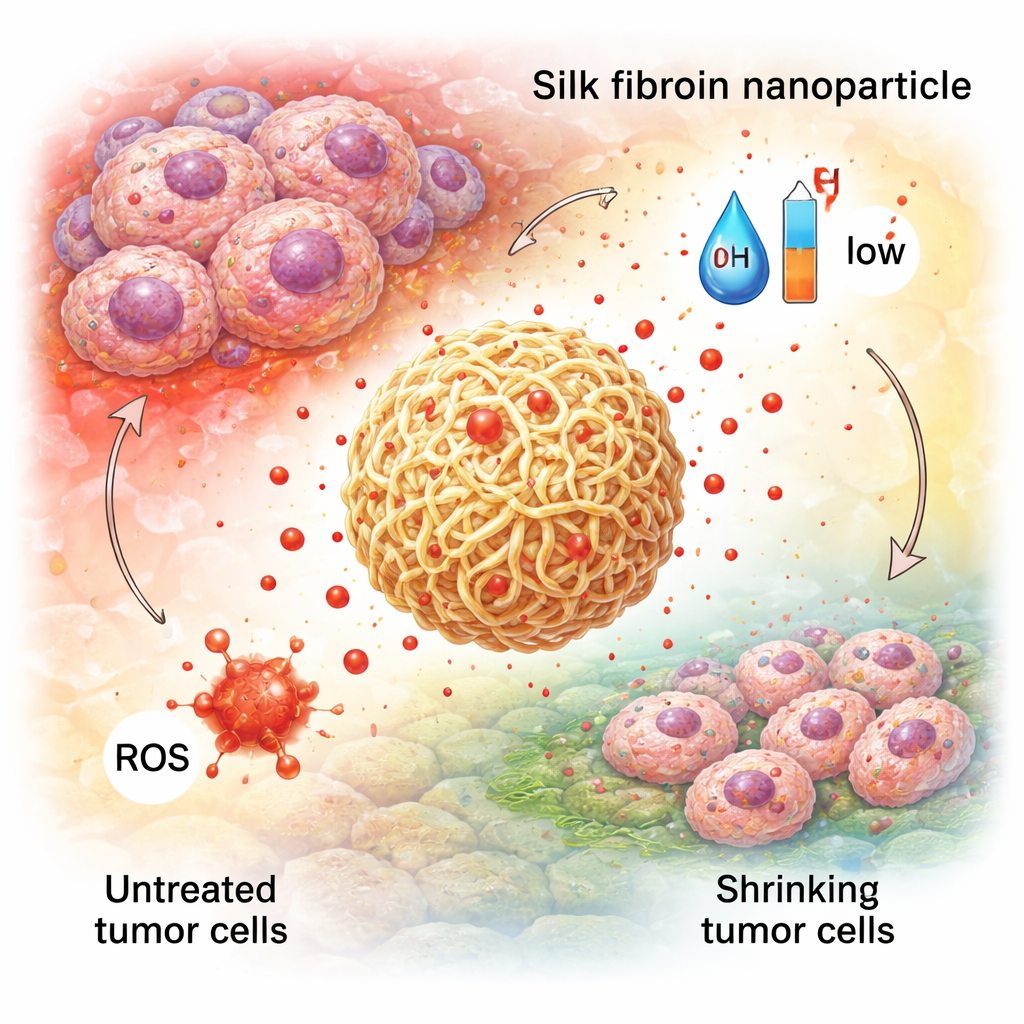
Because silk fibroin is rich in reactive chemical groups, it can be modified much like a Lego structure with extra pieces. Chemists attach small molecules, polymers, or biological “tags” to change how the particles interact with water, how fast they break down, or which cells they stick to. For instance, adding polyethylene glycol (PEG) can make particles more stable in blood and less visible to the immune system, while grafting short protein fragments or vitamins can help them bind to tumor cell receptors. Crucially, the beta‑sheet content and added linkers can be adjusted so that silk nanoparticles respond to the environment: they can loosen in acidic conditions, in the presence of certain enzymes, or in oxidized regions, releasing more drug exactly where tissues are diseased.
Early Successes in Cancer, Gut Disease, and Wound Repair
These designer particles have been tested in many disease models. In cancer, silk nanoparticles have carried chemotherapy drugs and even combinations of drugs plus light‑activated agents, showing better accumulation in tumors, more precise release in the harsh tumor environment, and fewer side effects than free drugs. In inflammatory bowel diseases, orally administered silk particles loaded with natural antioxidants or anti‑inflammatory compounds can stick to gut mucus, navigate through it, and release their payload in response to high levels of reactive molecules, helping to calm inflammation and repair the intestinal barrier. In tissue engineering and wound healing, silk nanoparticles are blended into scaffolds and hydrogels to slowly release growth factors or antibiotics, strengthening bone‑like materials and speeding the closure and clean healing of skin wounds.
Bridging the Gap from Lab Bench to Hospital Bed
Despite the promise, silk fibroin nanoparticles are still at the preclinical stage. The review highlights major obstacles: batch‑to‑batch differences from how silk is processed, traces of other proteins or solvents that could affect safety, and the difficulty of producing large volumes under strict pharmaceutical quality rules. Microfluidic manufacturing offers better control but is currently expensive and complex to scale. Regulators will also require a clear understanding of how these particles break down in the body, how they are cleared, and how reliably they can be made. The authors argue that greener processing methods, standardized protocols, continuous monitoring of particle quality, and close collaboration between academic labs and industry could gradually move silk nanoparticles from elegant lab demonstrations to everyday medical tools.
What This Could Mean for Patients
In plain terms, silk fibroin nanoparticles aim to deliver more medicine where it is needed and less where it is not. By using a biocompatible, biodegradable protein that can be shaped and tuned at multiple levels, scientists hope to reduce the harsh side effects of chemotherapy, provide more effective long‑term treatment for gut inflammation, and accelerate tissue repair after injury. While many engineering and regulatory hurdles remain, the work summarized in this article suggests that tomorrow’s “smart” drug carriers may be spun not from exotic synthetics but from the same silk that has clothed humans for thousands of years.
Citation: Bao, S., Yang, X., Reis, R.L. et al. Synthesis and application of silk fibroin nanoparticles for drug delivery. Commun Mater 7, 66 (2026). https://doi.org/10.1038/s43246-026-01108-x
Keywords: silk fibroin nanoparticles, targeted drug delivery, cancer nanomedicine, inflammatory bowel disease, tissue engineering