Clear Sky Science · en
Influence of grain size on the solid-state direct reduction of polycrystalline iron oxide
Why the Size of Tiny Grains Matters for Green Steel
Steelmaking is one of the world’s biggest sources of carbon dioxide, largely because iron ore is usually turned into metal using coal. A promising cleaner route swaps coal for hydrogen gas, which releases water instead of CO2. This study asks a deceptively simple question with big practical consequences: if the iron ore is made of crystals ("grains") of different sizes, does that change how well hydrogen can turn it into metal? The answer is yes—and the way those grains shape a hidden network of pores turns out to be crucial for designing greener steel processes and other porous materials.
From Ore to Metal with Hydrogen
To explore this, the researchers worked with hematite, a common iron oxide used in ironmaking. Instead of industrial pellets with many complexities, they created clean, dense, lab-made pellets containing either very large grains (around 30 micrometers across) or ultrafine grains (around 1 micrometer). They then exposed these pellets to pure hydrogen gas at 700 °C while carefully tracking how fast oxygen was removed—essentially weighing the samples in real time as they lost mass. This allowed them to compare the “reduction” behavior of the two grain sizes under identical, controlled conditions.
Fast Start Versus Strong Finish
The timing of the reaction turned out to depend strongly on grain size. At the beginning of the process—up to roughly one-third of the total oxygen removed—the large-grained hematite reduced more quickly. A sharp reduction front moved inward from the surface, in line with the classic “shrinking-core” picture, where an outer shell turns into metal while the inner core remains oxide. But as the reaction progressed further, the pattern flipped. For much of the remaining journey from oxide to metal, especially beyond about half-complete reduction, the ultrafine-grained samples pulled ahead and reduced more rapidly. When the heating was fast, the large-grained pellets tended to get “stuck” with more leftover oxide, while the ultrafine ones got closer to full conversion under the same schedule.
Hidden Highways for Gas and Water
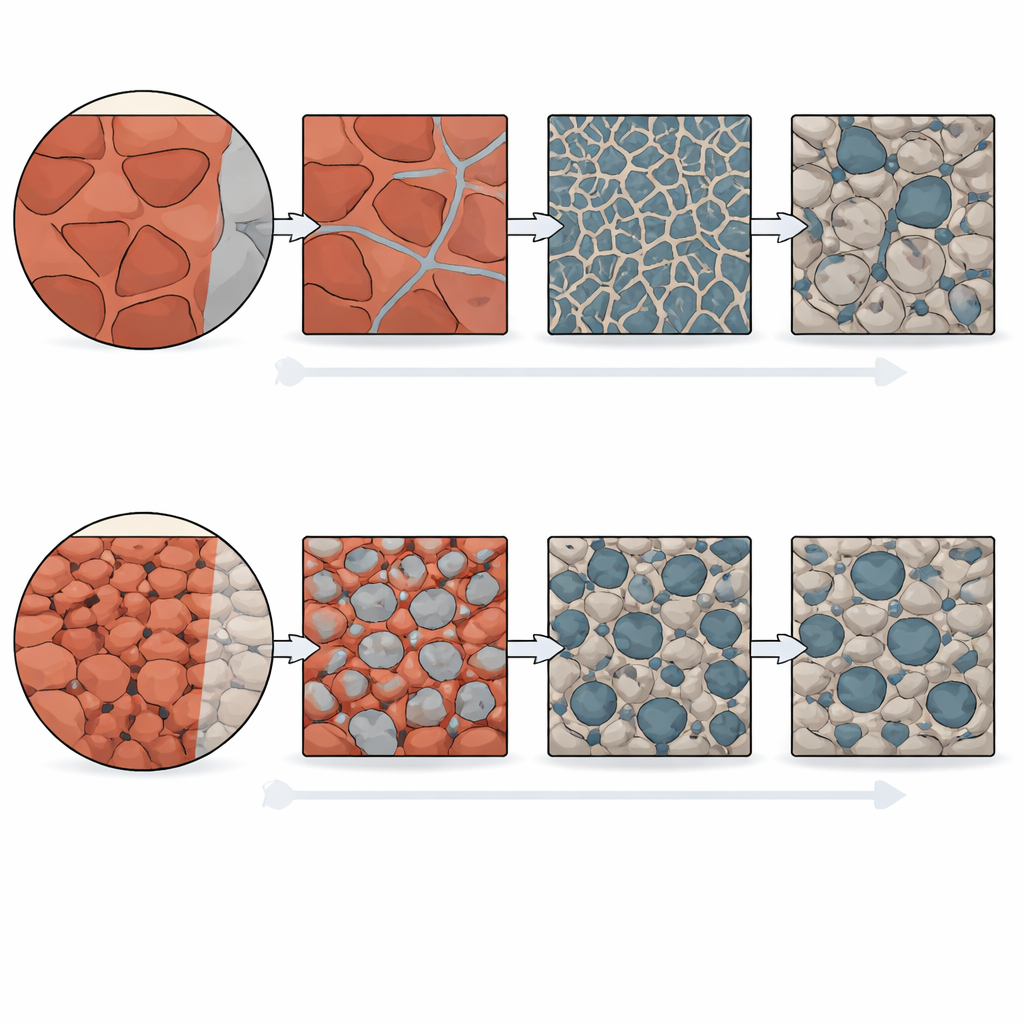
Peering inside partially reduced samples with advanced electron microscopy revealed why. As hematite transforms step-by-step into other iron oxides and then iron, it spontaneously develops pores—tiny empty spaces that act as highways for hydrogen to come in and water vapor to escape. In the large-grained material, these pores first appear as extremely fine, straight channels threading through the intermediate oxide layer. They are highly directional and well connected, which helps the reduction front advance quickly at early stages. However, because there are few grain boundaries and the grains are big, the resulting pore network is uneven from place to place. Some regions end up with very narrow channels and dense iron around trapped oxide pockets, which slow down the final clean-up of remaining oxygen.
Smoother Networks in Ultrafine Grains
By contrast, the ultrafine-grained hematite behaves differently. The multitude of tiny grains, each with its own crystal orientation, disrupts the formation of long, straight nano-channels. Instead, somewhat larger, more rounded pores form that are spread more evenly through the material. This network is less sharply directional but more uniform, leading to fewer isolated islands of oxide locked inside dense iron. During the slower, late-stage steps—when oxygen must move through solid metal—this more regular pore structure gives hydrogen and water vapor better access, allowing the reaction to proceed more completely. The team even identified a threshold grain size, between about 5 and 10 micrometers, above which the narrow, straight channels characteristic of large grains begin to appear.
Beyond Steel: Designing Better Porous Materials
Together, the experiments and imaging show that simply changing the initial grain size of iron oxide can tilt the balance between a fast early reduction and an efficient final clean-up. Large grains favor a rapid start by building very fine, directional gas pathways, but they also foster a patchy pore network that traps unreduced oxide. Ultrafine grains slow the initial advance but generate a more even, coarser pore system that helps the reaction reach a higher degree of completion. For green steel, this means that tuning grain size offers a powerful lever to balance speed and efficiency in hydrogen-based processes. More broadly, the insights apply to any technology that relies on controlled porosity in iron oxides—from batteries and fuel systems to materials for capturing carbon—where the invisible architecture of grains and pores can make all the difference in performance.
Citation: Ratzker, B., Ruffino, M., Shankar, S. et al. Influence of grain size on the solid-state direct reduction of polycrystalline iron oxide. Commun Mater 7, 82 (2026). https://doi.org/10.1038/s43246-026-01106-z
Keywords: hydrogen direct reduction, green steel, iron oxide microstructure, grain size effects, porous materials