Clear Sky Science · en
Enhanced reversible barocaloric effect at low pressure in neopentyl plastic crystal solid solutions
Making Cooling Cleaner and Greener
Air conditioners and refrigerators keep us comfortable, but they typically rely on gases that can leak and warm the planet. Scientists are exploring solid materials that can cool things when squeezed, offering a way to build compact, efficient fridges without harmful refrigerants. This paper shows how carefully mixing three simple organic molecules—relatives of sugar alcohols—creates a new solid that cools efficiently under relatively low pressures and works more reliably than earlier candidates.
How Squeezable Solids Can Replace Cooling Gases
Certain solids warm up when you compress them and cool down when you release the pressure. This behavior, known as the barocaloric effect, can be harnessed to move heat in much the same way that conventional refrigerators use the compression and expansion of gases. One especially promising material is neopentyl glycol (NPG), a small organic molecule that forms a “plastic crystal” in which molecules can reorient like spinning tops. When NPG switches between a more ordered and a more disordered state, it exchanges a large amount of heat, making it attractive for solid-state cooling. However, its transition temperature and the high pressures needed for reliable operation make it difficult to use in practical devices.
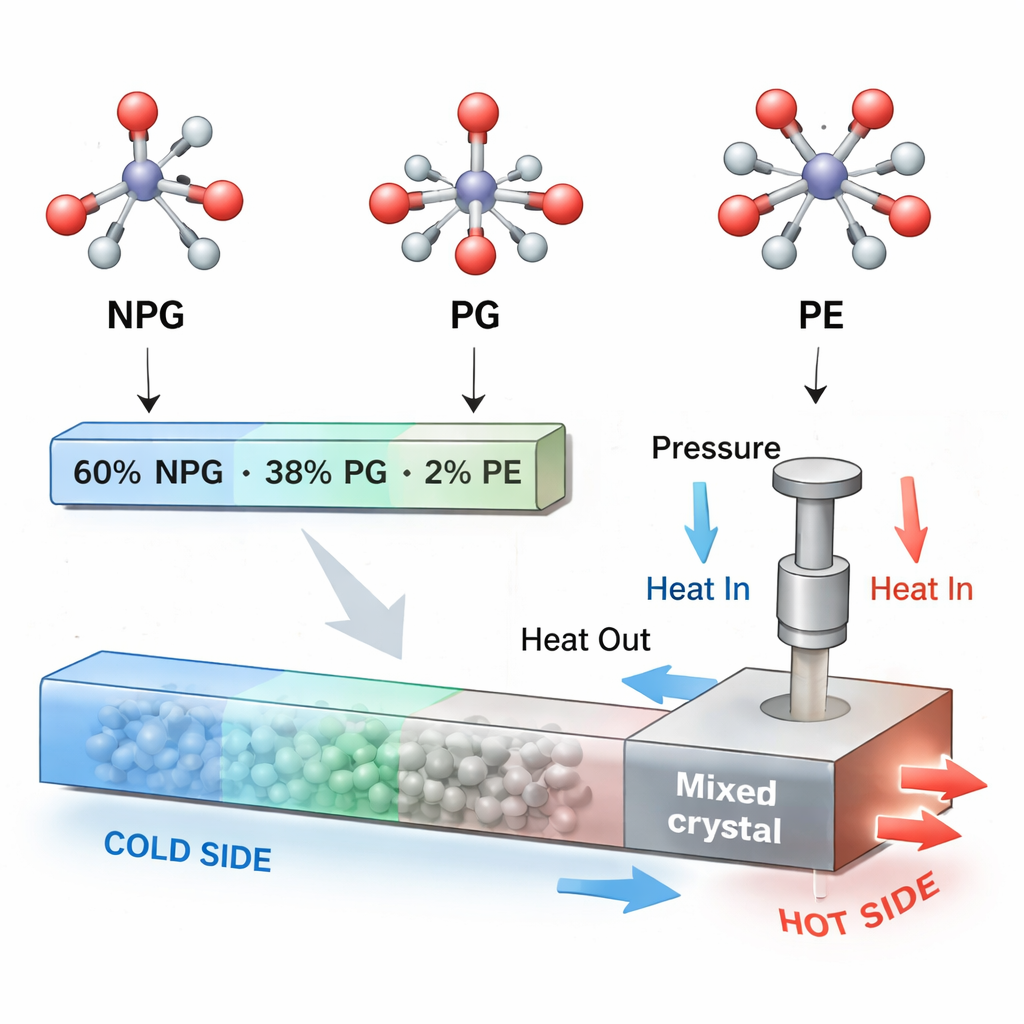
Blending Simple Molecules to Tune Performance
The researchers tackled this problem by mixing NPG with two closely related molecules, pentaglycerine (PG) and pentaerythritol (PE). All three have similar tetrahedral shapes but carry different numbers of hydroxyl (–OH) groups, which control how molecules lock together via hydrogen bonds in the solid. By starting with a 60:40 mixture of NPG and PG and then adding just 2% PE, they created a stable “ternary” solid solution that still shows a colossal barocaloric effect, but now at a more useful temperature and under moderate pressure. The key achievement is that the heat-exchange process becomes much more reversible: compared with pure NPG at the same pressure, the new mixture delivers about seven times more useful, repeatable cooling power, over a temperature window around twenty times wider.
What Happens Inside the Material When It Works
To understand why such a small compositional tweak has a big impact, the team probed both the structure and motion inside the crystals. Synchrotron X-ray diffraction revealed that, as the material is heated, it gradually transforms from a neat, layered crystal into a more symmetric, highly disordered plastic crystal. In the ternary mixture, this transition stretches over roughly 30 degrees Celsius, with both phases coexisting over a broad range. This extended coexistence softens the transition, reducing the sharp “start–stop” behavior that causes hysteresis and energy loss in simpler materials. The extra PE molecules subtly distort the hydrogen-bond network, particularly in certain crystallographic directions, which appears to make it easier for regions of the new phase to start forming and to grow.
Watching Heat Spots and Molecular Motions
Infrared cameras showed how the phase change spreads through the samples as they cool. Pure NPG tends to switch in a few long, needle-like fronts, while the mixed crystals show many tiny, scattered hot spots that flicker on and off. This indicates a much higher density of nucleation sites, where the new phase can start, and explains the smoother, more gradual transition. Neutron scattering experiments, which are sensitive to how hydrogen atoms move, further revealed that the energy barriers for key molecular rotations in the ternary blend are up to 50% lower than in pure NPG. In other words, the molecules in the mixed crystal can begin reorienting—and thus storing or releasing heat—more easily and at lower energetic cost, supporting efficient, low-pressure operation.
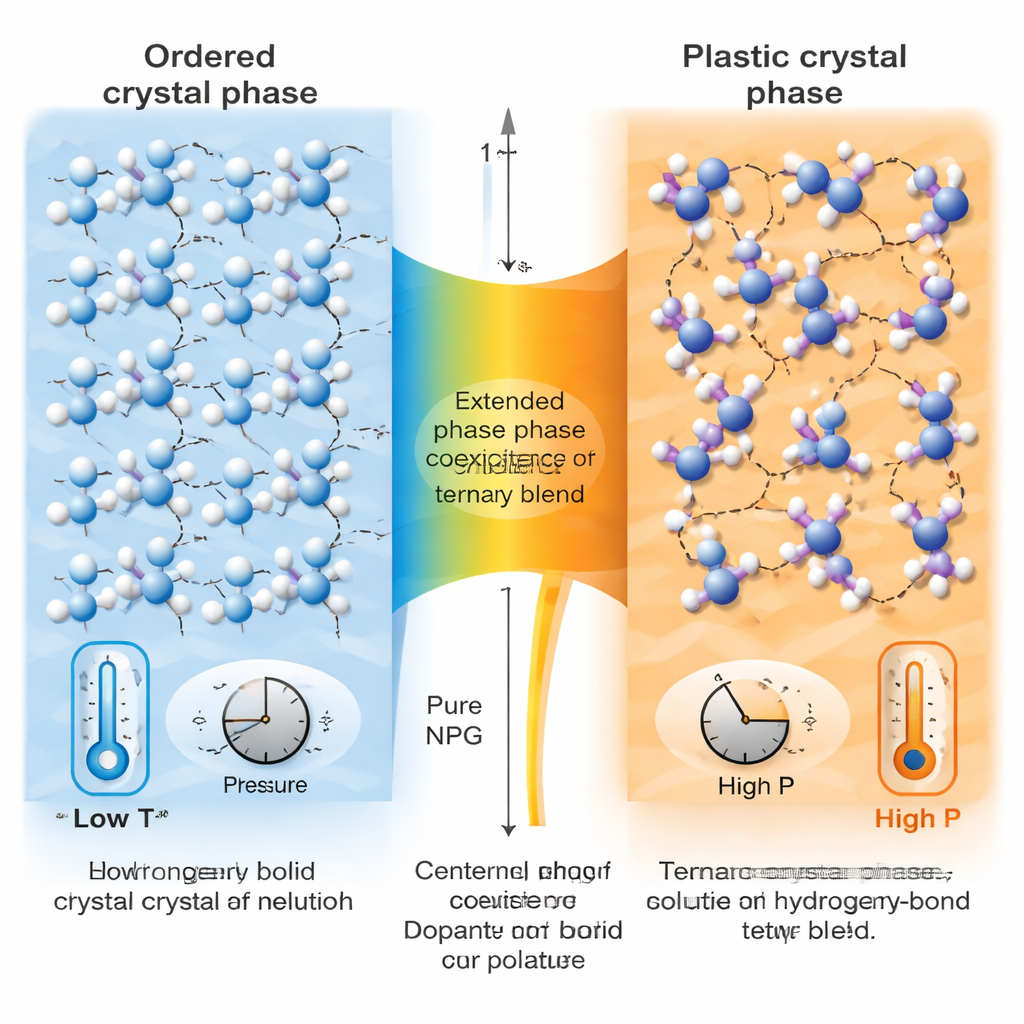
Why This Matters for Future Solid-State Fridges
In simple terms, this work shows that by blending and lightly “doping” closely related molecules, scientists can tame an otherwise temperamental cooling material, making it more reliable and efficient under realistic pressures. The new 60:38:2 NPG–PG–PE mixture keeps the strong cooling punch of NPG but widens the useful temperature range and dramatically improves reversibility, boosting the practical cooling capacity by about a factor of seventy at one kilobar of pressure. Because there are many families of similar plastic crystals and related molecular solids, this compositional design strategy could guide the development of next-generation, climate-friendly solid-state refrigerators and heat pumps.
Citation: Rendell-Bhatti, F., Dilshad, M., Beck, C. et al. Enhanced reversible barocaloric effect at low pressure in neopentyl plastic crystal solid solutions. Commun Mater 7, 72 (2026). https://doi.org/10.1038/s43246-026-01084-2
Keywords: barocaloric cooling, plastic crystals, solid-state refrigeration, hydrogen-bond networks, neopentyl glycol blends