Clear Sky Science · en
Non-resonant plasmon energy transfer processes for catalysis
Lighting Up Chemistry in a New Way
Chemists have long dreamed of copying plants by using light to power chemical reactions cleanly and efficiently. Yet many of the light-absorbing molecules used today are fragile, expensive, and picky about which reactions they can drive. This article explores a different strategy: using tiny gold particles as durable “antennae” for light, able to hand off energy to ordinary catalysts and even to simple molecules, opening paths to greener and more versatile chemical manufacturing.
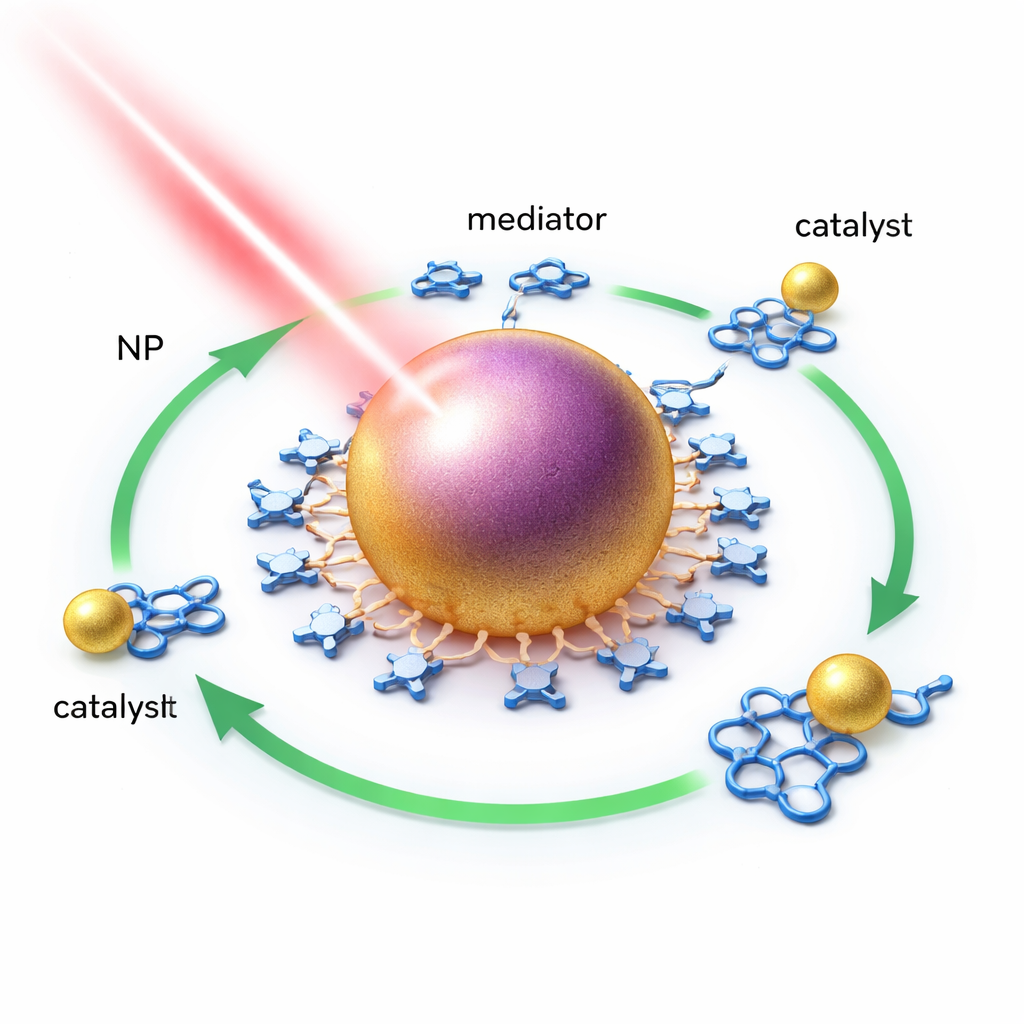
Why Tiny Gold Particles Matter
When very small pieces of gold are hit by light, the electrons inside them slosh back and forth in unison, a behavior known as a plasmon. This motion concentrates light energy into a tiny volume and briefly creates very energetic electrons and holes, often called “hot carriers.” Traditionally, to pass this energy to nearby molecules, the color of the light, the metal particle, and the molecule all had to be closely matched—like tuning a radio to just the right station. That strict matching has limited which catalysts and reactions could benefit from plasmon effects.
A Detour Around Energy Matching
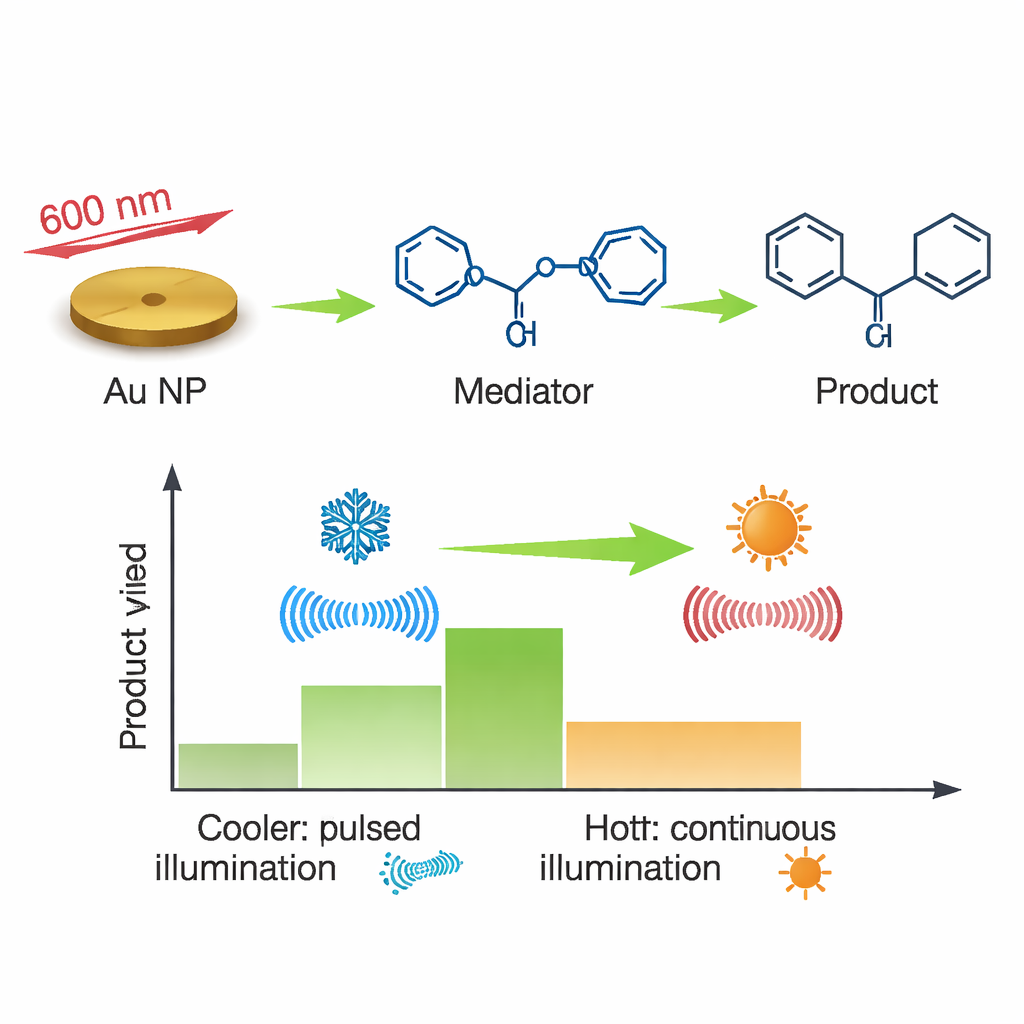
The researchers show that gold nanoparticles can bypass this tuning requirement by using an indirect, two-step handoff of energy. First, they attach a simple organic molecule, 1-naphthoic acid, to the nanoparticle surface. This “mediator” is chosen so that its excited state sits at just the right energy to pass power along to a specially designed gold catalyst complex. When the nanoparticles absorb light, they can transfer energy to the mediator, which then transfers it to the catalyst. Crucially, this works even with light that is too weak to excite the mediator or catalyst directly—evidence of a new, non-resonant route for moving energy.
Watching Energy Move, Frame by Frame
To prove that this handoff really occurs, the team used ultrafast spectroscopy, a kind of high-speed camera for electronic states. They first recorded the distinctive “fingerprint” of the catalyst in its energized form, a long-lived but non-glowing triplet state. Then they showed that this same fingerprint appears when light is absorbed by the iridium dye often used in photocatalysis and—more strikingly—when gold nanoparticles are excited instead. By carefully comparing how signals fade in the presence and absence of oxygen, they confirmed that the catalyst’s triplet state is indeed being formed and that its lifetime shortens when energy can leak back toward the nanoparticle or into oxygen.
Driving a Real Chemical Reaction
To move beyond spectroscopy, the authors tested whether this energy transfer could make an actual product. They chose a classic light-driven reaction: joining two styrene molecules into a four-membered ring called 1,2-diphenylcyclobutane. On their own, the gold particles, mediator, and styrene do nothing under red light. But when gold nanoparticles coated with the mediator are illuminated at a wavelength too low to excite the reactants directly, a small amount of the cyclobutane product appears. Adjusting the light to reduce local heating boosts the yield several-fold, matching the idea that brief, controlled bursts of energy, not overall warming, are responsible. This demonstrates that the non-resonant plasmon pathway can genuinely power bond-forming chemistry.
A New Platform for Light-Driven Catalysts
In simple terms, the study shows that gold nanoparticles can act like rugged solar antennas that collect light and funnel its energy, through a mediator, into otherwise unresponsive gold catalysts and even simple molecules. Because this mechanism does not require a perfect color match between light, particle, and catalyst, it greatly widens the menu of reactions that could be run with light instead of heat or harsh reagents. Over time, such plasmon-based energy transfer schemes could help chemists design more sustainable, tunable processes for making medicines, materials, and other high-value products, all by putting tiny pieces of gold to work as nanoscale power lines for light.
Citation: Andreis, A., Herrera, J., Mouriès-Mansuy, V. et al. Non-resonant plasmon energy transfer processes for catalysis. Commun Mater 7, 68 (2026). https://doi.org/10.1038/s43246-026-01077-1
Keywords: plasmonic catalysis, gold nanoparticles, energy transfer, photochemistry, light-driven reactions