Clear Sky Science · en
High-performance ionomer-free gas diffusion cathodes with low Pt loading for proton exchange membrane water electrolysis
Turning Water into Fuel with Less Precious Metal
Hydrogen made from water and renewable electricity is often promoted as a clean fuel for heavy industry, shipping and long-term energy storage. But today’s most efficient devices for splitting water rely on large amounts of platinum, one of the rarest and most expensive metals on Earth. This study shows a way to use almost one hundred times less platinum on one side of these devices, without sacrificing performance or stability, bringing affordable green hydrogen a step closer.
Why Cutting Platinum Use Matters
Modern proton exchange membrane water electrolyzers—compact devices that split water into hydrogen and oxygen—work well but lean heavily on two scarce “noble” metals. Iridium drives the oxygen-forming reaction at the anode, while platinum powers hydrogen formation at the cathode. Even though platinum is a superb catalyst in the lab, in real devices much of it sits buried in a thick layer mixed with a plastic-like ion-conducting binder. Only a fraction of the metal actually touches water, gas and membrane at the same time, which is required for the reaction to proceed. As a result, manufacturers compensate by adding more platinum, increasing both cost and material demand.
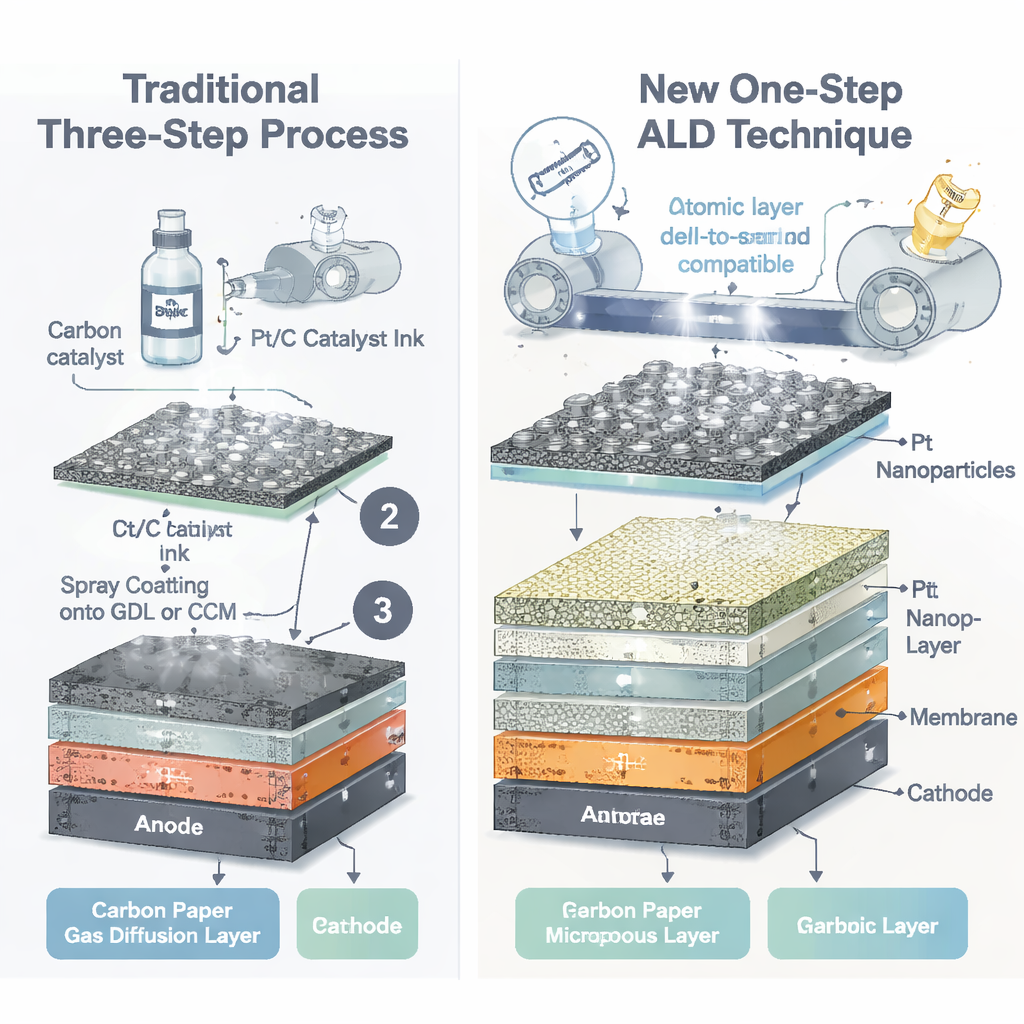
A One-Step Way to Place Atoms Precisely
The researchers tackled this problem by rethinking how the cathode is built. Instead of making a liquid ink and spraying it onto a support, they used a gas-based technique called atomic layer deposition (ALD). In ALD, the surface is exposed to alternating pulses of a platinum-containing vapor and a reactive gas, allowing platinum to grow as tiny, well-separated nanoparticles layer by layer. They applied this process directly onto a commercial gas diffusion layer—a porous carbon sheet that lets water and gas flow—optionally coated with an extra thin “microporous layer” that smooths the surface. By tuning the number of ALD cycles, they could control both how many platinum atoms landed and how large the particles became, all with nanometer precision.
Building a Thinner, Smarter Cathode Layer
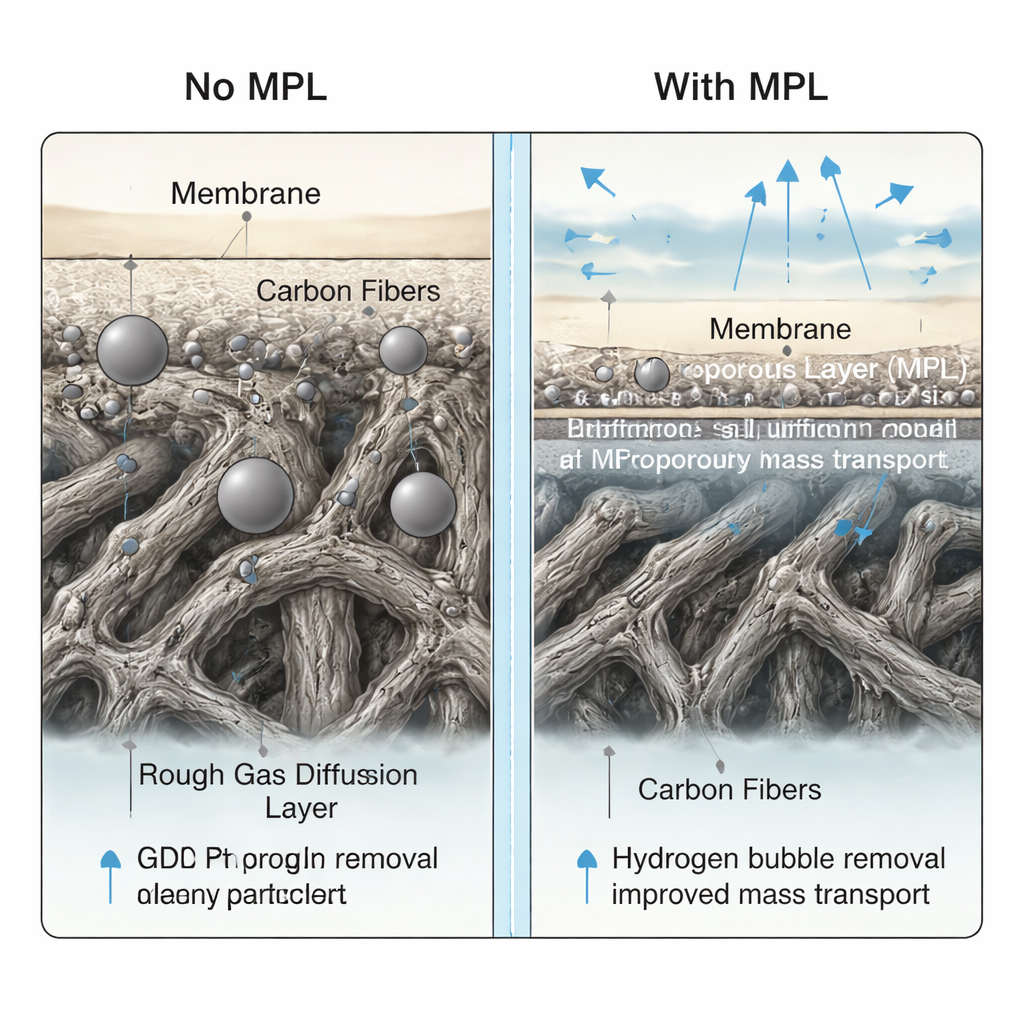
Careful imaging and surface analysis confirmed that the ALD method produced uniform platinum nanoparticles sitting mainly at the outer surface of the support rather than soaking deep inside. On the microporous layer, the particles were particularly small and evenly spread, with sizes often below two nanometers at the lowest metal loading. Because this thin, smooth layer makes good contact with the polymer membrane while remaining water-repellent, it helps hydrogen bubbles escape and keeps fresh water flowing to active sites. Electrical tests in full electrolyzer cells showed that these new cathodes, even with extremely low platinum amounts between about 1 and 5 micrograms per square centimeter, could match or outperform commercial reference electrodes whose platinum content is more than one hundred times higher.
Performance, Efficiency and Staying Power
To understand how and why the new design works so well, the team dissected the cell voltage into contributions from reaction speed, electrical resistance and gas transport. They found that when platinum was placed on a microporous layer using ALD, the speed of the hydrogen-forming reaction remained comparable to that of conventional, platinum-rich electrodes, despite the drastically lower metal content. At the same time, the thinner, well-organized catalyst region reduced problems with gas build-up that can otherwise waste energy. When the researchers normalized performance by the actual platinum mass, the advantage became stark: their best ionomer-free cathodes showed mass activities up to three orders of magnitude higher than standard commercial devices and outperformed the best results reported so far in the scientific literature.
Proving Durability Under Realistic Conditions
Using less platinum is only meaningful if the device remains stable during long-term use and under the fluctuating power typical of wind and solar energy. The team therefore ran their best electrodes for 200 hours at a high current, equivalent to industrially relevant hydrogen production rates. The cell voltage remained nearly constant, with only minute degradation. In a separate test that mimicked rapid swings in power input—by cycling the cell voltage between low and high values for 25,000 cycles—the electrodes again showed only tiny performance losses. Electrical measurements before and after these tests indicated that neither the intrinsic activity of the platinum nor the overall resistance of the cell changed appreciably.
What This Means for Green Hydrogen
In simple terms, this work shows how to “use every atom wisely.” By placing platinum exactly where it is needed, in a very thin layer at the interface between a smooth porous support and the membrane, the researchers achieve the same hydrogen output with roughly 99.5% less platinum on the cathode than today’s commercial designs. Because the ALD process can be adapted to roll-to-roll production, similar to how newspapers are printed, it offers a realistic path toward mass manufacturing. If combined with parallel efforts to reduce iridium use at the anode, such advances could make large-scale, efficient green hydrogen production both technically and economically feasible.
Citation: Chen, M., Piechulla, P.M., Mantzanas, A. et al. High-performance ionomer-free gas diffusion cathodes with low Pt loading for proton exchange membrane water electrolysis. Commun Mater 7, 67 (2026). https://doi.org/10.1038/s43246-026-01076-2
Keywords: green hydrogen, water electrolysis, platinum catalyst, atomic layer deposition, gas diffusion electrode