Clear Sky Science · en
Neoadjuvant PD-1 blockade in surgically resectable desmoplastic melanoma: cohort A of the phase 2 SWOG S1512 trial
Why this matters for people facing skin cancer
For many people, the word “melanoma” already sounds frightening. A rare form called desmoplastic melanoma can be especially difficult to treat because it often grows deep into sun‑damaged skin on the head and neck of older adults, making surgery disfiguring and complex. This study tested whether giving an immune‑boosting drug called pembrolizumab before surgery could shrink these tough tumors, make operations easier and safer, and improve long‑term outcomes.
A stubborn skin cancer in hard‑to‑treat places
Desmoplastic melanoma usually appears on skin that has seen years of intense sun, such as the scalp, face and neck. Instead of forming a dark mole, it often looks skin‑colored and blends into surrounding tissue. Under the microscope, it threads between nerves and deep structures, so surgeons may need to remove large areas of skin and underlying tissue to be confident all cancer is gone. Because these tumors often occur in older, more fragile patients, the side effects of big operations and radiation can seriously affect appearance, speech, vision or even the ability to chew and swallow.
Using the immune system before the scalpel
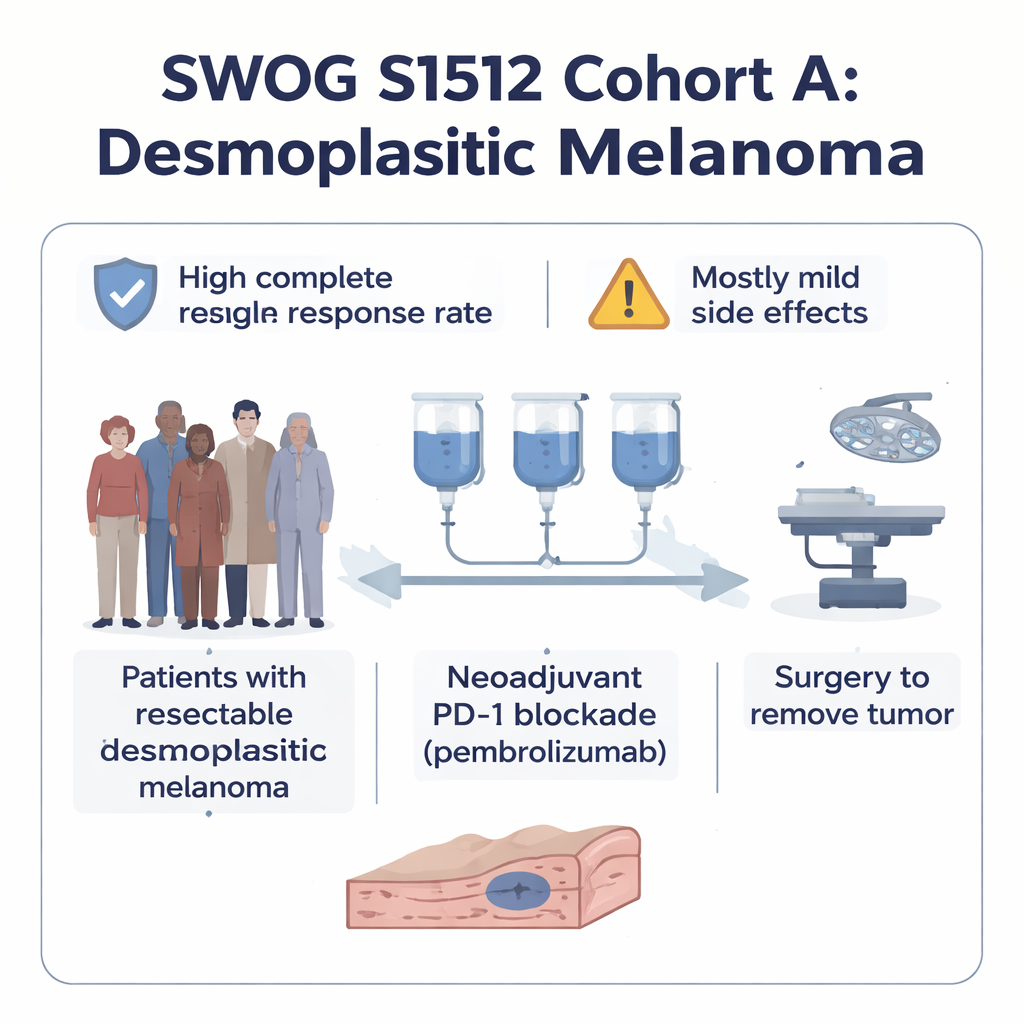
Scientists have learned that desmoplastic melanoma carries an unusually high number of DNA mutations caused by ultraviolet light, which can make it highly visible to the immune system. Earlier work suggested that drugs blocking PD‑1—a molecular “brake” that restrains immune cells—spark strong responses in advanced, inoperable cases. The SWOG S1512 trial asked whether the same approach, given before surgery in earlier‑stage disease, could wipe out much of the tumor in advance. In this trial, 28 people with resectable desmoplastic melanoma received three intravenous doses of pembrolizumab, each three weeks apart, followed by surgery to remove any remaining cancer.
What the trial found in the tumor samples
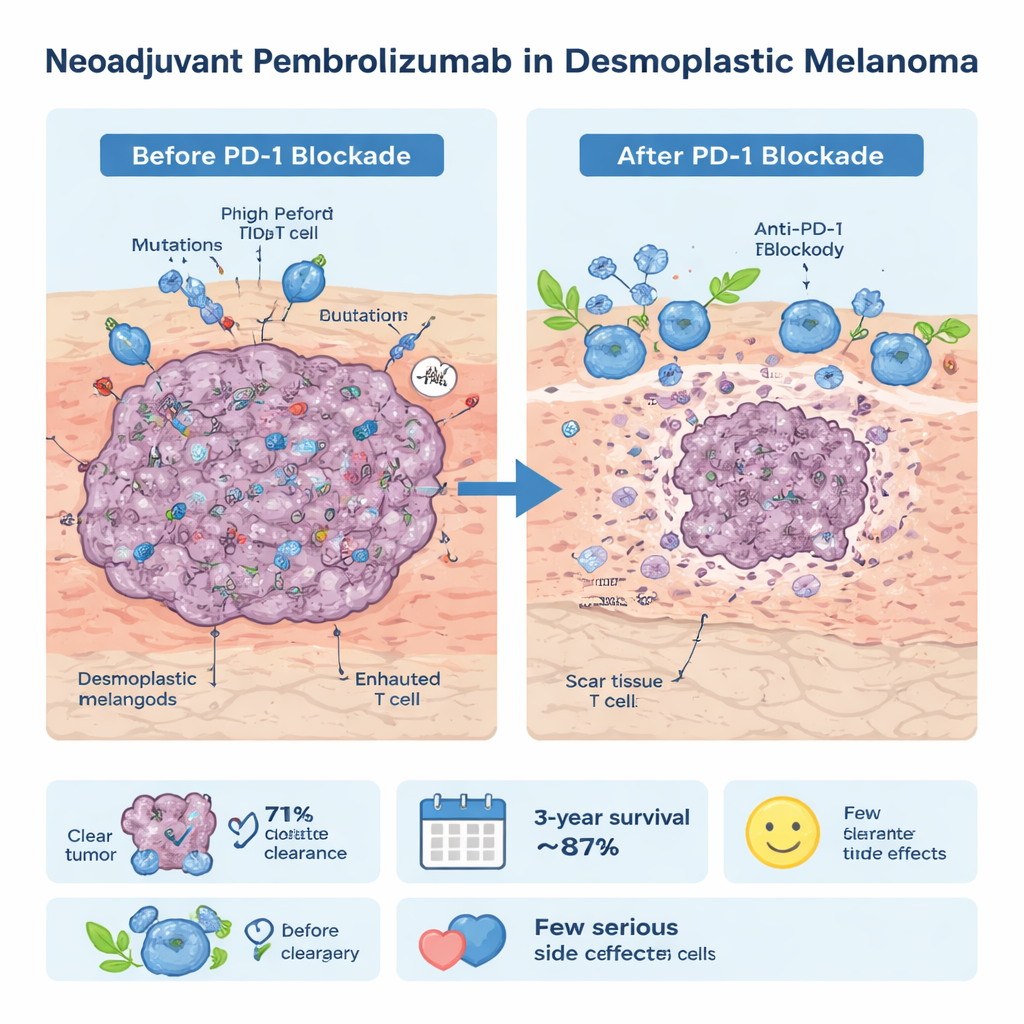
When surgeons later removed the tumors, pathologists carefully examined the tissue. In 20 of the 28 patients—about 71 percent—no living cancer cells could be found in the surgical specimen, a result called a pathological complete response. Central reviewers who re‑checked many of these samples confirmed a very similar rate of near‑complete or complete tumor destruction. Importantly, this success rate was far higher than what has been seen when PD‑1 drugs are used in the same way for more common types of melanoma. The benefit appeared in both “pure” desmoplastic tumors and those mixed with other melanoma cell types, suggesting the strategy is broadly useful within this rare subtype.
Safety, side effects and long‑term outlook
The immune treatment was generally well tolerated in this older group of patients, whose median age was 75. Most side effects, such as fatigue, rash or mild diarrhea, were manageable. Only two people (7 percent) had more serious treatment‑related problems, like significant mouth sores or inflammation of the colon, but both still went on to successful surgery. After a median follow‑up of three and a half years, three‑year overall survival was estimated at 87 percent, and no deaths were clearly linked to melanoma or to the treatment itself. Only a handful of patients saw their cancer return, and one person who achieved a complete visible response chose to skip surgery entirely and has remained free of relapse so far.
What this could mean for future care
These findings suggest that a short course of pembrolizumab before surgery can eradicate desmoplastic melanoma in the majority of patients, while causing relatively few serious side effects. For people who might otherwise need large, disfiguring operations or repeated procedures to chase persistent tumor, shrinking or eliminating the cancer first could allow smaller surgeries and reduce the need for radiation. Although larger studies and longer follow‑up are still needed, this work points toward a future in which carefully timed immune therapy makes this rare but challenging skin cancer both more curable and less devastating to live through.
Citation: Kendra, K.L., Bellasea, S.L., Eroglu, Z. et al. Neoadjuvant PD-1 blockade in surgically resectable desmoplastic melanoma: cohort A of the phase 2 SWOG S1512 trial. Nat Cancer 7, 272–282 (2026). https://doi.org/10.1038/s43018-025-01113-y
Keywords: desmoplastic melanoma, pembrolizumab, immunotherapy, neoadjuvant therapy, PD-1 blockade