Clear Sky Science · en
CAR-adapted PIK3CD base editing enhances T cell anti-tumor potency
Reprogramming Cancer-Fighting Cells
Chimeric antigen receptor (CAR) T cell therapy has transformed the treatment of some blood cancers, yet many patients still relapse or never respond. One key problem is that these engineered immune cells can burn out or fail to persist in the body long enough. This study asks a simple but powerful question: instead of completely redesigning CARs, what if we could subtly “retune” the internal wiring of T cells so they stay powerful for longer, and do so safely?
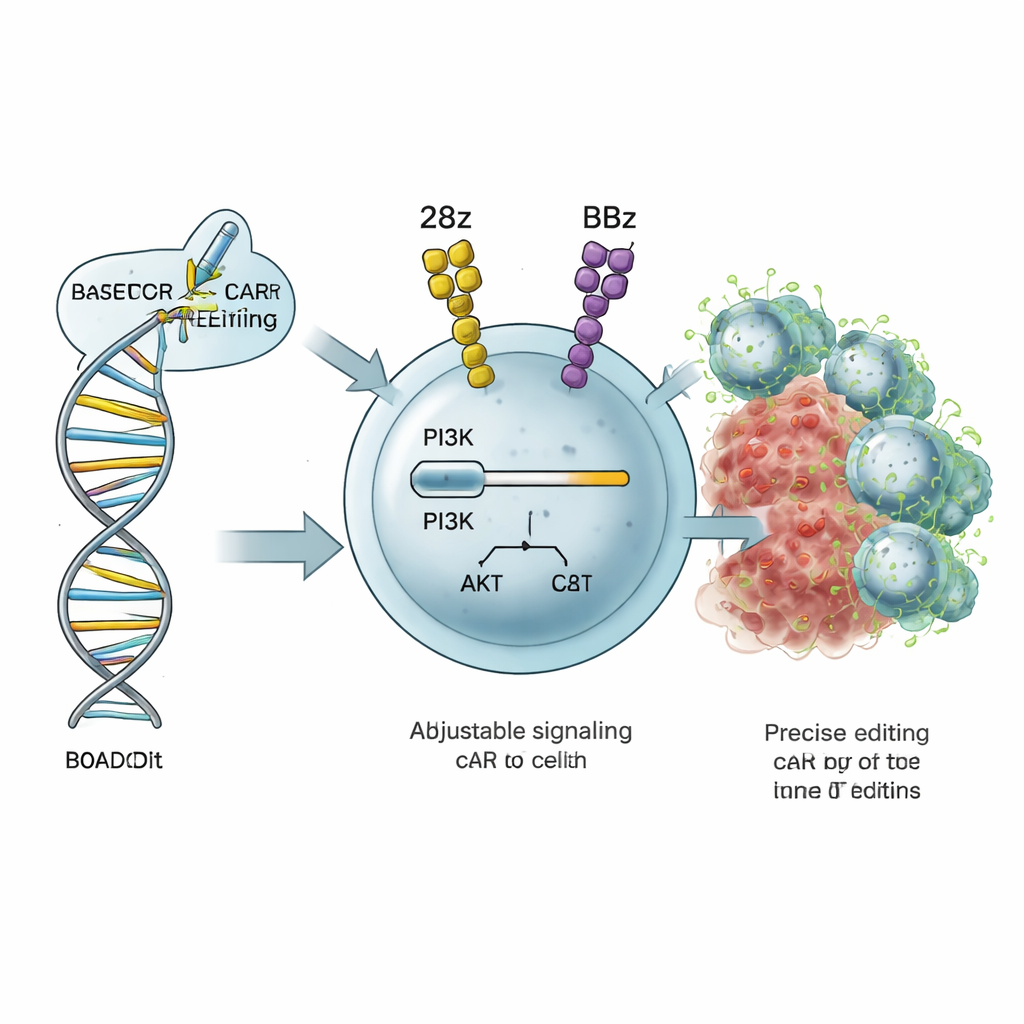
Aiming the T Cell’s Volume Knob
Cancer-killing T cells rely on internal signaling circuits that tell them how strongly to activate, how fast to divide and whether to become short-lived fighters or long-lived memory cells. A major circuit is the PI3K–AKT pathway, in which a protein called PI3Kδ—encoded by the PIK3CD gene—acts like a volume knob for activation and metabolism. The authors developed a strategy they call ROADSTAR, which uses base editing, a highly precise form of CRISPR genome engineering, to change single “letters” in PIK3CD. Instead of turning the pathway fully on or off, ROADSTAR searches for tiny mutations that gently increase or decrease signaling in a way that best matches the specific CAR design inside the T cell.
Discovering Helpful Mutations
The team built a library of 34 different single-letter changes in the regulatory region of PIK3CD and introduced them into human T cells already carrying one of two common CAR designs: 28z (based on the CD28 molecule) and BBz (based on 4‑1BB). These CARs differ in how they stimulate T cells: 28z tends to drive strong, rapid attack but also exhaustion, whereas BBz fosters better survival and memory but can be less aggressive. The edited CAR T cells were put through a “stress test” by repeatedly exposing them to leukemia cells. Using deep sequencing, the researchers tracked which mutations allowed CAR T cells to outcompete their unedited peers over several weeks.
Two mutations clearly stood out. In BBz CAR T cells, a change called E81K slightly boosted PI3Kδ activity, increasing signals down the PI3K–AKT pathway. In 28z CAR T cells, a different change, L32P, dampened PI3Kδ activity. Structural modeling suggested both edits subtly alter how PI3Kδ interacts with its regulatory partner, fine‑tuning the strength of signaling rather than switching it fully on or off. Importantly, the beneficial mutation for one CAR design was not the same as for the other, underscoring that each CAR architecture demands its own optimal internal tuning.
Stronger, Fitter and Longer-Lasting CAR T Cells
When the promising edits were tested individually, E81K made BBz CAR T cells more activated upon seeing tumor cells, better at proliferating and markedly more efficient at killing cancer targets, including those with low antigen levels. These cells developed into effector memory T cells that combined robust killing power with the ability to persist. In mouse models of leukemia and metastatic neuroblastoma, E81K‑edited BBz CAR T cells controlled tumors more completely and for far longer than unedited BBz cells, even defeating multiple rounds of tumor rechallenge. Detailed single‑cell RNA and metabolic profiling showed that E81K cells had enhanced mitochondrial function, higher spare respiratory and glycolytic capacity and fewer signs of exhaustion—essentially, they were more energetic and resilient.
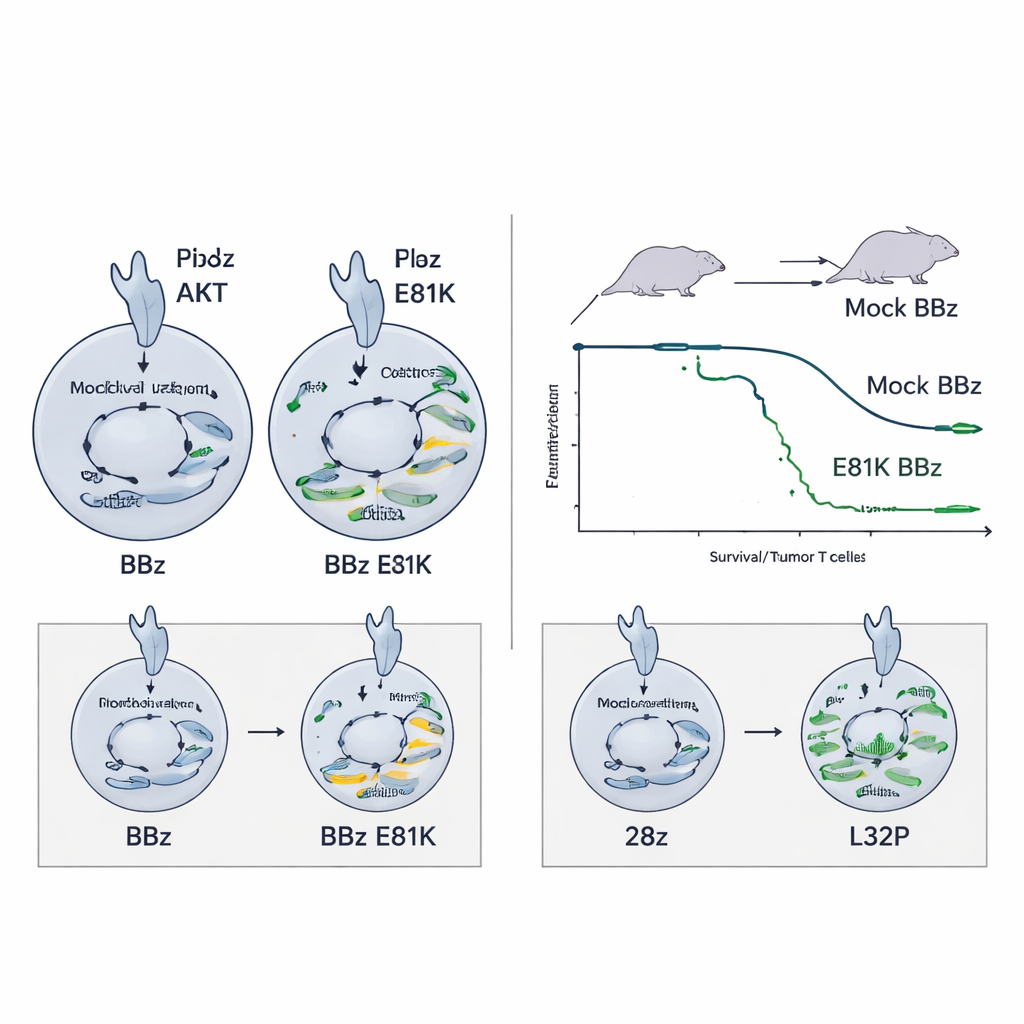
Preventing Burnout in Highly Active CARs
The same E81K boost did not help—and could even harm—28z CAR T cells, which already signal strongly. In that setting, extra PI3K activity increased markers of exhaustion without improving function. Here, the L32P mutation proved better suited: by modestly dialing down PI3K–AKT signaling, L32P‑edited 28z CAR T cells showed reduced baseline activation but preserved high killing capacity and proliferated more over time. They also maintained a less differentiated, memory‑like state, including a stem‑cell‑like memory pool that is thought to sustain long‑term responses. In a demanding neuroblastoma model, L32P‑modified 28z CAR T cells improved survival compared to their unedited counterparts.
Safety and Future Promise
A central concern with permanently altering immune signaling is the risk of uncontrolled growth or secondary cancers. The authors therefore performed extensive safety checks. E81K‑edited BBz CAR T cells did not grow abnormally in culture, did not cause organ damage or inflammatory crises in mice and showed no signs of lymphoma or tissue invasion months after infusion. Clinical databases likewise show no enrichment of these specific PIK3CD mutations in T cell leukemias. Overall, the work demonstrates that carefully chosen single‑letter genome edits can act as precision dials for T cell behavior—boosting anti‑tumor power and staying power while respecting each CAR’s unique biology. This ROADSTAR approach could help craft the next generation of CAR T therapies that are both more effective and more reliable across blood cancers and, potentially, solid tumors.
Citation: Bucher, P., Brückner, N., Kortendieck, J. et al. CAR-adapted PIK3CD base editing enhances T cell anti-tumor potency. Nat Cancer 7, 368–383 (2026). https://doi.org/10.1038/s43018-025-01099-7
Keywords: CAR T cells, PI3K signaling, base editing, cancer immunotherapy, T cell metabolism