Clear Sky Science · en
De novo H3.3K27M-altered diffuse midline glioma in human brainstem organoids to dissect GD2 CAR T cell function
Why this childhood brain cancer study matters
Diffuse midline glioma is one of the deadliest childhood brain cancers, striking very young children in the brainstem and leaving families with almost no effective treatment options. Because tumors sit deep in the brain and are rarely operated on, researchers have little tissue to study and few ways to test new therapies safely. This study builds a realistic “mini–brainstem in a dish” and uses it to watch how a promising immunotherapy—GD2 CAR T cells—behaves over weeks, including why it sometimes fails.
Building a mini brainstem in the lab
The team started with human stem cells and guided them, step by step, to grow into three-dimensional brain organoids that resemble the brainstem region called the pons, where these tumors usually arise. By carefully timing growth signals, they produced organoids rich in the same kinds of support cells, or glia, that normally live in this area. Detailed genetic analyses showed that the cell types and their maturation followed patterns seen in early human brain development, especially the glial lineages typical of the pons and neighboring medulla. This meant the organoids could serve as a realistic backdrop for studying how this cancer starts and grows.
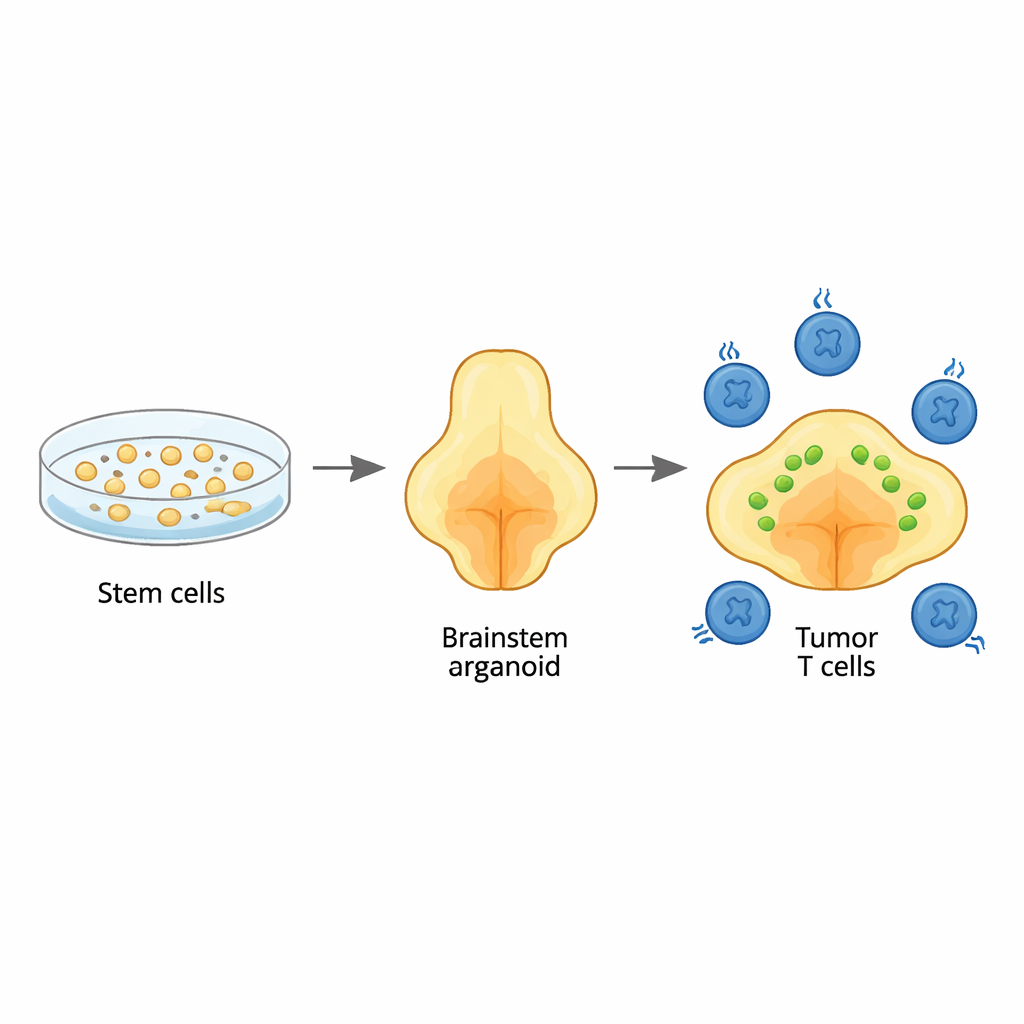
Recreating the child’s tumor inside the organoid
To mimic diffuse midline glioma, the researchers inserted three key genetic changes into young organoids: a hallmark mutation in a histone protein (H3.3K27M), loss of the tumor suppressor TP53, and an activating change in the growth receptor PDGFRA. When introduced at a very early developmental stage, these alterations turned select glial-like cells cancerous. The resulting tumors spread diffusely through the organoids, just as they do through children’s brainstems, and carried the same broad DNA and RNA signatures seen in patient tumors. Single-cell sequencing revealed a mix of cancer cell states, including immature, pons-specific glial cells that match those found in actual pediatric tumors, underscoring that the model faithfully recapitulates the human disease.
Watching immune therapies fight—and tire—over time
With this tumor-in-a-brainstem model in place, the team added GD2 CAR T cells—patients’ T cells engineered to recognize a molecule called GD2 on tumor cells—and followed both tumor size and T cell behavior for up to a month. Some organoids showed strong tumor shrinkage, others only partial control, mirroring the mixed outcomes seen in clinical trials. Single-cell RNA sequencing of more than 20,000 CAR T cells exposed to the tumors uncovered a surprising diversity of T cell states. Some cells were highly cytotoxic “hit men,” others were activated but not fully lethal, and a distinct group displayed hallmarks of exhaustion, with dampened cancer-killing genes and high expression of inhibitory “brakes.” The exhausted cells closely resembled worn-out T cells seen in chronic infections and human tumors, indicating that the organoid system captures clinically relevant T cell fatigue driven by ongoing contact with cancer.
Finding powerful but short-lived T cells
Digging deeper, the researchers identified a subset of CAR T cells marked by a surface protein called NCAM1 that were especially potent killers in the short term. When these NCAM1-positive cells were isolated and used alone, they controlled tumors more strongly at first than their NCAM1-negative counterparts. However, they also became exhausted and dwindled more quickly, leading to weaker control over time. This trade-off between immediate power and persistence helps explain why CAR T therapies can produce dramatic but temporary responses and suggests that pre-selecting or reshaping T cell subsets could optimize treatment durability.
How brain immune cells can blunt therapy
In the real brain, tumors are surrounded not only by neurons and glia but also by microglia, the brain’s resident immune cells. To capture this layer of complexity, the scientists added microglia derived from stem cells into their organoids. These cells matured, adopted shapes and gene expression patterns specific to microglia found in diffuse midline glioma, and produced molecules associated with an immunosuppressive environment. When CAR T cells were added to tumor-bearing organoids that contained microglia, the T cells shifted further toward exhausted and low-effector states, and tumor control worsened. At the same time, inflammatory factors linked to treatment side effects, such as IL-6, rose, suggesting the model can also help study CAR T–related toxicities.
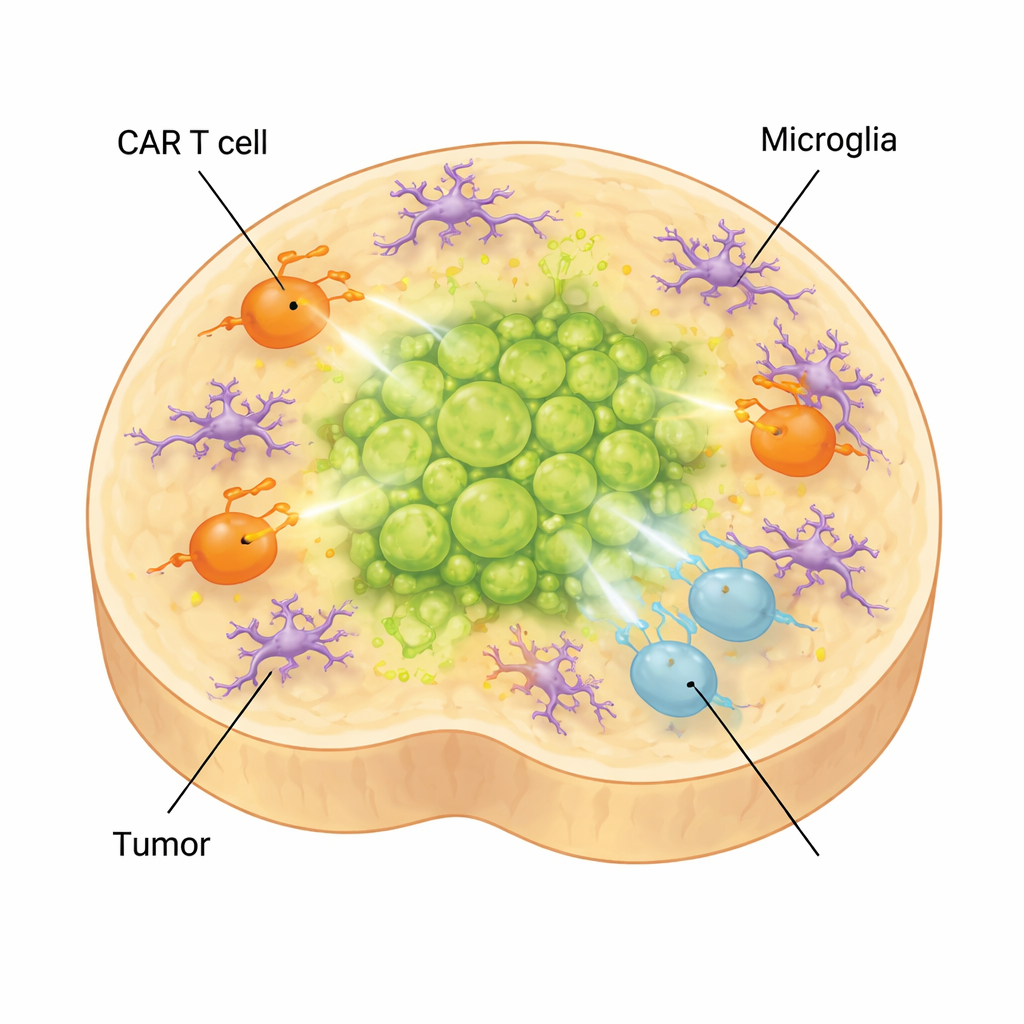
What this means for children with this cancer
For families facing diffuse midline glioma, this work does not yet offer a new cure, but it provides a powerful testing ground to get there faster. By recreating a child’s brainstem environment and tumor in a dish, scientists can now observe, in real time, how different tumor cells, CAR T cell types and microglia interact over weeks. This allows them to pinpoint which engineered T cells are most effective, how and when they become exhausted, and how brain-resident immune cells undermine their efforts. Ultimately, such organoid models could be personalized for individual patients and used to fine-tune immunotherapies before they are given, improving the odds that future CAR T cell treatments will be both safer and more effective for children.
Citation: Bessler, N., Wezenaar, A.K.L., Ariese, H.C.R. et al. De novo H3.3K27M-altered diffuse midline glioma in human brainstem organoids to dissect GD2 CAR T cell function. Nat Cancer 7, 316–333 (2026). https://doi.org/10.1038/s43018-025-01084-0
Keywords: diffuse midline glioma, brain organoids, CAR T cells, pediatric brain cancer, microglia