Clear Sky Science · en
PLIN5 phosphorylation orchestrates mitochondria lipid-droplet coupling to control hepatic lipid flux and steatosis
Why Liver Fat Matters
Many people have extra fat in their livers without feeling sick, yet this silent buildup can eventually lead to inflammation, scarring and even cancer. This study asks a simple but crucial question: when waves of fat arrive in the liver during fasting or from a rich "Western" diet, how do liver cells decide whether that fat will be burned for energy or stored safely, and when does this safety system start to fail?
Little Powerhouses and Tiny Fat Bubbles
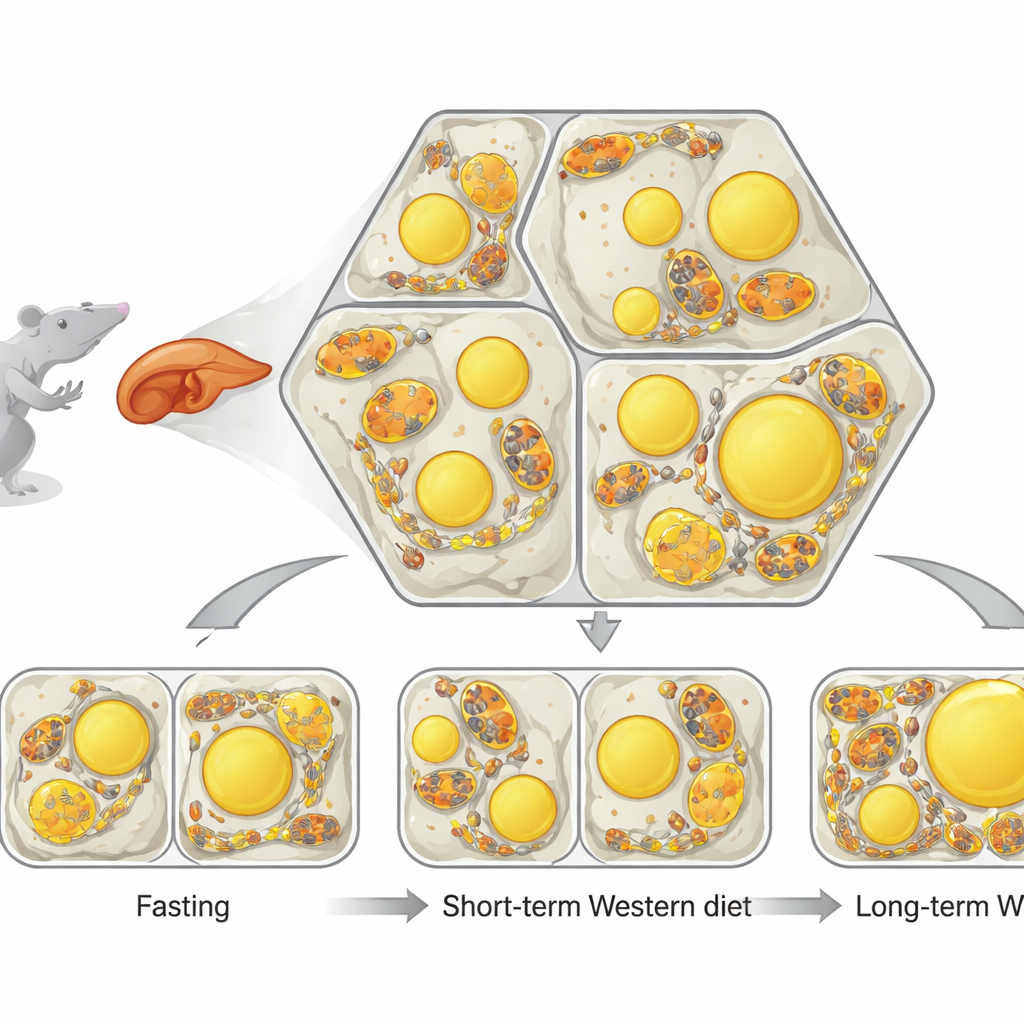
The liver is organized into repeating hexagon-like units where blood flows from one side to the other. Along this path, liver cells specialize: some mostly burn fat for energy, while others mainly make and store fat. Inside each cell, two structures are key. Mitochondria are the cell’s power plants, and lipid droplets are small fat-filled bubbles used for storage. The researchers developed a powerful imaging and analysis pipeline, called single-cell phenotypic profiling (scPhenomics), to map the size, shape and arrangement of these structures in thousands of cells directly in tissue slices. They found that even in healthy animals, mitochondrial shape and lipid droplet content form distinct spatial patterns that mirror where burning versus storage normally happens in the liver.
Fasting Versus a Rich Diet
Next, the team asked what happens when the liver is suddenly flooded with fat. Fasting drives fat out of body fat stores and into the liver, while a short-term Western-style diet delivers extra fat directly from food. In fasted mice, lipid droplets increased throughout the liver, and mitochondria became longer and often wrapped closely around these fat bubbles, forming many contact sites. In mice fed a Western diet for several weeks, the liver also filled with fat, but the droplets were arranged differently and mitochondria stayed mostly separate and more rounded. This showed that the way fat arrives—through fasting or chronic rich feeding—triggers very different structural responses inside liver cells, even when overall fat content looks similar.
A Molecular Switch for Fat Handling
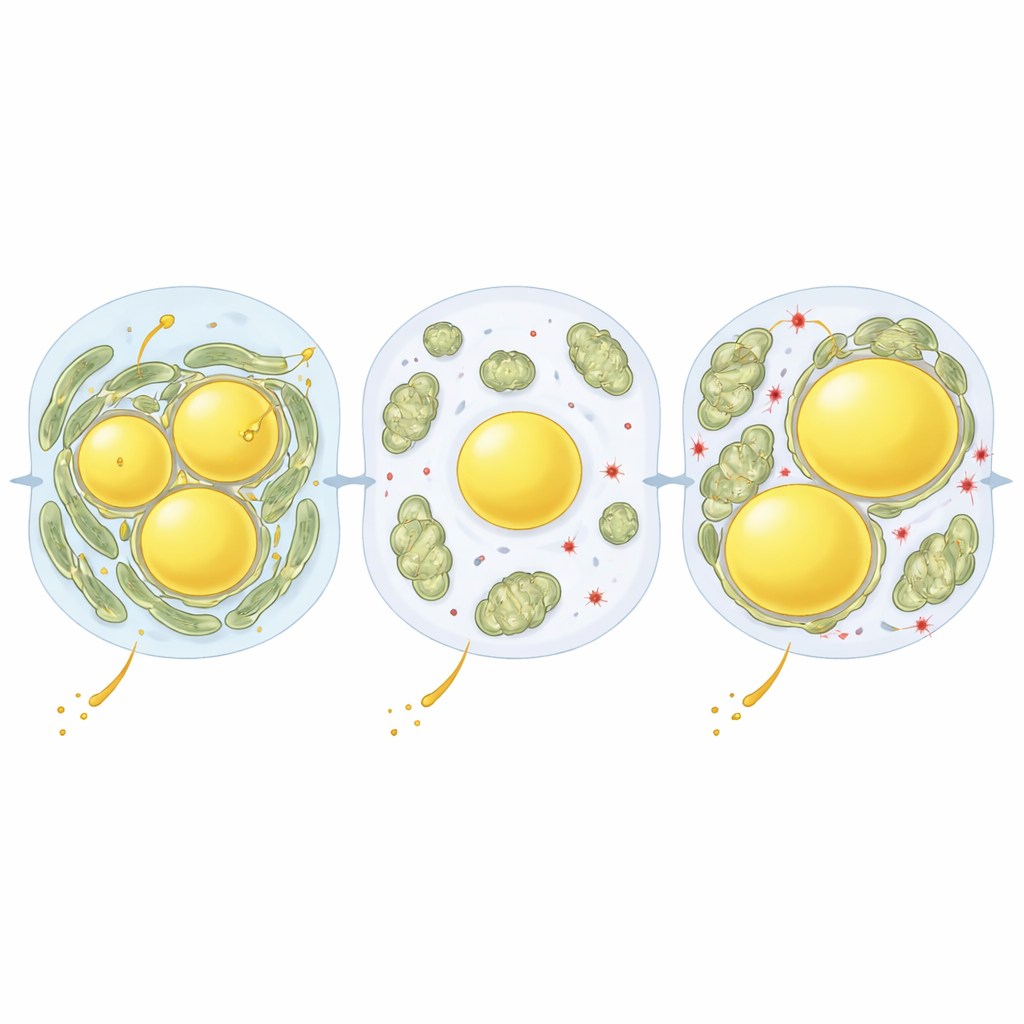
To uncover what controls these organelle contacts, the authors combined their imaging with protein measurements in liver cells taken from different zones and diet conditions. One standout protein was perilipin‑5 (PLIN5), which coats lipid droplets and can tether mitochondria to them. Fasting boosted PLIN5 levels and positioned it right at the interface between mitochondria and droplets. The researchers then used a liver-targeted virus to raise PLIN5 levels and to test versions of the protein that mimic different on/off states of a single chemical tag called phosphorylation. A version that could not be phosphorylated (S155A) strongly increased mitochondria–droplet contacts and enlarged fat droplets, even on a normal diet. A version that mimicked constant phosphorylation (S155E) did the opposite: mitochondria rarely touched droplets, which stayed fewer and smaller.
Protecting Cells From Fat-Driven Damage
The team then looked at what this meant for liver health under a Western diet. When mice ate this diet for several weeks, the non-phosphorylatable PLIN5 variant (S155A) drove more fat into droplets and raised liver triglyceride content, yet markers of damaging oxidative stress were actually lower. By contrast, the phospho-mimic version (S155E) left more free fat and signs of a more oxidizing, potentially harmful environment, with lower antioxidant reserves. In effect, close partnerships between mitochondria and lipid droplets appeared to serve as a safety valve, channeling excess fatty acids into neutral storage and limiting the chemical chain reactions that damage membranes and proteins. Over longer periods of Western diet feeding, however, mitochondria–droplet contacts increased again and were associated with heavier fat loading in both mice and human liver samples, hinting that the same protective wiring can become overburdened and contribute to disease progression.
A New Handle on Liver Fat Disease
Altogether, this work shows that liver cells actively rewire the physical relationships between their power plants and fat stores to cope with changing nutrient conditions. PLIN5, and particularly its phosphorylation state at a single site, acts like a fine-tuning knob that strengthens or loosens mitochondria–lipid droplet coupling. Tight coupling promotes the packaging of potentially toxic free fats into safer triglyceride stores and helps buffer short-term nutritional stresses, such as fasting or brief exposure to a rich diet. When this adaptive program is blunted, or when high-fat exposure persists for months or years, the balance tips toward lipotoxicity and oxidative damage. By revealing this structural "circuit" inside liver cells, the study suggests that drugs that modulate PLIN5 activity or mitochondria–droplet contacts could offer a new way to slow or prevent fatty liver disease in people.
Citation: Kang, S.W.S., Brown, L.A., Miller, C.B. et al. PLIN5 phosphorylation orchestrates mitochondria lipid-droplet coupling to control hepatic lipid flux and steatosis. Nat Metab 8, 587–603 (2026). https://doi.org/10.1038/s42255-026-01476-1
Keywords: fatty liver disease, liver metabolism, mitochondria, lipid droplets, perilipin 5