Clear Sky Science · en
Fatty acids promote uncoupled respiration via ATP/ADP carriers in white adipocytes
Why fat cells can help burn energy
Most people think of body fat as a silent storehouse of calories. This study reveals a surprisingly active side of white fat cells: under the right conditions, they can waste fuel and generate heat. By uncovering a new way that fat cells let energy "leak" out, the work points to fresh strategies for helping the body burn more calories and potentially combat obesity.
When stored fat is turned back into fuel
In everyday life, white fat cells spend much of their time storing energy as triglycerides—large droplets of fat. During exercise, fasting or cold exposure, stress signals trigger these cells to break down triglycerides in a process called fat breakdown, releasing fatty acids and glycerol. The researchers confirmed that when this breakdown is stimulated in white fat cells grown in the lab, the cells sharply increase their use of oxygen, a sign that their internal power plants, the mitochondria, are working harder. Blocking the first step of fat breakdown stopped both the release of fatty acids and the rise in oxygen use, showing that the process depends on fatty acids themselves.
Free fatty acids as on–off switches for energy burning
Fatty acids leaving the fat cell do not all exit the tissue; some remain inside. The team showed that these leftover free fatty acids are key to turning up mitochondrial activity. When a protein in the cell culture medium was used to soak up fatty acids, the cells released more fat overall but their oxygen use fell, indicating that high internal fatty acid levels—not just fat breakdown in general—drive the extra respiration. Conversely, blocking the rebuilding of triglycerides, a pathway that normally re-packages fatty acids, raised oxygen consumption and further lowered the electrical charge across the mitochondrial membrane. Together, these findings suggest that free fatty acids push mitochondria into a leaky, energy-wasting mode.
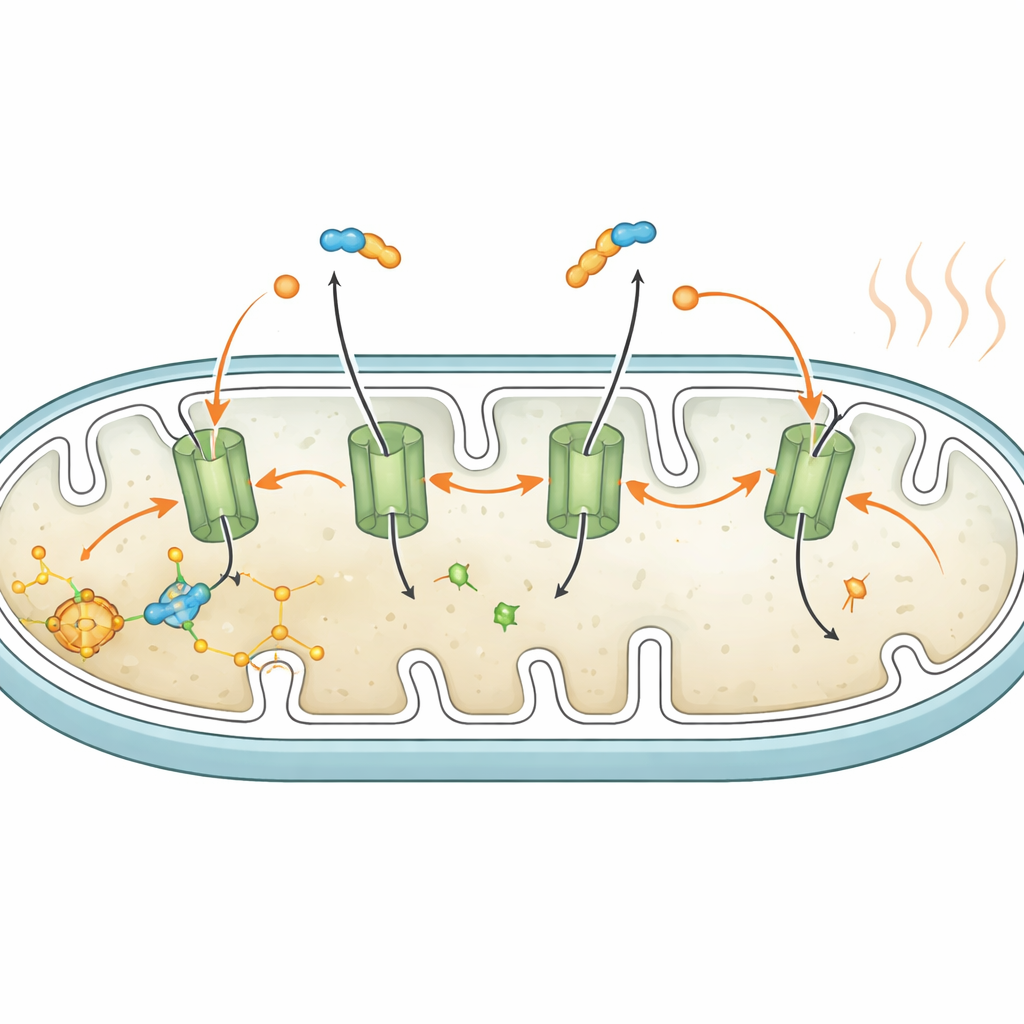
A new player in heat-producing respiration
In classic heat-producing brown fat, a protein called UCP1 pokes holes in the mitochondrial barrier so that the energy from food is released as heat rather than stored as useful cellular fuel. White fat cells have little or no UCP1, so the authors searched for another route. They focused on the ATP/ADP carrier, a transport protein that normally exchanges the cell’s energy currency across the mitochondrial barrier. Using both a specific chemical blocker and genetic tools to reduce this carrier, they showed that it is required for the fatty-acid-driven leak: blocking the carrier preserved the mitochondrial charge and prevented the rise in oxygen use during fat breakdown, even though fat breakdown itself proceeded normally. This points to free fatty acids working together with the carrier to create a controlled leak of protons across the mitochondrial barrier, turning on uncoupled respiration.
From cell culture to living animals
To test whether this energy leak matters in whole animals, the researchers used mice in which a signaling protein called STAT3 had been removed specifically from fat cells. These animals carry out fat breakdown normally but have reduced fatty-acid-driven respiration in their white fat. Under usual laboratory conditions, where brown fat is active and handles most cold defence, these mice stayed warm like their normal littermates. However, when the mice were made obese and kept at a temperature where brown fat is largely inactive, the animals lacking STAT3 in their fat became cold-sensitive and showed a weaker rise in overall oxygen use during cold exposure or drug-induced fat breakdown. Importantly, similar changes were not seen when STAT3 was deleted only in brown fat, pointing to a specific role for white fat.
White fat as a hidden heater
The study concludes that in white fat cells, free fatty acids generated during active fat breakdown can bind to the ATP/ADP carrier in mitochondria and trigger an energy leak that produces heat instead of useful cellular fuel. In obese animals kept warm, this uncoupled respiration in white fat makes a meaningful contribution to maintaining body temperature and overall energy expenditure, especially when brown fat and muscle shivering are limited. Because white fat is abundant in adults, particularly in obesity, gently enhancing this leak could offer a new way to increase everyday calorie burning and support weight-loss therapies, provided it can be done safely and without harming other tissues.
Citation: Ahmadian, M., Aksu, A.M., Dhillon, P. et al. Fatty acids promote uncoupled respiration via ATP/ADP carriers in white adipocytes. Nat Metab 8, 572–586 (2026). https://doi.org/10.1038/s42255-026-01467-2
Keywords: white adipose tissue, fatty acids, mitochondrial uncoupling, thermogenesis, obesity