Clear Sky Science · en
PFKM governs metabolic shifts throughout skeletal muscle differentiation
Why muscle cells need smart sugar use
When we exercise or simply stand up from a chair, our skeletal muscles spring into action, burning sugar to power every contraction. But young muscle stem cells and fully formed muscle fibers do not handle sugar in the same way. This study reveals a built‑in switching system—centered on a single enzyme called PFKM—that decides whether glucose is burned for immediate energy or rerouted to protect and rebuild cells. Understanding this switch could open new paths for treating muscle weakness, aging muscles, and inherited metabolic diseases.
A traffic light for cellular fuel
Glucose entering a cell can follow several routes. One major road, glycolysis, rapidly converts sugar into energy and building blocks for growing muscle fibers. Another route, the pentose phosphate pathway, produces molecules that defend cells against damaging oxygen stress and support DNA and lipid synthesis. The researchers focused on PFKM, a version of the enzyme phosphofructokinase-1 that sits at a key junction in glycolysis. By measuring hundreds of metabolites over minutes to hours after activating a growth signal called Wnt, they found that cells quickly piled up early glycolysis intermediates while the product of PFKM dropped, and pentose phosphate pathway metabolites rose. This suggested that cells were actively throttling PFKM to divert sugar toward protective chemistry instead of pure energy production. 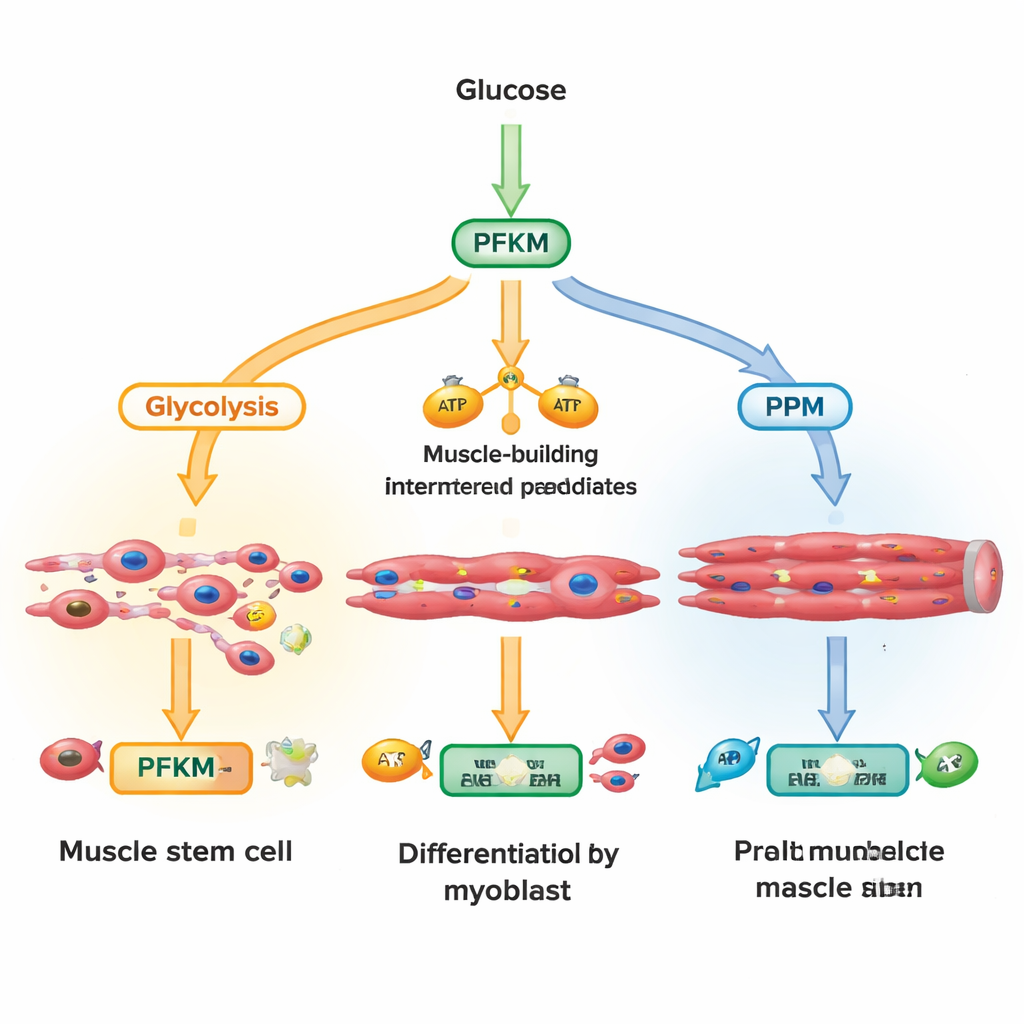
Tagging and removing the gatekeeper enzyme
To see how PFKM is controlled, the team tracked where the protein sits inside cells. Under resting conditions, PFKM floated throughout the cytoplasm. Within minutes of Wnt stimulation, it gathered into bright dots that overlapped with lysosomes, the cell’s protein‑shredding compartments. Two sister enzymes, PFKL and PFKP, did not move or change levels, revealing that only the muscle‑type PFKM was targeted. Biochemical tests showed that blocking lysosome function prevented PFKM loss, while inhibiting the proteasome—the other main protein disposal system—had no effect. Sequence analysis uncovered a short “methyl arginine degron” motif unique to PFKM. The enzyme PRMT1 added a specific methyl mark on an arginine within this motif, and this tag allowed microautophagy machinery, including the protein VPS4, to pull PFKM into lysosomes for degradation. Disabling PRMT1 or VPS4 stabilized PFKM and blocked its removal.
From stem cell to working muscle fiber
Using large human single‑cell datasets, the authors mapped PFKM levels across many muscle cell types. Muscle stem cells, which lie dormant until needed for repair, had very little PFKM but expressed many pentose phosphate pathway genes and lysosomal components. As cells committed to becoming muscle and fused into multinucleated fibers, PFKM transcripts and protein rose sharply, while Wnt targets and lysosomal genes waned. In cultured human and mouse muscle cells, Wnt rapidly drove PFKM into lysosomes in early, single‑nucleus cells but not in mature multinucleated fibers. This pattern supports a model in which undifferentiated cells keep PFKM low—via lysosomal degradation—to favor protective metabolism, then re‑express it as they transition into energy‑hungry contractile fibers.
What happens when the switch is stuck
To test how crucial PFKM is for building muscle, the team reduced its levels with RNA interference. Cells with low PFKM struggled to turn on hallmark muscle genes, produced less myosin protein, and formed fewer and smaller multinucleated fibers, even though overall cell numbers were unchanged. Metabolite profiling revealed reduced downstream glycolytic intermediates and tricarboxylic acid cycle fuels, but increased pentose phosphate genes and markers, alongside enhanced resistance to oxidative stress. Importantly, supplying cells with 3‑phosphoglycerate—a glycolytic intermediate that normally lies downstream of PFKM—rescued many of the differentiation defects. Muscle markers and fiber formation rebounded, showing that missing metabolites, rather than loss of the PFKM protein itself, were the main bottleneck. 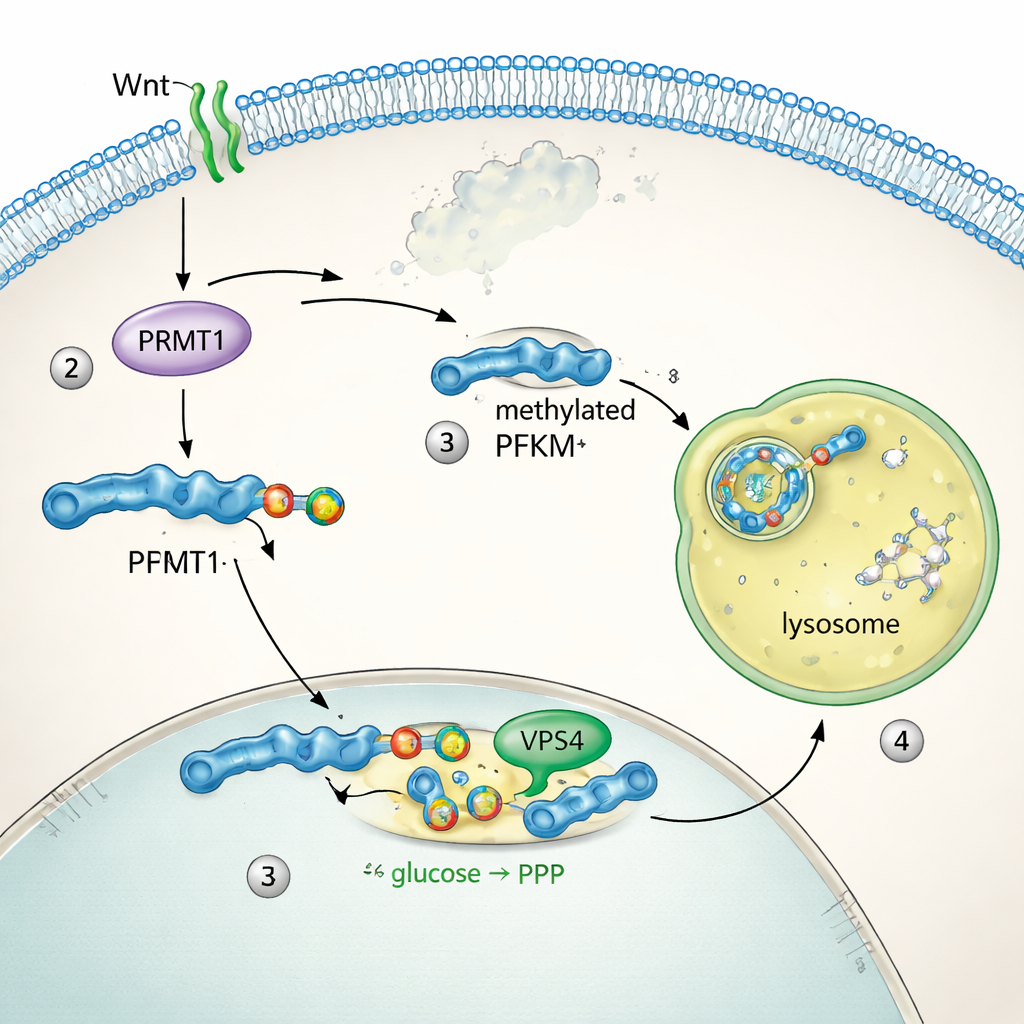
Implications for muscle health and repair
To a non‑specialist, the central message is that muscle cells do not simply “burn sugar” more or less; they carefully reroute it between energy production and cell protection depending on their stage of life. PFKM acts like a controllable valve at this junction. In stem‑like cells, Wnt‑driven tagging and lysosomal destruction of PFKM redirect glucose into a pathway that shields cells and prepares them for future growth. As cells mature into working fibers, they rebuild PFKM stores and swing metabolism toward high‑output energy use. Disrupting this balance, as seen in rare PFKM deficiency disorders, derails normal muscle development. By uncovering the molecular handle on this switch, the study suggests that future therapies might fine‑tune muscle regeneration or protect muscles in diseases and aging by modestly adjusting PFKM activity or supplying the right downstream metabolites.
Citation: Campos, M., Nguyen, S.T., Kong, X. et al. PFKM governs metabolic shifts throughout skeletal muscle differentiation. Nat Metab 8, 489–505 (2026). https://doi.org/10.1038/s42255-026-01457-4
Keywords: skeletal muscle differentiation, glucose metabolism, PFKM enzyme, pentose phosphate pathway, lysosomal protein degradation