Clear Sky Science · en
Feeding-regulated glycogen metabolism drives rhythmic liver protein secretion
Why the Liver’s Daily Rhythm Matters
Most of us think about what we eat, but not when our liver quietly packages and sends out vital proteins into the bloodstream. This study reveals that the liver’s protein “shipping schedule” is anything but constant: it follows a daily rhythm that depends strongly on when we eat and on how the liver uses stored sugar, known as glycogen. Understanding this timer system helps explain why meal timing, obesity, and certain rare genetic diseases can disrupt hormones, clotting factors, and many other blood proteins that keep the body in balance.
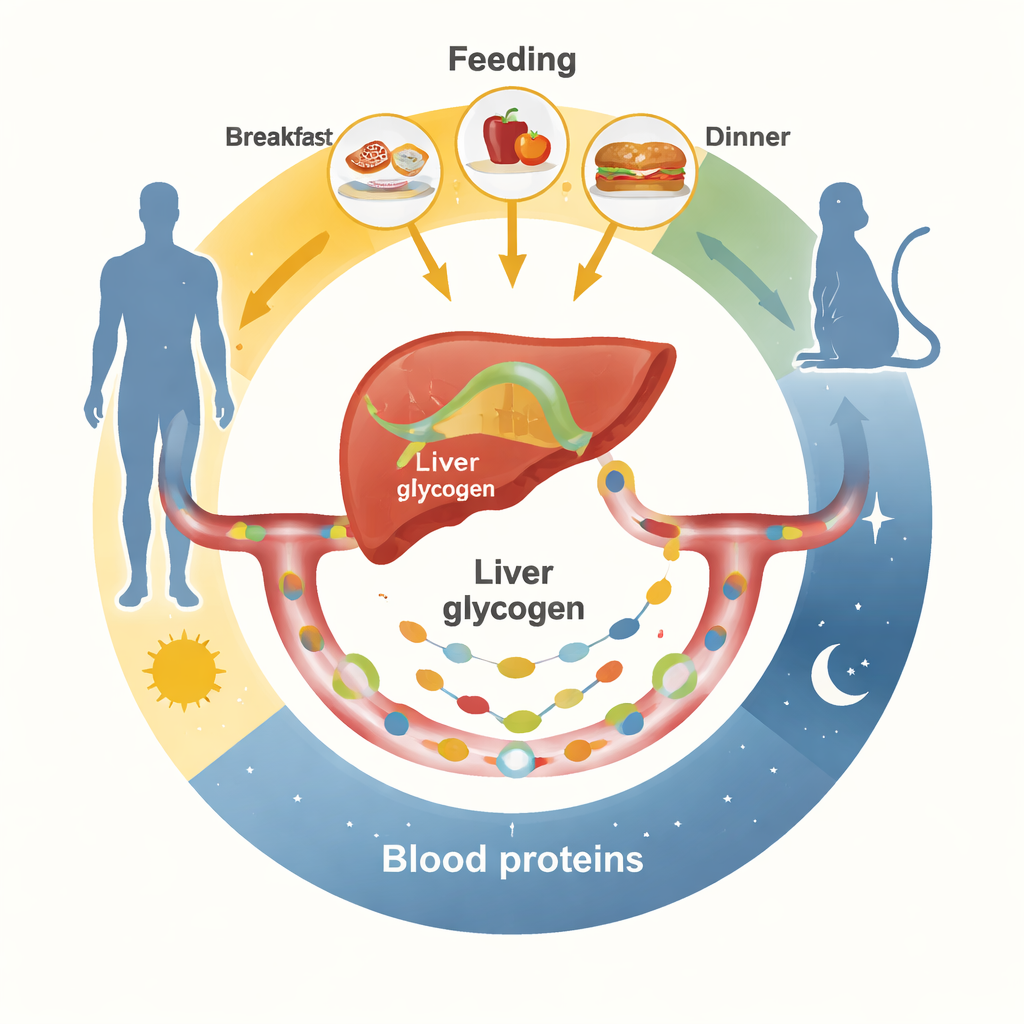
Meals Set the Beat for Blood Proteins
The researchers first tracked hundreds of blood proteins over 24 hours in healthy men under two controlled eating patterns. In one, volunteers ate regular meals; in the other, the same total calories were spread evenly across the waking day. With regular meals, many blood proteins rose and fell in clear daily waves, often peaking around early morning and late afternoon. These included metabolic hormones, clotting factors, immune proteins, and transporters made largely by the liver. When food was sipped slowly and continuously instead, most of these rhythms flattened out. Similar experiments in mice, with food available either all the time or only in defined day or night windows, showed that changing feeding schedules powerfully reshaped which proteins in the blood were rhythmic and when they peaked.
The Liver’s Secretory Line Runs on a Clock
To find out how these patterns arise, the team looked inside the liver. They found that the proteins involved in the classic “secretory pathway” — where newly made proteins enter the endoplasmic reticulum (ER), are modified in the Golgi apparatus, and are then released — also rise and fall over the day. Rather than being a constant conveyor belt, this pathway is tuned by time of day. Most rhythmic blood proteins carried the molecular “signal peptides” that mark them for this pathway, and blocking ER–Golgi transport in mouse liver slices sharply reduced protein release. In contrast, markers of cell damage and protein breakdown did not show strong daily changes, indicating that it is secretion, not destruction, that chiefly drives the observed blood rhythms.
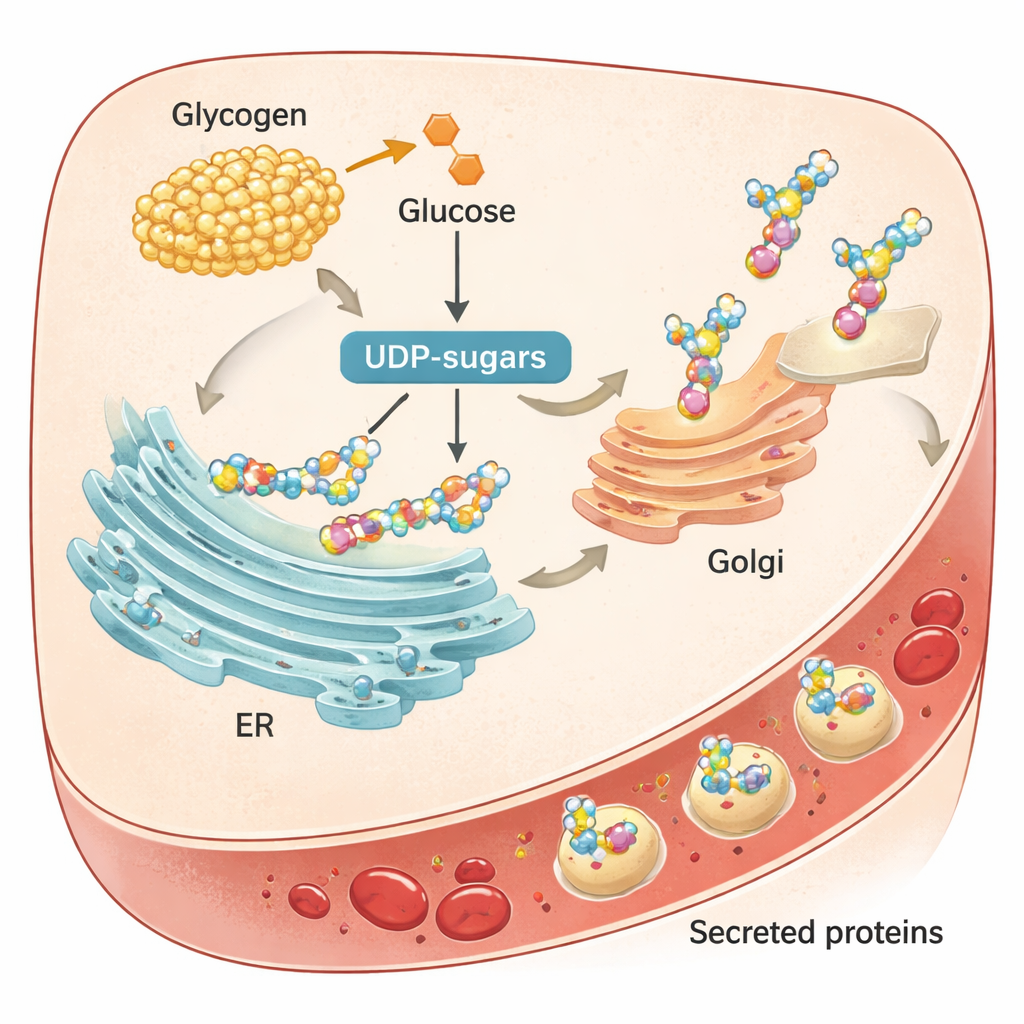
Glycogen: Stored Sugar Fueling Protein Packaging
The authors then linked these timing effects to how the liver handles glycogen, its main carbohydrate store. After meals, the liver builds up glycogen; during fasting, it breaks glycogen down, a process called glycogenolysis. The breakdown products feed into “UDP-sugars,” the activated sugar units used to decorate proteins with sugar chains, a modification called glycosylation. In mice, levels of UDP-sugars oscillated strongly over the day, in step with the activity of enzymes that build and break glycogen. When the team blocked the key liver glycogenolysis enzyme PYGL with a drug, glycogen piled up, blood sugar dipped during fasting, and the supply of UDP-sugars fell. This led to less glycosylation of liver proteins, signs of stress in the ER, and reduced secretion of several important proteins into the blood.
Stress Signals and Disease Links
Disrupting glycogen breakdown did more than just slow protein export; it triggered the cell’s quality-control alarm system. Liver cells treated with the PYGL-blocking drug activated the unfolded protein response, a set of ER stress pathways that can boost protein degradation and selectively alter which proteins are secreted. In obese mice, which naturally have altered glycogen handling, the normal daily swings in glycogen were blunted, and the number of rhythmic blood proteins fell. The same was true when normal mice underwent time-restricted feeding that effectively reduced calories. Finally, in a large human genetic study, people carrying variants in genes tied to glycogen storage diseases or congenital glycosylation disorders showed altered levels of many blood proteins, reinforcing the link between glycogen metabolism, protein glycosylation, and secretion.
What This Means for Everyday Health
For a non-specialist, the message is that the liver runs a timed export business for blood proteins, using stored sugar as the fuel that powers proper packaging and release. When feeding patterns, internal clocks, or glycogen-processing genes are disturbed, this schedule falters, changing the levels of hormones, clotting factors, and immune proteins in the bloodstream. These changes may help explain why meal timing, obesity, and certain rare metabolic conditions are linked to wide-ranging health problems. The work suggests that not just what we eat, but when we eat, can influence the liver’s daily shipping rhythm—and that doctors may need to consider time of day and eating schedules when interpreting blood tests.
Citation: Weger, M., Mauvoisin, D., Hoyle, D. et al. Feeding-regulated glycogen metabolism drives rhythmic liver protein secretion. Nat Metab 8, 327–349 (2026). https://doi.org/10.1038/s42255-026-01453-8
Keywords: circadian rhythm, liver glycogen, protein secretion, meal timing, metabolism