Clear Sky Science · en
Lamin A/C-regulated cysteine catabolic flux modulates stem cell fate through epigenome reprogramming
How the Cell’s Skeleton Talks to Its Metabolism
Why do some cells stay youthful and flexible in what they can become, while others age or lock into the wrong identity? This study uncovers a surprising conversation inside our cells between the nuclear “scaffold” that shapes DNA and a sulfur‑containing nutrient, cysteine. By showing how this dialogue steers stem cells toward healthy fates—or toward premature ageing—the work hints that what cells “eat” and how they process it could someday be tuned to prevent certain genetic diseases and perhaps even slow aspects of ageing.
The Hidden Role of the Nuclear Shell
Deep inside each cell, DNA is pressed against a protein mesh called the nuclear lamina, built partly from proteins called lamin A and C. These lamins not only give the nucleus its shape; they also help decide which genes are locked down and which remain accessible. The authors studied mouse embryonic stem cells in their most flexible, or “naive,” state and found that lamin A/C normally keeps a check on two metabolic enzymes, CBS and CTH, which help make and break down cysteine. When lamin A/C is present and active, these enzymes are held back and cysteine processing stays low, supporting a stable, youthful stem cell state. 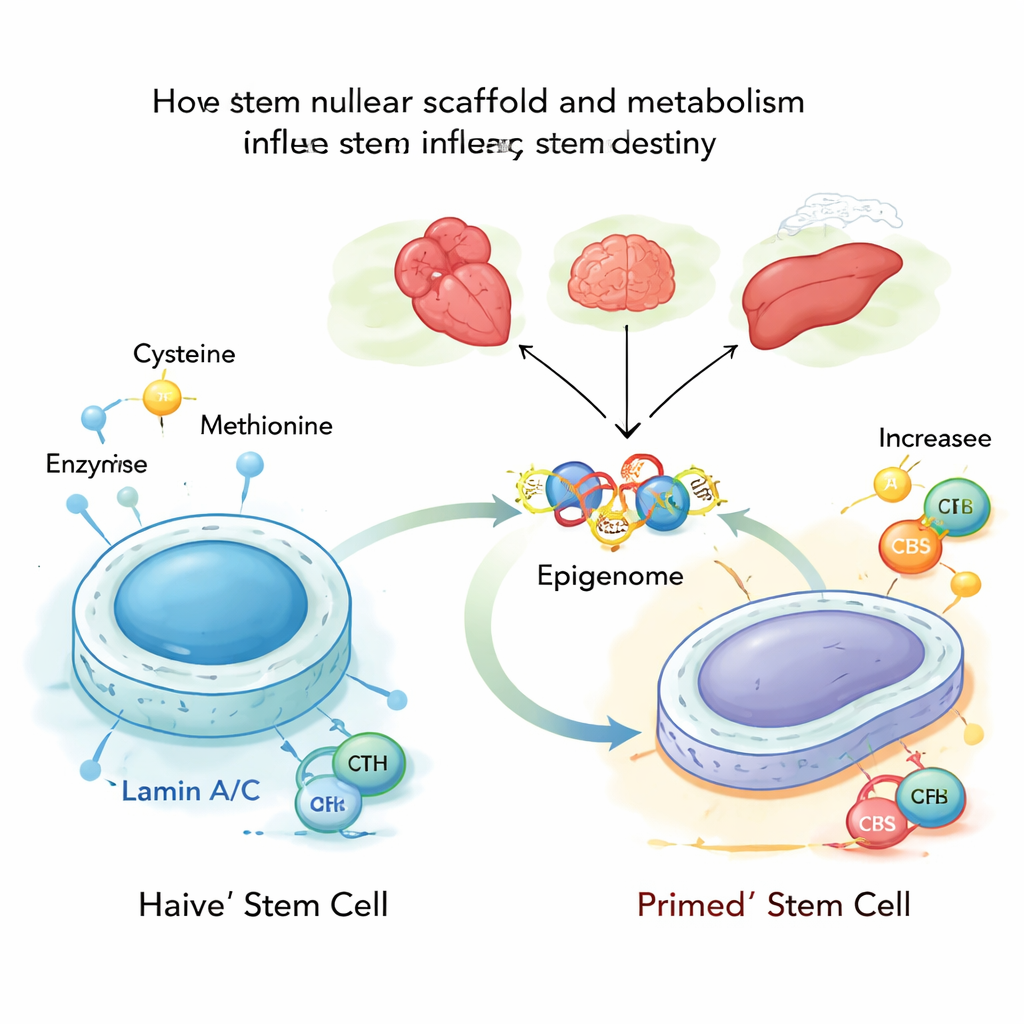
When Structure Fails, Metabolism Speeds Up
Next, the researchers removed lamin A/C or introduced a disease‑linked mutation that mimics a human premature‑ageing disorder called Hutchinson–Gilford progeria. Without normal lamin A/C, the CBS and CTH genes become more accessible and more strongly bound by a transcription factor called SP1, turning them on. That change pushes more cysteine through a metabolic pathway that ends in acetyl‑CoA, a small molecule that carries acetyl groups used to loosen DNA packing. As cysteine is increasingly funneled into acetyl‑CoA, the acetyl groups are preferentially added to specific spots on histone proteins—especially two positions on histone H3 known as K9 and K27—making nearby genes easier to switch on.
From Chemical Tags to Cell Fate Decisions
These shifts in histone “tags” act like a global retuning of the cell’s control panel. In naive stem cells, boosting CBS and CTH, or simply supplying more cysteine, pushes cells toward a more advanced, “primed” state that is closer to forming real tissues. Genes involved in heart development and other lineages become more active, and the three‑dimensional folding of DNA changes to favor these programs. Conversely, dialing down CBS and CTH or limiting cysteine helps restore a more naive, youthful identity even when the nuclear lamina is damaged. In embryonic models, this metabolic mis‑wiring led to skewed formation of the three germ layers—the early building blocks of all organs—and abnormal bias toward heart muscle cells unless CBS and CTH were rebalanced. 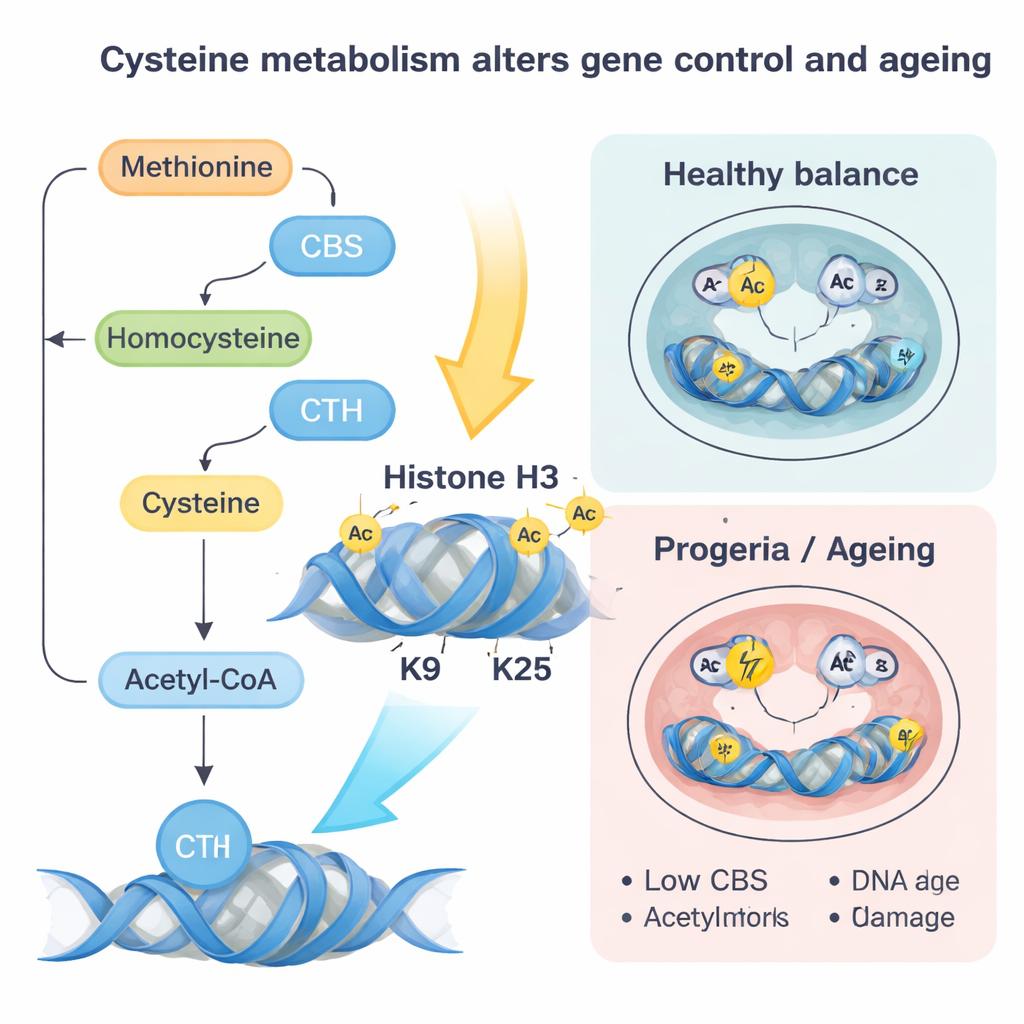
Metabolism, DNA Marks and Premature Ageing
The progeria‑like lamin mutation told a complementary story. Here, CBS and CTH levels drop, reducing cysteine flux and acetyl‑CoA from this route. At the same time, homocysteine is shunted back into the methionine cycle, increasing levels of SAM, the universal donor of methyl groups. That favors extra methyl tags, especially a mark called H3K9me3 placed by the enzyme SUV39H1, which tightens chromatin and is linked to impaired DNA repair. The authors showed that gently restoring CBS or restricting methionine lowered SAM, reduced these repressive methyl marks, improved DNA repair, decreased oxidative stress and senescence markers, and partially rescued heart cell differentiation. Similar drops in CBS and rises in repressive marks were seen in normal aged mouse hearts and in human fibroblasts from older people and progeria patients, suggesting this circuit operates in natural ageing as well.
What This Means for Health and Ageing
For a non‑specialist, the message is that the cell’s structural framework and its metabolism are tightly linked: when the nuclear shell made of lamin A/C is disturbed, the way cysteine is used changes, which in turn rewrites chemical marks on DNA‑packaging proteins. These marks decide which genes turn on as stem cells build the body and help determine how well cells repair damage over a lifetime. By carefully tuning enzymes like CBS and CTH—or by adjusting the supply of nutrients such as methionine and cysteine—it may be possible to correct some of the mis‑writing of these marks in lamin‑based diseases and perhaps mitigate aspects of premature or normal ageing.
Citation: Wang, Y., Shi, H., Wittig, J. et al. Lamin A/C-regulated cysteine catabolic flux modulates stem cell fate through epigenome reprogramming. Nat Metab 8, 431–453 (2026). https://doi.org/10.1038/s42255-025-01443-2
Keywords: stem cells, nuclear lamina, cysteine metabolism, epigenetics, ageing