Clear Sky Science · en
Elastic heterogeneity governs anomalous dynamic scaling in a soft porous crystal
Why tiny pores and hidden stresses matter
Materials that can soak up and release molecules on demand lie at the heart of future technologies for clean water, efficient batteries, and smart sensors. This article explores how a special class of “soft” porous crystals—materials whose internal scaffolding can bend and swell—take up gas or liquid molecules in surprisingly uneven ways. By zooming in on how the material’s stiffness changes from place to place as it fills, the authors reveal that hidden mechanical stresses can speed up or slow down absorption, generate tiny creases on the surface, and lead to unusual patterns in how quickly molecules spread.
Crumpling sponges made of crystal
Soft porous crystals, such as flexible metal–organic frameworks, behave a bit like sponges made from ordered crystal lattices. When guest molecules enter their pores, they do more than simply occupy space: they can stretch or compress the framework and make some regions stiffer than others. The study focuses on this “elastic heterogeneity”—the fact that different parts of the same crystal can become harder or softer as molecules move in. To capture these effects without tracking every atom, the authors build a coarse-grained model in which each site in a regular grid can hold at most one guest molecule, and the grid itself can expand and stiffen locally when occupied. Molecules are allowed to enter only from the bottom edge, mimicking real situations where uptake is controlled by external surfaces.
Corners that drink faster than flat surfaces
Simulations show that the crystal does not fill uniformly. Instead, when adsorption is strong enough to destabilize the empty state, dense “domains” of molecules first develop at the bottom corners of the crystal. These corner regions can relax mechanical stress more easily than the flat surface, so they act as preferred gateways for uptake. As time passes, the corner domains grow both upward and sideways, while smaller domains appear along the bottom surface. Because the contribution of corners is relatively larger in smaller samples, the overall uptake rate depends on the lateral size of the crystal: smaller crystals, with more influence from corners, fill faster than larger ones. This size dependence directly ties macroscopic performance to the way stresses are relieved at edges.
Wrinkles from softer gaps between stiff patches
Another striking feature is the emergence of creases on the surface. Adsorbed regions that have become harder and slightly expanded clamp down on neighboring unfilled regions, which remain softer and more compressible. These unfilled patches are effectively squeezed between rigid domains, so the surface buckles and forms a series of wrinkles or creasing lines. As molecules continue to enter, adsorbed domains merge into larger clusters and the spacing between creases gradually increases. The simulations also reveal a pronounced asymmetry between filling and emptying: during desorption, soft, unfilled channels tend to extend into the bulk from the surface, so corners empty more slowly than flat regions. This reversal stems from the same elastic contrast but with the roles of hard and soft areas switched.
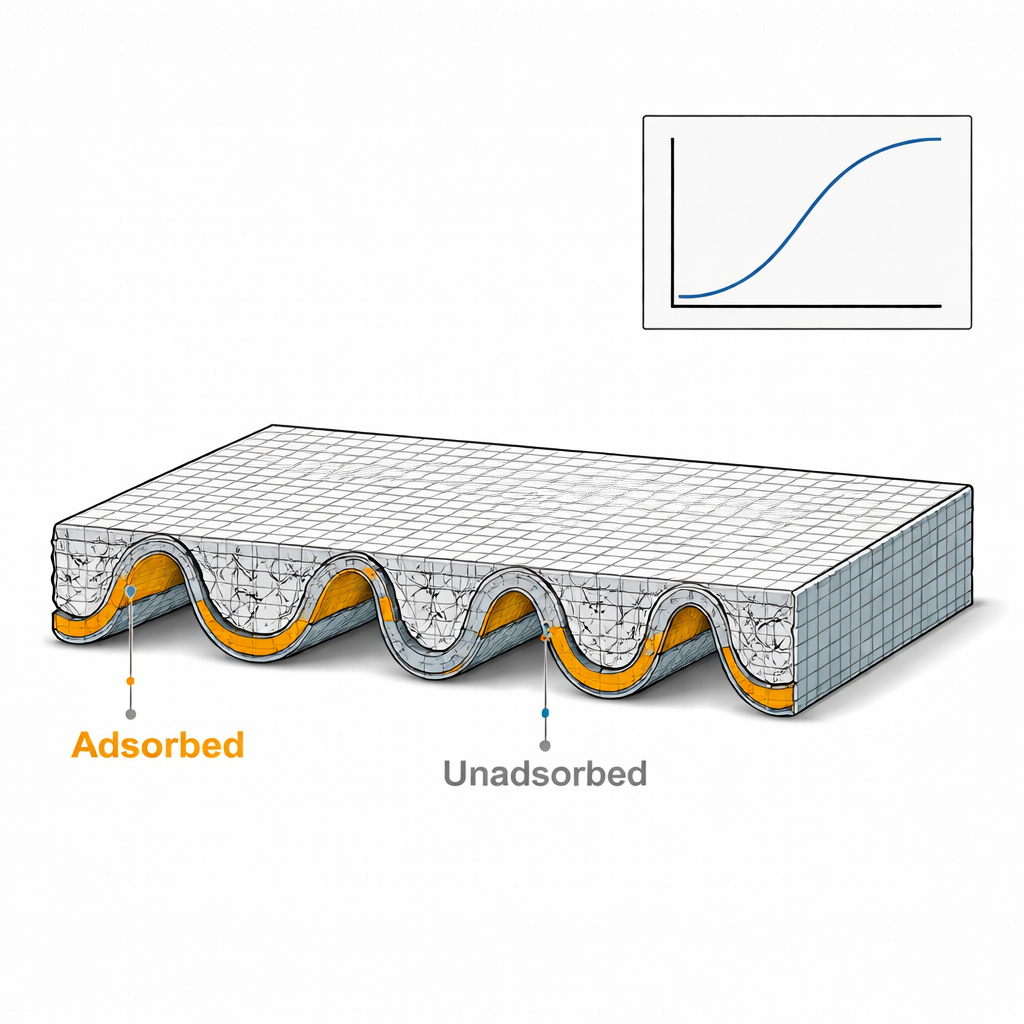
Patterns that break usual rules of growth
To make sense of these evolving patterns, the authors borrow ideas from studies of growing surfaces and rough fronts, where simple power laws often relate how fast structures grow to their size. Here, they analyze how the distribution of adsorbed molecules fluctuates along the surface, using measures of correlation, roughness, and spectral content. They find that the characteristic spacing between creases increases roughly as the cube root of time, indicating a slow coarsening controlled by redistribution of molecules within the crystal rather than by simple diffusion from outside. More importantly, the fluctuations at small and large length scales do not follow the same scaling rules: local roughness grows differently from global roughness, and the spectrum of fluctuations does not match known categories from classic interface-growth theories. This “anomalous dynamic scaling” points to a new class of cooperative behavior, driven by feedback between adsorption and elastic deformation.
Toward smarter, shape-shifting materials
In accessible terms, the article shows that how a soft porous crystal fills is governed not just by how easily molecules can move, but also by how the material bends and stiffens in response. Corners act as privileged entry points, squeezed gaps on the surface wrinkle into creases, and the overall spreading of molecules follows unusual time laws that standard models cannot capture. By identifying elastic heterogeneity as the key control knob, the work suggests practical strategies for engineering responsive materials: by tuning how much different regions harden or soften on uptake, designers could steer where molecules enter, how fast they spread, and how the material’s shape changes under external stimuli. This understanding could guide the creation of next-generation systems for water harvesting, catalysis, sensing, and energy storage, where mechanical feedback is deliberately used to program function.
Citation: Mitsumoto, K., Takae, K. Elastic heterogeneity governs anomalous dynamic scaling in a soft porous crystal. Commun Phys 9, 36 (2026). https://doi.org/10.1038/s42005-026-02508-8
Keywords: soft porous crystals, elastic heterogeneity, molecular adsorption, metal-organic frameworks, surface creasing