Clear Sky Science · en
Search for superconducting icosahedral hydrides via coordination number engineering
Why this matters for future technologies
Superconductors—materials that carry electricity with zero resistance—could transform power grids, medical scanners, and even future computers. But most known superconductors work only at extremely low temperatures or under crushing pressures. This study explores a clever new way to design superconducting materials made of hydrogen and metals, aiming to push their operating temperatures higher and their pressure requirements lower. By carefully arranging how many hydrogen atoms surround a heavy metal atom, the authors show how to "engineer" superconductivity into new compounds.
Building superconductor candidates like molecular Lego
The researchers focus on a family of materials called hydrides, which are rich in hydrogen. Hydrogen is light and vibrates easily, traits that naturally favor superconductivity when electrons interact with these vibrations. Instead of randomly testing thousands of combinations, the team starts from a known compound, BaReH9, where rhenium (Re) is surrounded by nine hydrogen atoms in a well-defined cluster. They then systematically ask: what happens if we squeeze in more hydrogen around rhenium and change how these atoms are connected? This design principle—tuning the number of neighboring atoms, known as the coordination number—acts like a structural dial for superconducting behavior.
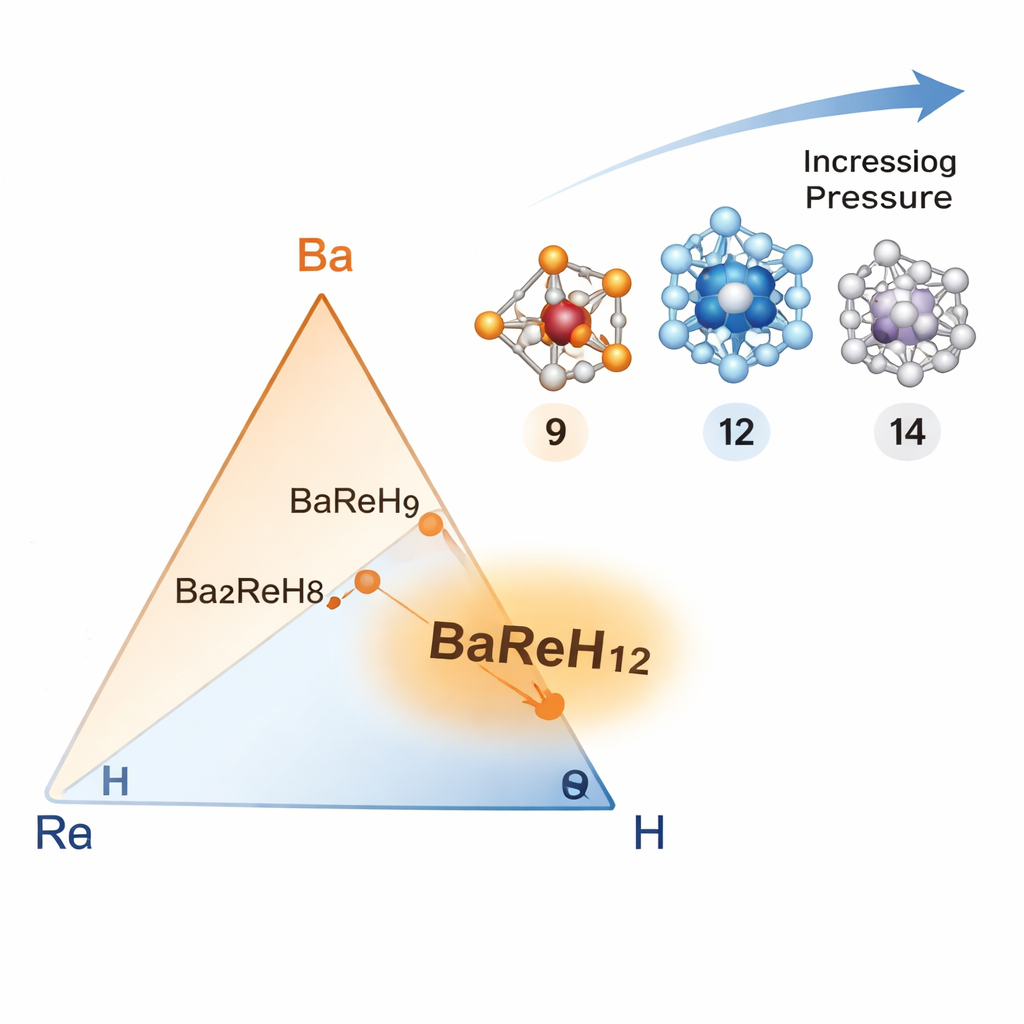
Discovering a twelve‑hydrogen cage with powerful effects
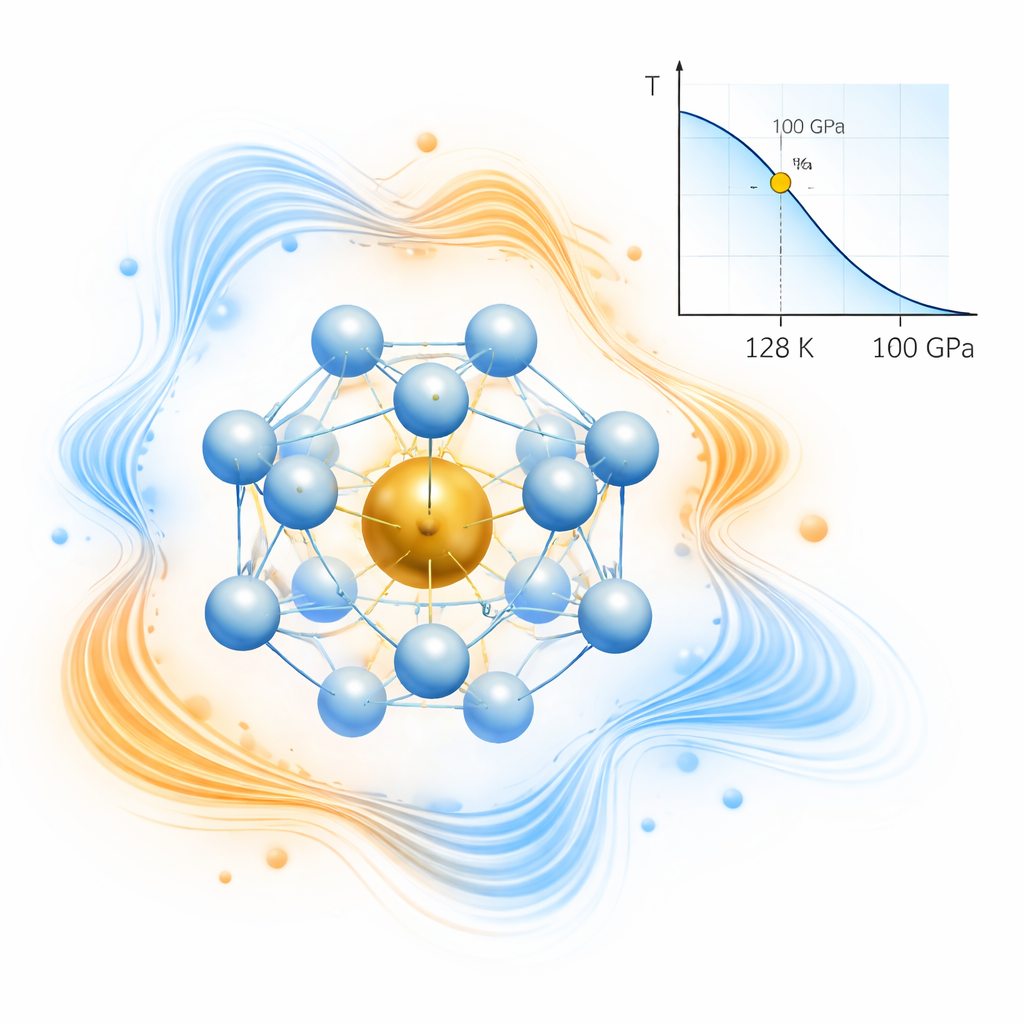
Using advanced computer simulations under very high pressures, the authors map out which combinations of barium (Ba), rhenium, and hydrogen are stable. They identify several promising compounds, including Ba2ReH8 and, most importantly, BaReH12. In BaReH12 at about 100 billion times atmospheric pressure (100 GPa), each rhenium atom is wrapped by 12 hydrogen atoms arranged in a near‑perfect icosahedral cage. This highly symmetrical structure forms a special unit, written as [ReH12]2−, that behaves like a building block for superconductivity. The calculations show that this compound can become superconducting at temperatures around 128 kelvin—more than halfway from absolute zero to room temperature, and notably high for such a chemically simple system.
How extra electrons and gentle hydrogen bonds help
Beyond geometry, the number of electrons inside each hydrogen–metal unit turns out to be crucial. Units with an odd number of electrons tend to be metallic, meaning their electrons move freely—an essential condition for superconductivity. BaReH12 has such an odd‑electron unit, which helps it conduct. At the same time, the hydrogen atoms between neighboring cages do not form very strong bonds; they are linked just enough to interact, but not so tightly that the electronic states become rigid. This combination—odd electron count, high symmetry, and relatively weak hydrogen–hydrogen bonding—produces strong coupling between electrons and atomic vibrations, the mechanism that underlies conventional superconductivity in these hydrides.
When more pressure becomes too much
As the pressure increases further, the tidy 12‑hydrogen cage begins to distort. In a higher‑pressure form of BaReH12, four hydrogen atoms are shared between neighboring rhenium centers, raising the coordination number to 14 and lowering the symmetry. This structural twist weakens the interaction between electrons and vibrations and causes the superconducting transition temperature to drop to around 40 kelvin. Similarly, another compound, Ba2ReH8, has a different hydrogen shell and extra barium atoms that push the hydrogen cages farther apart. It still becomes superconducting, but only near 19 kelvin. These comparisons highlight how sensitive superconductivity is to subtle changes in atomic arrangement.
Simple rules for designing better superconductors
Overall, the study proposes a clear recipe for discovering new high‑temperature hydride superconductors. Start with hydrogen‑and‑metal units that carry an odd number of electrons, surround them with positively charged atoms like barium that donate electrons and stabilize the structure, and aim for highly symmetrical cages with gently bonded hydrogen atoms between them. By treating these hydrogen cages as tunable building blocks, scientists gain a powerful new toolkit for exploring materials that might one day superconduct at practical temperatures and less extreme pressures—bringing applications from loss‑free power transmission to more compact magnets closer to reality.
Citation: Song, H., Du, M., Zhang, Z. et al. Search for superconducting icosahedral hydrides via coordination number engineering. Commun Phys 9, 59 (2026). https://doi.org/10.1038/s42005-026-02494-x
Keywords: superconducting hydrides, high pressure materials, hydrogen rich compounds, coordination number engineering, BaReH12