Clear Sky Science · en
Diels-Alder reaction affords circumpyrene tetracarboxydiimide with excited state intramolecular charge transfer character
Why this glowing molecule matters
Electronics made from carbon-based molecules promise flexible phones, light-emitting wallpapers, and ultra-thin solar cells. To reach that future, chemists need molecules that both conduct charges efficiently and interact strongly with light, especially in the red and near‑infrared part of the spectrum. This paper reports a new family of such molecules, built by enlarging a tiny slice of graphene and decorating it so that, when light hits, electrical charge shifts inside the molecule in a highly controlled way.
Building a bigger light-harvesting core
The work centers on polycyclic aromatic hydrocarbons—flat, fused-ring “nanographenes” that resemble small flakes of graphene. The team starts from a highly luminescent nanographene called dibenzo[hi,st]ovalene, which already behaves as an efficient light emitter. Their goal is to both enlarge this carbon framework and attach special side groups, called imides, that like to pull electrons toward themselves. By flanking an electron‑rich carbon core with electron‑poor imides, they aim to create an acceptor–donor–acceptor layout that naturally encourages charge to shift across the molecule when excited by light. 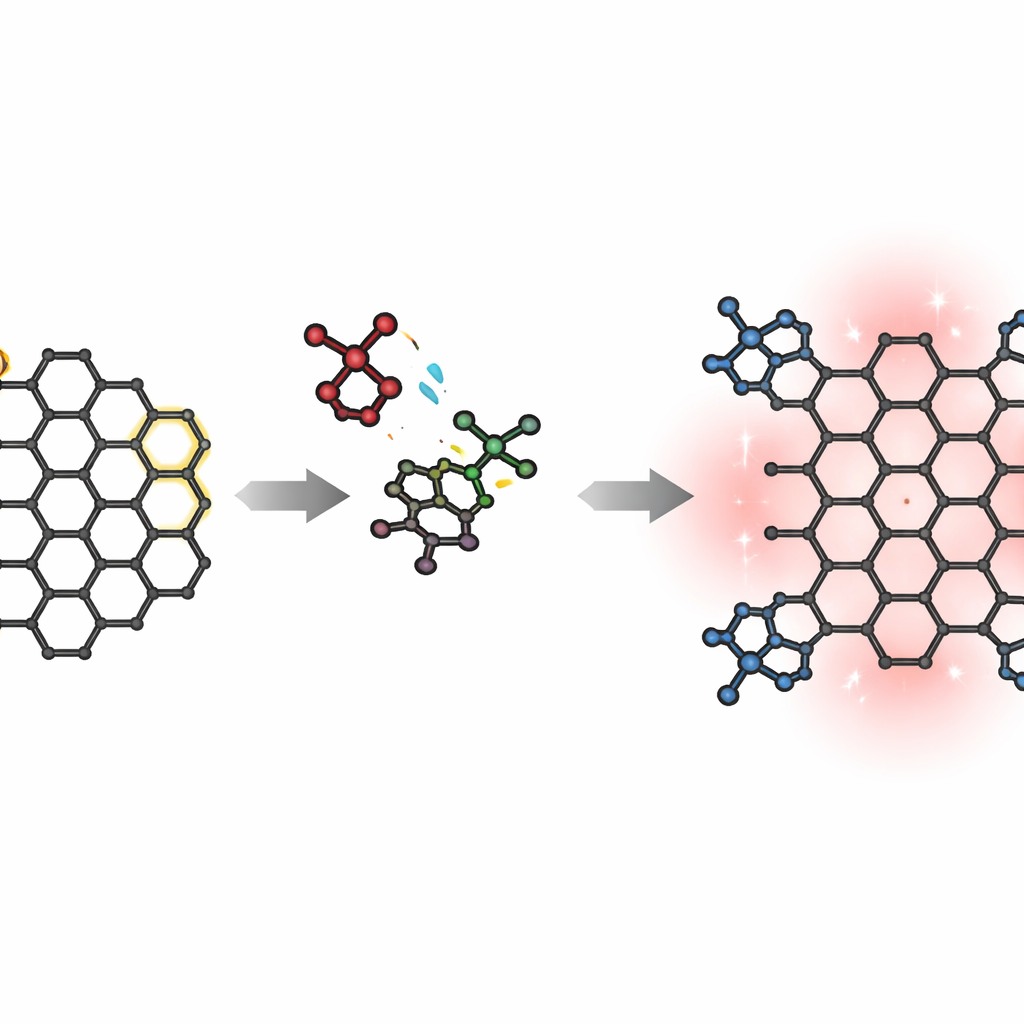
Snapping new rings into place
To reshape the nanographene, the researchers use a classic bond‑forming method known as the Diels–Alder reaction, which can fuse new rings onto pre‑existing aromatic systems. They direct this reaction to “bay” regions—indentations along the edge—of the starting nanographene. By reacting it with maleimide building blocks at high temperature, they obtain two main products: a mono‑adduct with one new ring and one imide, and a di‑adduct called circumpyrene tetracarboxydiimide, where both bay regions are extended and four imide groups are installed. Although this strategy sounds straightforward, the yields of the fully extended di‑adduct are modest, hinting at a subtle reaction pathway.
Uncovering a hidden reaction step
To understand why the second ring‑forming step is sluggish, the authors turn to quantum‑chemical calculations. These simulations reveal that once the first Diels–Alder addition is fully “aromatized” back into a flat graphene‑like structure, it becomes surprisingly inert toward a second addition. Instead, the most likely route to the di‑adduct passes through a partially hydrogenated intermediate that is only partly re‑flattened. This species has a lower barrier for adding a second maleimide unit. The calculations therefore explain the experimental finding: if the mono‑adduct fully relaxes into its most stable, aromatic form, it essentially stops reacting. Any strategy that favors the partially aromatized intermediate should improve the formation of the desired, fully extended product.
Making red light and moving charge
Once in hand, the new circumpyrene tetracarboxydiimide shows striking optical behavior. Compared with earlier circumpyrene molecules lacking imide groups, its absorption and emission are shifted toward longer wavelengths, into the deep‑red region, and its fluorescence becomes significantly brighter. Electrochemical measurements and theoretical models confirm that the imide groups pull down the molecule’s energy levels, shrinking the gap between the states that absorb and emit light. Detailed spectroscopy across different solvents reveals broad, red‑shifted absorption and emission bands that grow stronger in more polar liquids—classic fingerprints of an internal charge‑transfer state, where excitation moves electron density from the central nanographene core toward the imide “endcaps.” 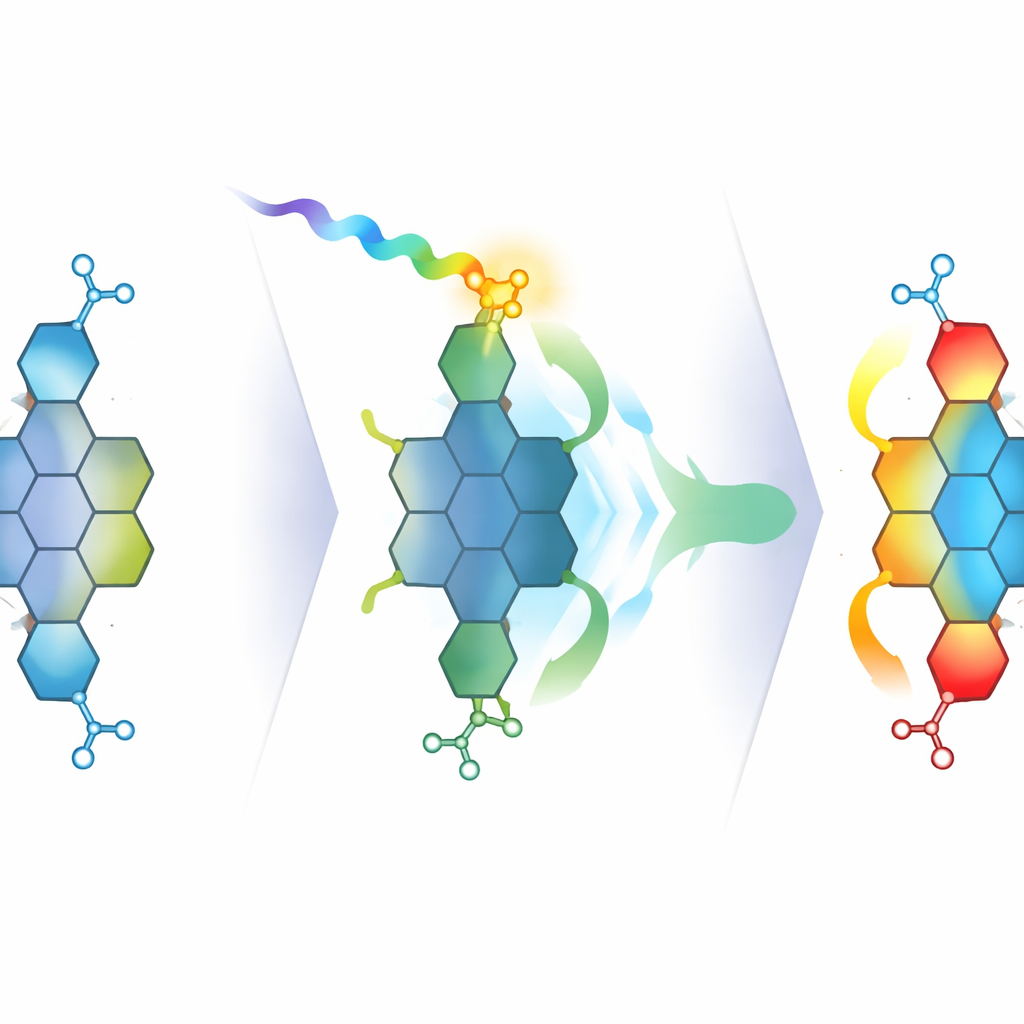
Watching charge shift in real time
To visualize how this charge‑transfer state forms and evolves, the team uses ultrafast transient absorption and two‑dimensional electronic spectroscopy. These techniques track, on femtosecond to picosecond timescales, how the molecule’s excited states appear, transform, and relax after a very short light pulse. When they excite the main absorption band, they mainly populate a more localized excited state on the carbon core. But when they excite slightly further into the red, they directly access a bright charge‑transfer state that shows its own distinct signals and persists for hundreds of picoseconds. The two‑dimensional maps confirm that this state is not merely a dark by‑product reached by relaxation, but a genuine optical transition that can be addressed directly by light.
From designer molecules to future devices
In simple terms, the researchers have devised a way to grow a larger, carefully wired piece of graphene that lights up brightly and internally shifts charge when illuminated. The key synthetic insight is that a partially relaxed reaction intermediate must be captured before it fully settles, while the key physical insight is that the attached imide groups create a built‑in pathway for charge transfer without sacrificing brightness. Such molecules are attractive building blocks for organic solar cells, optical switches, and bioimaging probes, where efficiently turning absorbed light into separated charges—or into bright, red‑to‑near‑infrared emission—is crucial.
Citation: Chen, Q., Guizzardi, M., Méndez, F. et al. Diels-Alder reaction affords circumpyrene tetracarboxydiimide with excited state intramolecular charge transfer character. Commun Chem 9, 122 (2026). https://doi.org/10.1038/s42004-026-01946-3
Keywords: nanographene, Diels–Alder, charge transfer, organic optoelectronics, fluorescent dyes