Clear Sky Science · en
ABEL-FRET bridges the timescale gap in single-molecule measurements of the structural dynamics in the A2A adenosine receptor
Watching Tiny Cell Switches in Real Time
Many of today’s medicines work by flipping molecular “switches” on cell surfaces called receptors. These switches constantly change shape as they turn signals on and off, but most tools can only see the very fastest or the very slowest motions—not both. This paper introduces a way to watch a single, drug‑target receptor in solution for much longer than before, revealing how it lingers in particular shapes that are crucial for drug action.
Why These Shape‑Shifting Proteins Matter
The study focuses on a major class of cell‑surface proteins known as G protein‑coupled receptors, or GPCRs. These proteins control vital processes including vision, mood, blood pressure, and immune responses, and more than a third of approved drugs act on them. One well‑studied member, the A2A adenosine receptor, helps regulate sleep, pain, inflammation, and brain signaling, and is a promising target for treating disorders such as Parkinson’s disease and cancer. GPCRs are highly flexible: when different molecules bind, they push the receptor into distinct shapes that favor some signaling pathways over others. That very flexibility, however, makes it hard to design drugs based on static snapshots from X‑ray or cryo‑electron microscopy.
Following a Single Receptor Without Pinning It Down
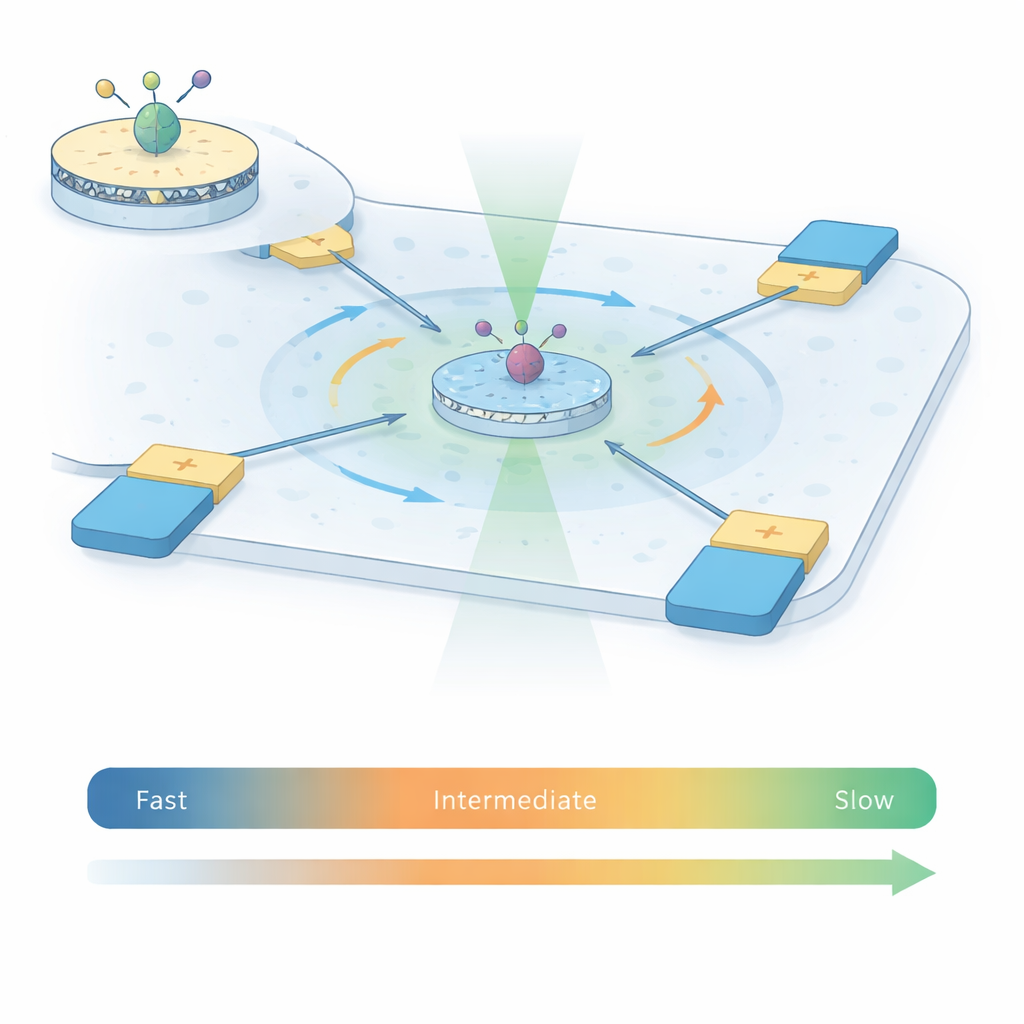
To capture how A2A receptors move, the authors combine two powerful techniques into what they call ABEL‑FRET. First, they reconstitute single receptors into tiny patches of artificial membrane called nanodiscs, giving each protein a more natural environment than detergent alone. They attach a pair of fluorescent dyes to two moving parts on the inside of the receptor. As the receptor changes shape, the distance and relative orientation between these dyes change, altering the energy transfer between them—a phenomenon known as Förster resonance energy transfer (FRET). Second, instead of sticking the receptor to a surface, they use an Anti‑Brownian Electrokinetic (ABEL) trap: a microfluidic chamber with electrodes that sense where a fluorescent particle is and gently push it back to the center, counteracting random Brownian motion.
Bridging the Missing Time Window
Traditional single‑molecule FRET experiments either watch freely diffusing receptors for only a few thousandths of a second, or immobilized receptors on a surface for seconds to minutes. Each approach covers a different time window. Using the ABEL trap, this work keeps individual A2A receptors in view for one to two seconds while they remain free in solution, roughly 100 times longer than in diffusion‑limited experiments. This extended viewing time allows the team to measure how the FRET signal fluctuates within each trapped burst and across thousands of receptors, under four conditions: without ligand, with an antagonist, and with two different activating molecules (agonists). Statistical tools borrowed from signal analysis—variance, correlation, and recurrence analyses—let them distinguish random photon noise from genuine, slow structural changes in the receptor.
Hidden Long‑Lived Shapes Revealed
The FRET readouts show that receptors populate multiple distinct conformations that do not fully interconvert during typical observation times of a few hundred milliseconds. In all conditions, the spread of FRET values is far broader than expected from noise alone, revealing structural heterogeneity: different molecules sit in different long‑lived shapes. When activating molecules bind, the average FRET level shifts upward, indicating that the receptor spends more time in an “active‑like” arrangement of its inner helices. Yet even then, the correlation analyses show that once a receptor is in a high‑ or intermediate‑FRET state, it has a strong chance of remaining there for at least hundreds of milliseconds. These results update earlier estimates based on faster experiments, extending characteristic “dwell times” in long‑lived states from mere milliseconds to well over one‑tenth of a second.
A New Map of the Receptor’s Energy Landscape
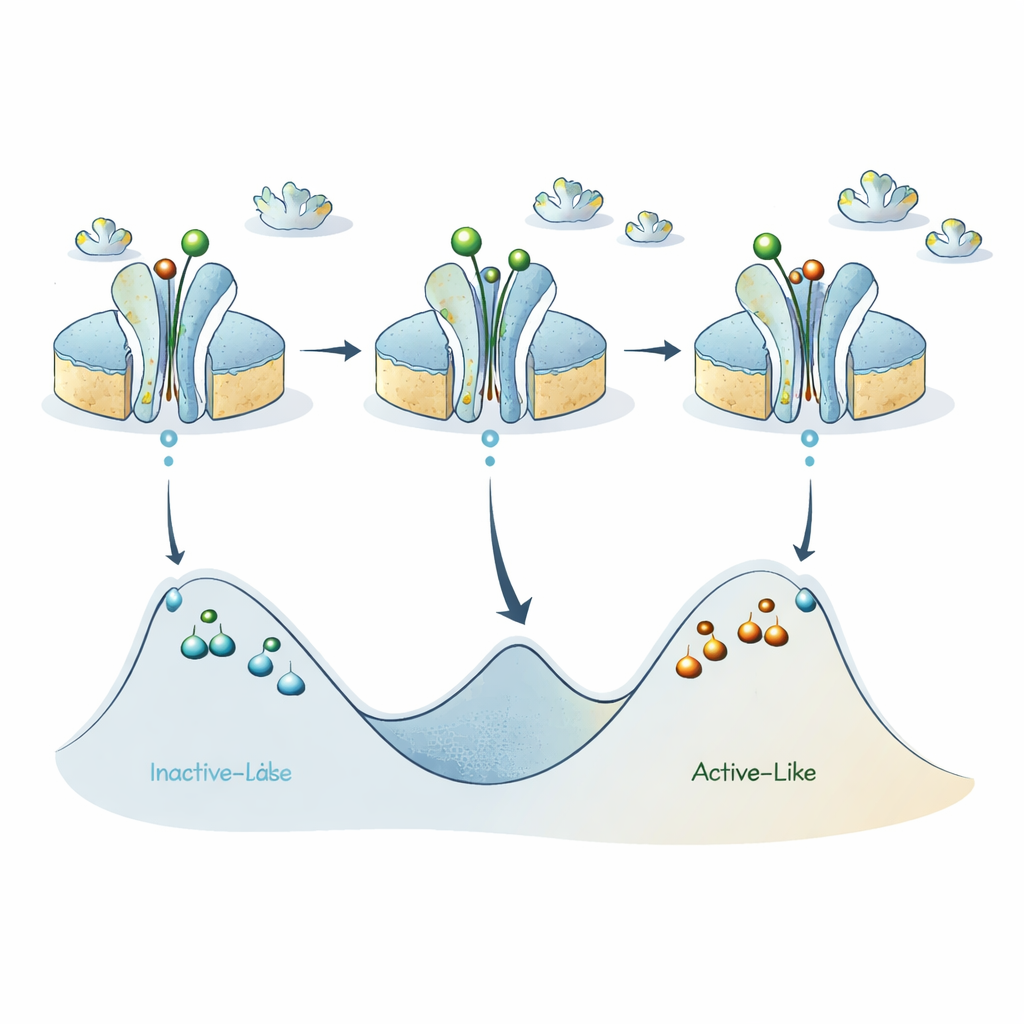
Putting these pieces together, the authors refine an earlier model in which the A2A receptor toggles mainly between an inactive‑like state and an active‑like state. Their new data suggest that each of these two broad categories actually hides several sub‑states separated by sizable energy barriers, so that individual receptors can remain “stuck” in a given version of active‑like or inactive‑like for surprisingly long times. Activating ligands lower the barrier between the main inactive‑ and active‑like basins, promoting rapid switching on the sub‑millisecond scale, but the internal barriers within each basin stay high, giving rise to the long‑lived sub‑states that ABEL‑FRET detects.
What This Means for Future Medicines
For non‑specialists, the key message is that a drug target such as the A2A receptor does not simply flip between “off” and “on.” Instead, it explores a rugged landscape of shapes, some of which persist long enough to matter for how signals are transmitted inside cells and how drugs work over time. By extending how long individual receptors can be watched in their natural, untethered state, ABEL‑FRET fills a crucial gap between ultrafast and very slow measurements. This approach can now be applied to many membrane proteins, offering a more complete, time‑resolved picture of how potential drug targets breathe, shift, and respond to therapeutic compounds.
Citation: Maslov, I., Borshchevskiy, V., Pérez, I. et al. ABEL-FRET bridges the timescale gap in single-molecule measurements of the structural dynamics in the A2A adenosine receptor. Commun Chem 9, 114 (2026). https://doi.org/10.1038/s42004-026-01941-8
Keywords: G protein-coupled receptors, single-molecule FRET, adenosine A2A receptor, protein conformational dynamics, ABEL trap