Clear Sky Science · en
Mechanistic study on the sulfate migration in glycosaminoglycans during MS fragmentation
Why shifting chemical tags on sugars matters
Our cells are coated with long sugar chains that help control processes as diverse as blood clotting, immune defense, and how viruses latch onto cells. Many of these chains carry tiny chemical tags called sulfate groups whose exact positions along the sugar backbone act like a molecular barcode, telling proteins when and where to bind. Scientists rely heavily on mass spectrometry, a powerful weighing technique, to read these barcodes. This study reveals that during such measurements, sulfate tags can quietly shift positions, potentially fooling researchers about how these biologically crucial sugars are really built.
Complex sugar chains with critical tags
Glycosaminoglycans are long, linear sugar chains often attached to proteins on cell surfaces. Their sulfate decorations are not random; slight changes in where a sulfate group sits on a given sugar unit can radically alter how the chain interacts with growth factors, clotting proteins, or pathogens. Because of this, scientists try to determine not just how many sulfates are present but exactly where they are placed. Mass spectrometry, often combined with controlled breakage of the sugar chains, is one of the main ways to do this. However, previous hints suggested that charged groups like sulfates might move around during the measurement, complicating efforts to read out the true pattern of modifications.
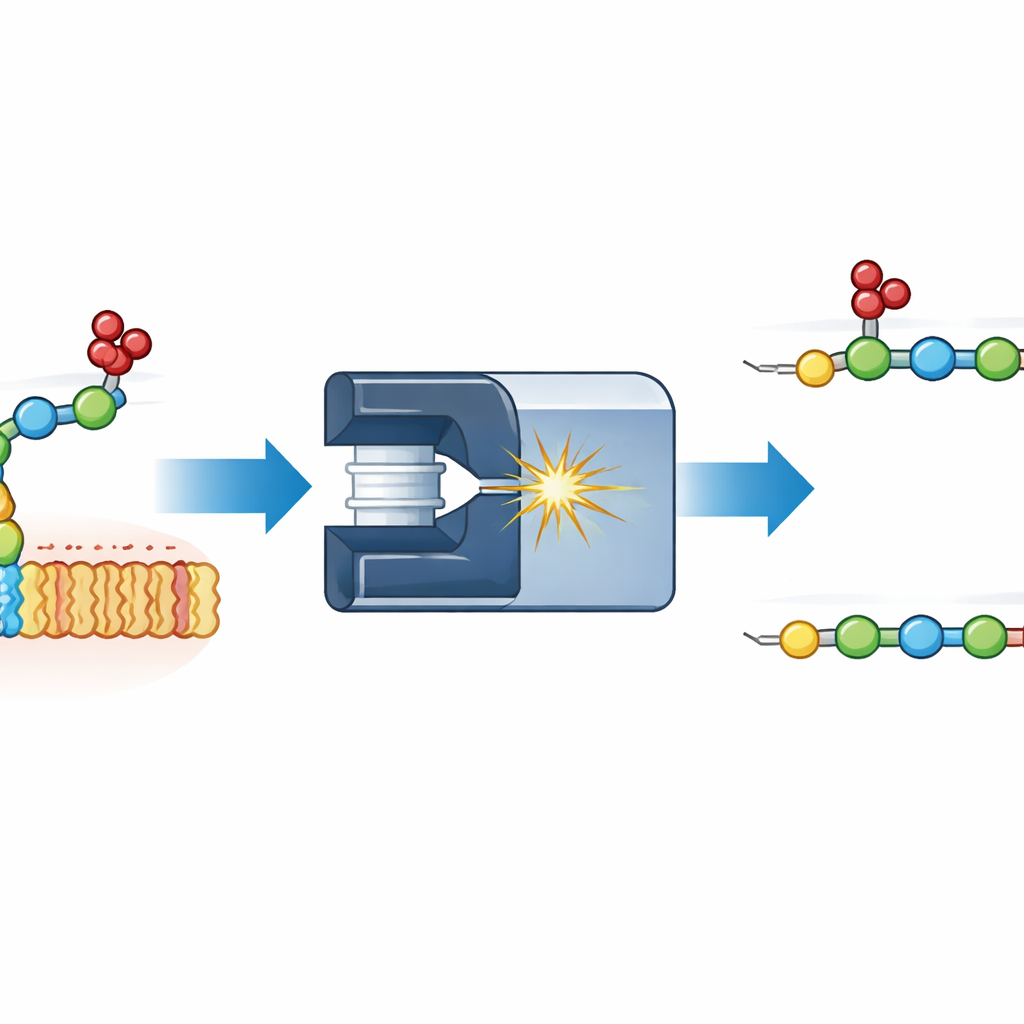
Watching sulfates move during measurement
The authors focused on a simple model: a two-sugar fragment of heparan sulfate, one of the best-known glycosaminoglycans. They attached different fluorescent labels to one end of the disaccharide and fragmented it inside a mass spectrometer. By measuring how the resulting pieces drifted through a gas under an electric field, a technique called ion mobility, they could distinguish shapes that otherwise have the same mass. An unexpected fragment appeared that was heavier by exactly one sulfate group in the wrong place: instead of staying on the first sugar, the sulfate had migrated onto the second. Comparisons with carefully synthesized reference compounds showed that the migrating sulfate could land at two distinct spots on the second sugar, giving two different shapes that were cleanly resolved by ion mobility.
Pinpointing new landing sites and testing labels
To better understand where the sulfate ended up and whether other positions were possible, the team combined their measurements with detailed computer simulations. They calculated many possible three-dimensional shapes for the candidate structures and predicted how each should travel through the gas phase. Only sulfates located at two specific sites on the second sugar—known to specialists as the 6O and 3O positions—matched the experimental behavior, while other hypothetical locations appeared unlikely. The researchers then tested whether the attached labels themselves might be driving the reshuffling, by swapping the original label for three simpler ones. In every case, sulfate migration persisted and produced similar kinds of fragments, indicating that the label choice has little effect on whether migration happens, though it can subtly alter how easily the different products can be told apart.
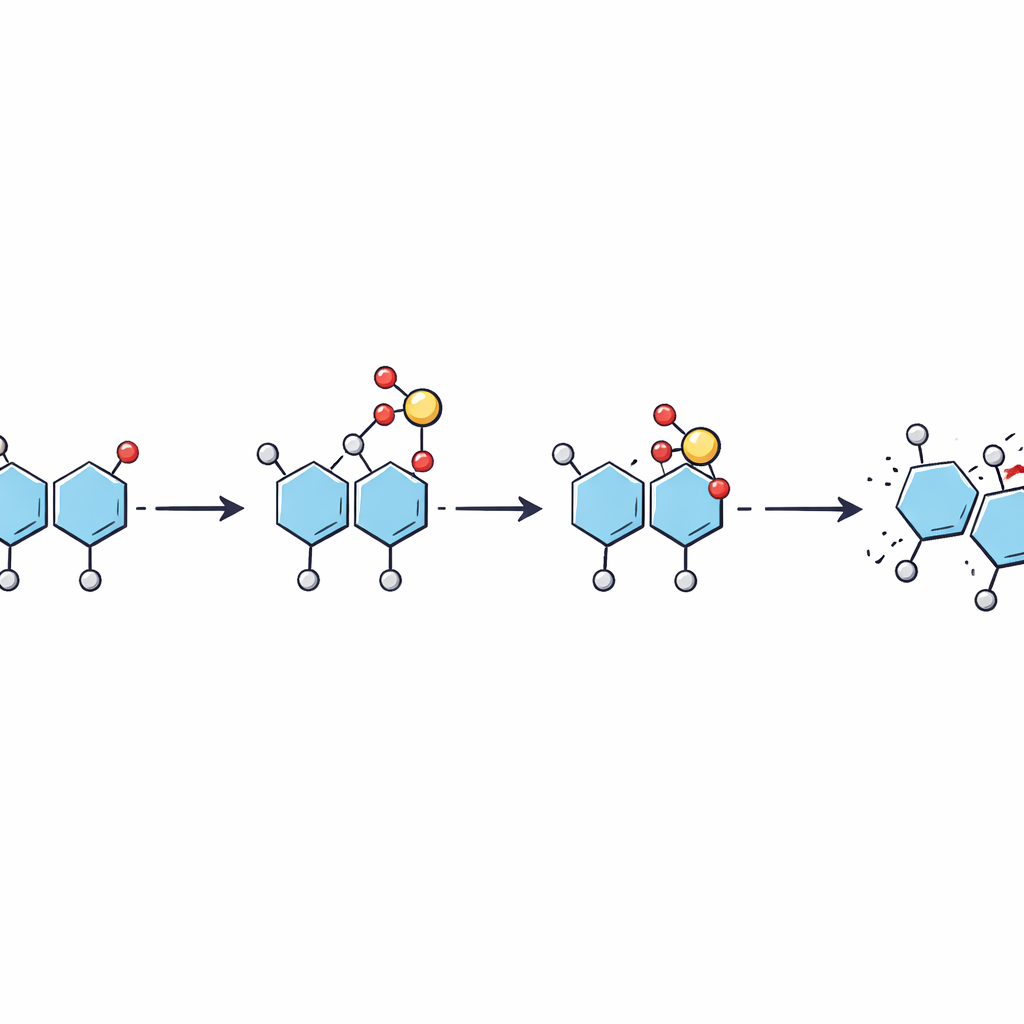
Stepwise hopping instead of a single jump
Using energy-tuned fragmentation experiments together with further simulations, the authors pieced together a step-by-step picture of how the sulfate moves. As the ion is energized in the mass spectrometer, a mobile proton first activates the sulfate group, which then hops from the original sugar onto a particular site on the neighboring sugar as the bond between them breaks. This produces a fragment in which the sulfate sits in an intermediate position. With additional energy, the sulfate can shift again along the same sugar to a more stable site. The work suggests that these rearrangements can occur at lower energies than those needed to break the sugar backbone, meaning they can happen quietly in the background of routine analyses.
What this means for decoding sugar structures
For non-specialists, the key message is that the chemical tags on important biological sugars are not always frozen in place during analysis; they can slide along the chain while the molecule flies through the mass spectrometer. The study shows in detail that, at least for a representative heparan sulfate fragment, a sulfate group can migrate from one sugar unit to another and then settle at new positions, producing misleading fragments that mimic genuine structural features. This means that some past and future measurements could misread the sulfate code unless additional techniques, such as ion mobility and advanced modeling, are used to catch these hidden moves. The work calls for more systematic studies to learn how widespread such sulfate migration is, so that researchers can more reliably connect sugar patterns on cell surfaces to health and disease.
Citation: Polewski, L., Yaman, M., Tokić, M. et al. Mechanistic study on the sulfate migration in glycosaminoglycans during MS fragmentation. Commun Chem 9, 130 (2026). https://doi.org/10.1038/s42004-026-01939-2
Keywords: heparan sulfate, glycosaminoglycans, mass spectrometry, sulfate migration, ion mobility