Clear Sky Science · en
Binding pose depth modulates photoswitchable ligands’ efficacy at the 5-HT2A receptor
Shining Light on Switchable Brain Drugs
Imagine a medicine you could turn on and off with a flash of light, precisely steering brain receptors while avoiding side effects. This study explores exactly that idea for a key serotonin receptor linked to mood, perception, and psychedelic drugs. The researchers dug into why two nearly identical light-sensitive molecules behave so differently—one acting almost like a perfect on/off switch, the other stubbornly refusing to turn fully off. Their answer comes down to a surprisingly simple factor: how deep the molecule sits inside the receptor.
Light-Driven Medicines in the Brain
Light-activated drugs, also called photopharmacological tools, are designed so that a beam of light flips part of the molecule between two shapes, like a bent and a straight version. Those shapes can change how strongly the drug grips its target. In this work, the target is the human 5-HT2A receptor, a protein on brain cells that responds to serotonin and is central to both antipsychotic and psychedelic effects. The molecules studied are derivatives of N,N-dimethyltryptamine (DMT), modified with a light-sensitive azobenzene unit. Two versions differ only in where a tiny methoxy group is placed on a ring—either in a “para” or “meta” position—yet their biological behavior under light is dramatically different.
Two Nearly Twin Molecules, Very Different Switches
In cell tests, the para version, called compound 1, behaves almost like a digital switch. In the dark, its “trans” shape barely activates the receptor and even blocks it slightly, acting like a weak antagonist. When light flips it into the “cis” shape, the same molecule becomes a moderate activator, turning the receptor partly on. The meta version, compound 2, refuses to cooperate in this way: both its light and dark forms keep the receptor fairly active, never delivering a true “off” state. The central puzzle of this paper is why such a minimal chemical tweak—just sliding one small group around a ring—so strongly changes how the receptor responds.
Peering Inside with Atom-Level Movies
To solve this, the team used extensive all-atom molecular dynamics simulations, effectively running detailed movies of every atom in the receptor, the surrounding membrane, water, and each ligand over a total of about 80 microseconds. They simulated both molecules in each of their light-controlled shapes, and in both inactive and active forms of the 5-HT2A receptor. By tracking known structural “microswitches” inside the protein—such as a key tryptophan that flips, a salt bridge that breaks, and the motion of sodium and water deep in the core—they could tell when the receptor was leaning toward an off or on state. They also compared how closely the new molecules mimicked the binding posture of LSD, a well-studied partial activator, using the overlap of their ring systems as a guide.
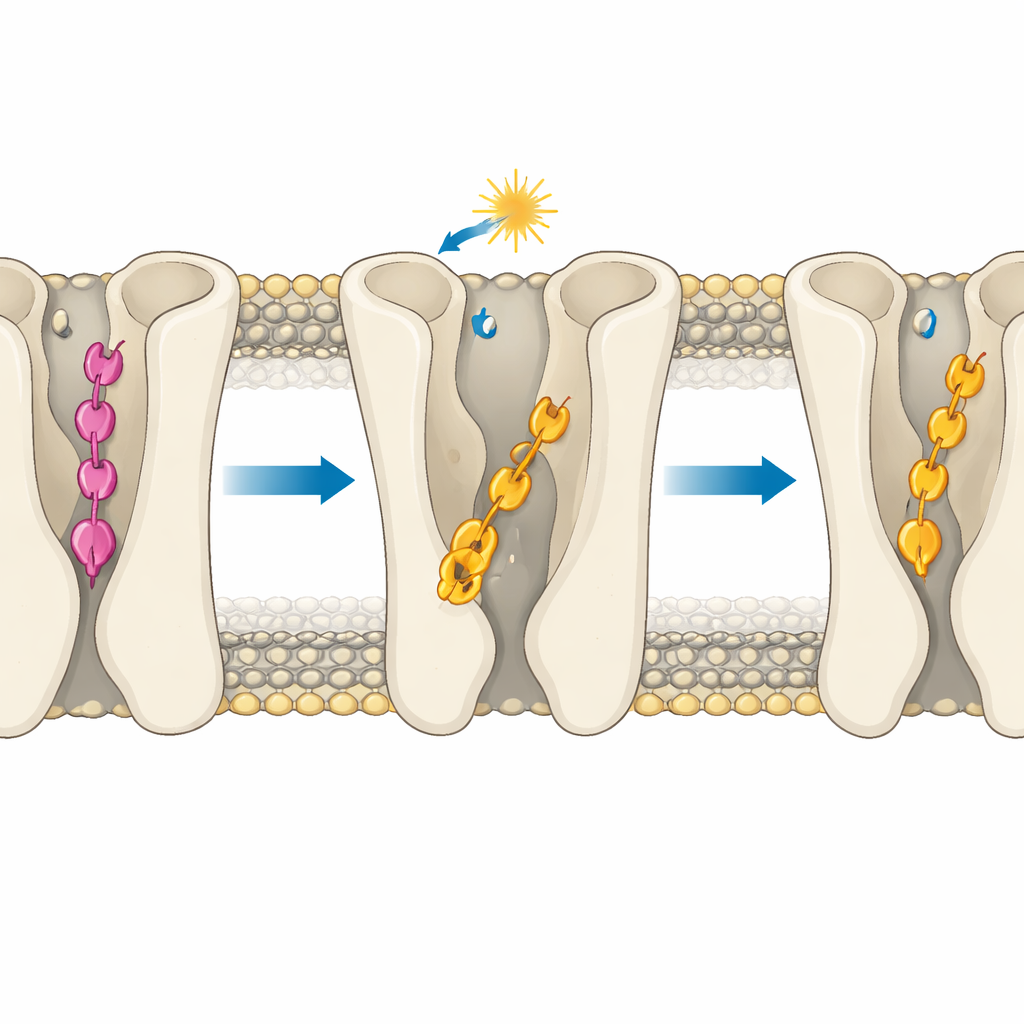
Depth Matters More Than Shape Alone
The core insight is that the molecules’ effectiveness is governed mainly by how deep they insert into the receptor’s binding pocket. In the inactive receptor, the para compound’s trans form makes a special set of hydrogen bonds that pull it unusually deep, below the region where typical activating contacts occur. This “over-insertion” prevents it from touching key polar residues that help switch the receptor on, so it stabilizes the off state. The meta compound’s trans form cannot make the same anchoring contacts and instead sits higher up in a more LSD-like pose that is compatible with activation, explaining its residual activity. When light flips both molecules into their cis forms, they generally move into shallower, more activator-like positions. Yet even here, geometry matters: in the active receptor, cis compound 2 can slide deeper into a hydrophobic tunnel between two helices and form a persistent hydrogen bond that reinforces its strong agonist behavior, while cis compound 1 is sterically blocked from doing so.
Fine Control of Internal Water and Ions
The simulations also show that binding depth subtly tunes an internal sodium pocket and water pathway that are known to influence receptor activation. Deep, rigid binding by the para compound in its trans form keeps a sodium ion tightly caged and the surrounding region relatively dry, features associated with an off state. In contrast, the more mobile binding of the meta compound or the cis forms allows more water to invade and loosens the sodium environment, priming the receptor for activation. A key aromatic “toggle switch” residue can flip orientation more readily when ligands are flexible and not over-anchored, further nudging the receptor toward an on-like state, especially with cis compound 2.
Design Rules for Future Light-Controlled Drugs
For a non-specialist, the message is that the exact depth at which a drug sits in its receptor pocket can make the difference between off, partly on, and strongly on—even when the chemical change looks tiny on paper. By showing how one photoswitchable ligand can over-insert and lock the receptor off, while a closely related one stays at an activation-friendly depth, the study offers a clear design rule: control insertion depth as carefully as you control light-induced shape changes. These insights could guide the creation of next-generation light-sensitive compounds that treat brain disorders with unprecedented precision, turning signaling pathways up or down in a controlled, reversible way while minimizing side effects.
Citation: Weber, V., Salvadori, G., Natale, F. et al. Binding pose depth modulates photoswitchable ligands’ efficacy at the 5-HT2A receptor. Commun Chem 9, 121 (2026). https://doi.org/10.1038/s42004-026-01936-5
Keywords: photopharmacology, serotonin 5-HT2A receptor, photoswitchable ligands, GPCR activation, molecular dynamics simulations