Clear Sky Science · en
RNA−Iron complexes catalyse prebiotic oxygen generation
Ancient Air From an Oxygen-Free World
Long before plants began pumping oxygen into the sky, Earth’s surface was largely starved of breathable air. Yet life still had to survive occasional bursts of damaging chemicals such as hydrogen peroxide, a close cousin of household bleach. This study explores a surprising possibility: simple RNA molecules, working together with dissolved iron, may have quietly generated small amounts of oxygen and helped early life cope with toxic chemistry billions of years before modern enzymes and photosynthesis evolved. 
A Young Planet With Hidden Dangers
When the earliest ancestors of life emerged more than four billion years ago, Earth’s atmosphere held almost no free oxygen. The seas, however, were rich in soluble iron, and natural processes such as sunlight hitting minerals and rocks reacting with water could create reactive oxygen species, including hydrogen peroxide. These molecules are double‑edged swords: they can drive useful chemistry but also damage delicate biological structures. Geological and genetic clues suggest that even the earliest organisms needed ways to manage these bursts of oxidative stress, long before sophisticated protein enzymes and plant‑like photosynthesis existed.
RNA and Iron Team Up
The researchers focused on RNA, the versatile genetic and catalytic polymer thought to play a starring role in life’s origins. They noticed that a specific metal‑binding pocket in a modern ribosomal RNA resembles the way iron is held inside heme, the reactive center of today’s peroxide‑destroying enzymes. This structural mimicry raised a question: could RNA, when bound to iron instead of its usual magnesium, act like a primitive catalyst for breaking down hydrogen peroxide into harmless water and molecular oxygen? To probe this, they tested several short and long RNA fragments, as well as RNA‑like molecules with slightly different backbone chemistry, under oxygen‑free, iron‑rich conditions meant to echo early Earth’s environment.
Testing Tiny Catalysts
Using a color‑changing “blue bottle” reaction that reports the appearance of oxygen, the team found that most of the RNA constructs, when partnered with ferrous iron, sped up the decomposition of hydrogen peroxide. The full‑length ribosomal RNA showed the strongest effect, but a much smaller three‑letter RNA segment (the universal CCA tail found at the end of transfer RNAs) and a ribosome‑mimicking RNA analog also worked. A two‑letter RNA lacking the right arrangement of phosphate groups did not, underscoring the importance of how the backbone holds the metal. Further measurements suggested that active complexes use four nearby oxygen atoms from the RNA backbone to tightly cradle a single iron ion, echoing the four nitrogen atoms that bind iron in heme. Kinetic analysis showed that at least one of these RNA–iron systems behaves much like a rudimentary enzyme, with reaction rates that increase and then level off as hydrogen peroxide concentration rises. 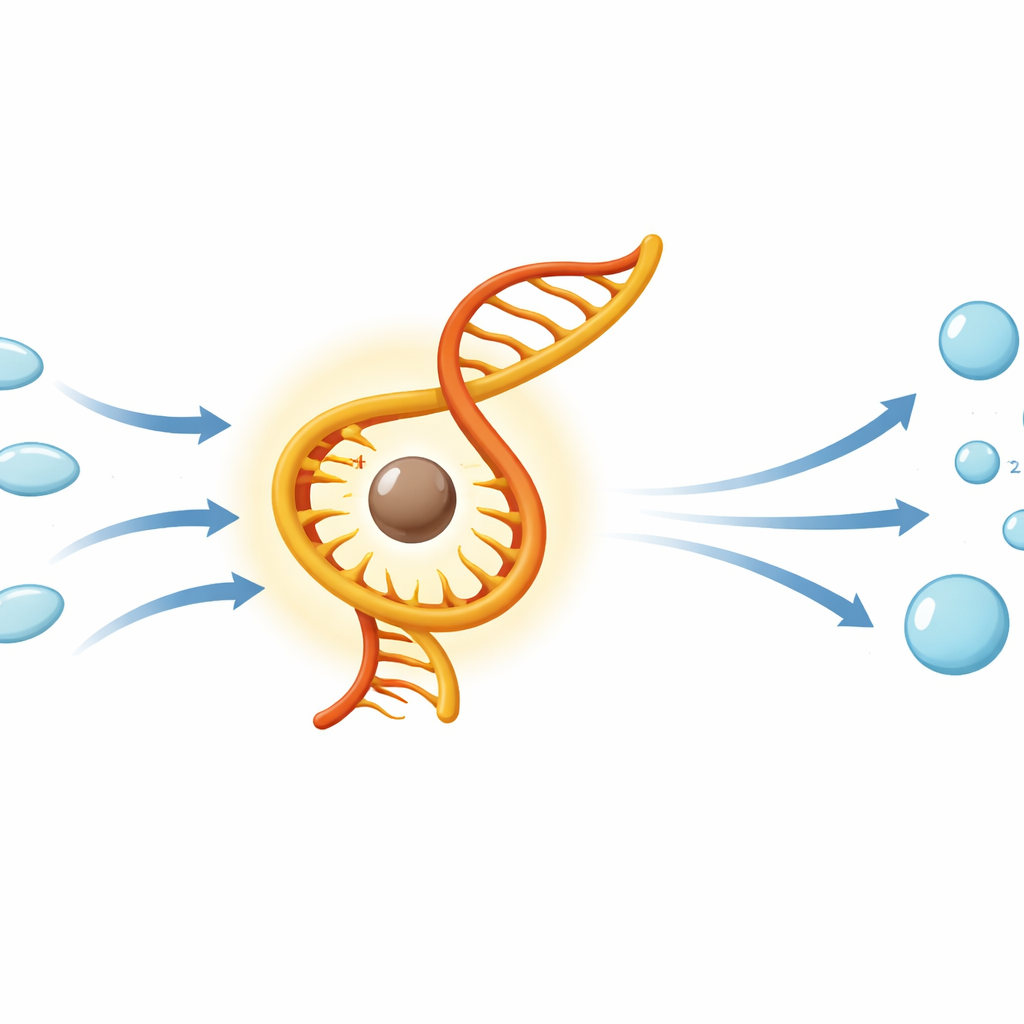
Watching Electrons Move
To peek into the reaction’s inner workings, the authors turned to electron paramagnetic resonance spectroscopy, a technique that senses unpaired electrons in metal centers. When CCA RNA, iron, and hydrogen peroxide were mixed, the magnetic signature of the iron changed over time, revealing intermediate high‑energy states similar to those seen in modern iron‑based enzymes that break down peroxide. Signals consistent with a fleeting “ferryl” species—iron in an unusually oxidized state linked to a nearby radical—appeared and then faded as the reaction proceeded. Over longer periods, the iron ended up in a more oxidized form, but it remained dissolved, suggesting that RNA not only helped drive the chemistry but also kept otherwise insoluble iron suspended in solution.
Rewriting Early Oxygen Stories
The authors propose that such RNA–iron complexes could have acted as early molecular guardians, detoxifying hydrogen peroxide and, as a side effect, releasing small pulses of molecular oxygen into otherwise oxygen‑free environments. They do not argue that this mechanism alone oxygenated the planet; photosynthetic organisms later did the heavy lifting. Instead, they suggest that RNA’s ability to both generate and withstand oxidative conditions may have given it a survival edge, helping to shape the chemistry of life before proteins took over most catalytic duties. In this view, traces of oxygen on the young Earth may have been, at least in part, the quiet handiwork of primitive RNA bound to iron.
Citation: Wang, YC., Tu, JH., Yu, LC. et al. RNA−Iron complexes catalyse prebiotic oxygen generation. Commun Chem 9, 124 (2026). https://doi.org/10.1038/s42004-026-01935-6
Keywords: origin of life, early Earth chemistry, RNA catalysis, reactive oxygen species, prebiotic oxygen