Clear Sky Science · en
Scaffold-client behavior and structural organization in multicomponent protein condensates as revealed by studying tau/TDP-43 droplets
Proteins That Behave Like Tiny Oil Droplets
Inside our cells, many proteins do not stay evenly mixed in the watery interior. Instead, they clump into tiny liquid droplets, a bit like oil in water. This study looks at how two such proteins, tau and TDP-43, which are closely linked to Alzheimer’s disease and other dementias, gather into droplets together. By uncovering how these droplets form, mix, and organize, the work offers clues to why harmful protein aggregates appear in aging brains and how we might one day control them.
Why These Two Brain Proteins Matter
Tau and TDP-43 are both known to form toxic aggregates in neurodegenerative diseases. Traditionally, scientists studied each of them on its own, but autopsies of human brains increasingly show that both proteins can end up in the same damaged regions. That raises a key question: when tau and TDP-43 are present together in cells, how do they share space inside droplets, and does this shared environment encourage disease-related aggregation? The authors chose a simplified but realistic model using tau and the low-complexity tail of TDP-43 (the LCD), which is the portion most responsible for droplet formation and eventual solidification.
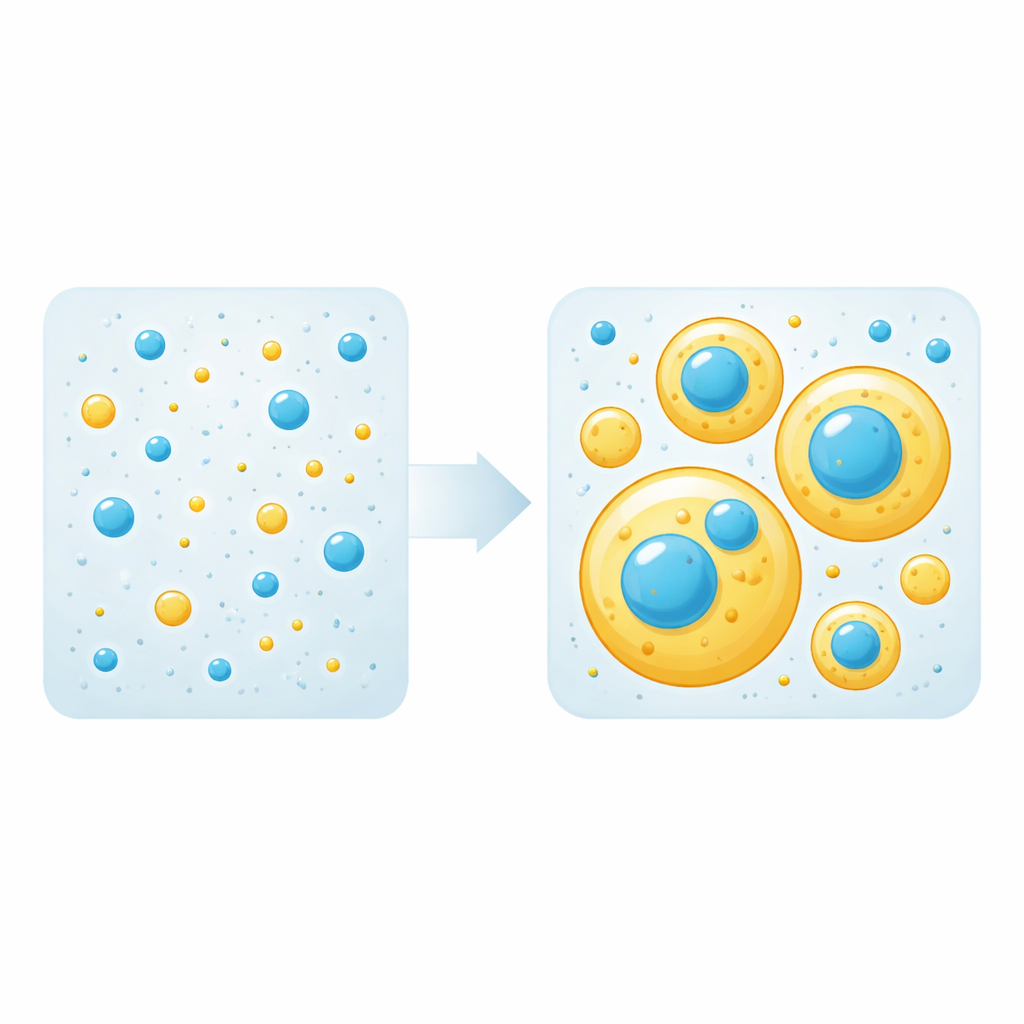
Droplets, “Scaffolds,” and “Clients”
Liquid droplets like these form when protein–protein attractions outweigh protein–water attractions, causing a solution to split into a dense phase (droplets) and a dilute phase. In mixtures, only some proteins are needed to build droplets; these are called “scaffolds.” Others simply hitch a ride into droplets as “clients.” The team first measured the concentration threshold at which each protein alone begins to form droplets. They then mixed different amounts of tau and TDP-43 LCD and watched, using fluorescence microscopy, whether droplets appeared and how the proteins arranged themselves. Strikingly, they found that either protein can play the role of scaffold or client depending purely on its concentration: above its own threshold it builds droplets, below that threshold it is passively recruited into droplets formed by the other.
Layered Droplets and a Mysterious Halo
When both proteins were abundant enough to form droplets on their own, they did not blend into a single uniform droplet. Instead, larger tau-rich droplets wrapped around smaller TDP-43–rich droplets, creating a layered, multiphase structure. Measurements of how the droplets spread on glass showed that TDP-43 droplets have higher surface tension, so it is energetically favorable for them to sit inside the more wetting tau droplets. An eye-catching feature emerged at these interfaces: tau accumulated at especially high levels as a bright “halo” coating the surface of TDP-43 droplets, even when tau itself was below its own droplet-forming threshold. Over time, TDP-43–rich regions tended to stiffen into a more solid-like material, while tau-rich regions remained liquid, suggesting different propensities for turning into long-lived aggregates.
Tuning Interactions and Probing the Microscopic Mechanism
The authors next tested how specific molecular forces control which protein acts as scaffold or client. They selectively weakened hydrophobic (water-repelling) interactions that drive TDP-43 LCD condensation using 1,6-hexanediol, and separately weakened the electrostatic (charge-based) interactions that drive tau condensation by raising salt levels. In each case, disabling one protein’s key interactions prevented it from forming droplets and turned it into a client inside droplets of the other, mimicking what happens when its concentration is lowered. To understand the halo more deeply, they varied salt to see how much tau entered TDP-43 droplets and used computer simulations that treat each amino acid as a simplified bead. These simulations reproduced the halo and showed that the first ~40 amino acids of tau’s negatively charged N-terminal region anchor at the droplet surface, while the rest of the molecule extends outward into the surrounding liquid, creating an “amphiphilic” (part droplet-loving, part water-loving) shell. Experiments with a shortened tau fragment lacking this N-terminal stretch confirmed that, without it, the halo disappears.

What This Means for Brain Disease
Together, the results reveal a general set of rules for how multicomponent protein droplets assemble and organize. In this tau/TDP-43 system, which mirrors situations in diseased neurons, concentration and interaction strength jointly determine which proteins build droplets and which simply occupy them. Surface tension and amphiphilic arrangements then sculpt layered droplets and halos that can concentrate multiple disease-linked proteins in the same confined space, potentially encouraging them to harden into aggregates. For a lay observer, the key message is that tau and TDP-43 do not just clump randomly; they self-organize into structured liquid droplets whose architecture is governed by simple physical principles. Understanding and eventually manipulating these principles could open new avenues for slowing or preventing harmful protein buildup in neurodegenerative diseases.
Citation: Monnaka, V.U., Shipley, B., Boyko, S. et al. Scaffold-client behavior and structural organization in multicomponent protein condensates as revealed by studying tau/TDP-43 droplets. Commun Chem 9, 126 (2026). https://doi.org/10.1038/s42004-026-01933-8
Keywords: protein condensates, liquid-liquid phase separation, tau, TDP-43, neurodegeneration