Clear Sky Science · en
Nitrogen carrier gas enhancement in GC-MS via ethylene dopant improves sensitivity and preserves EI-like spectra
Keeping vital lab tools running in a helium shortage
Modern chemistry labs rely on gas chromatography–mass spectrometry (GC–MS) to track pollutants, ensure food safety, and support medical testing. Most of these instruments use helium, a gas that is becoming expensive and sometimes unavailable as reserves dwindle. This study explores whether a far cheaper and virtually limitless gas, nitrogen, can be made to work almost as well, simply by adding a small amount of ethylene. The authors show that this tweak can restore much of the lost sensitivity without changing the familiar "fingerprint" patterns that chemists depend on to identify molecules.
Why changing the carrier gas matters
GC–MS instruments separate complex mixtures in a thin column and then weigh and break apart molecules in a detector. A steady stream of carrier gas pushes molecules through the column. Helium has been the gold standard because it gives sharp peaks and strong signals, but global supply disruptions have driven up prices and even forced some labs to shut down instruments. Nitrogen is cheap and can be generated on-site from air, but under normal conditions it delivers only a tiny fraction of helium’s performance. That makes it hard to spot trace pollutants, pesticides, or other low-level targets that regulations require. Finding a way to make nitrogen-based GC–MS nearly as sensitive as helium, without new hardware or new data libraries, would be a major practical win.
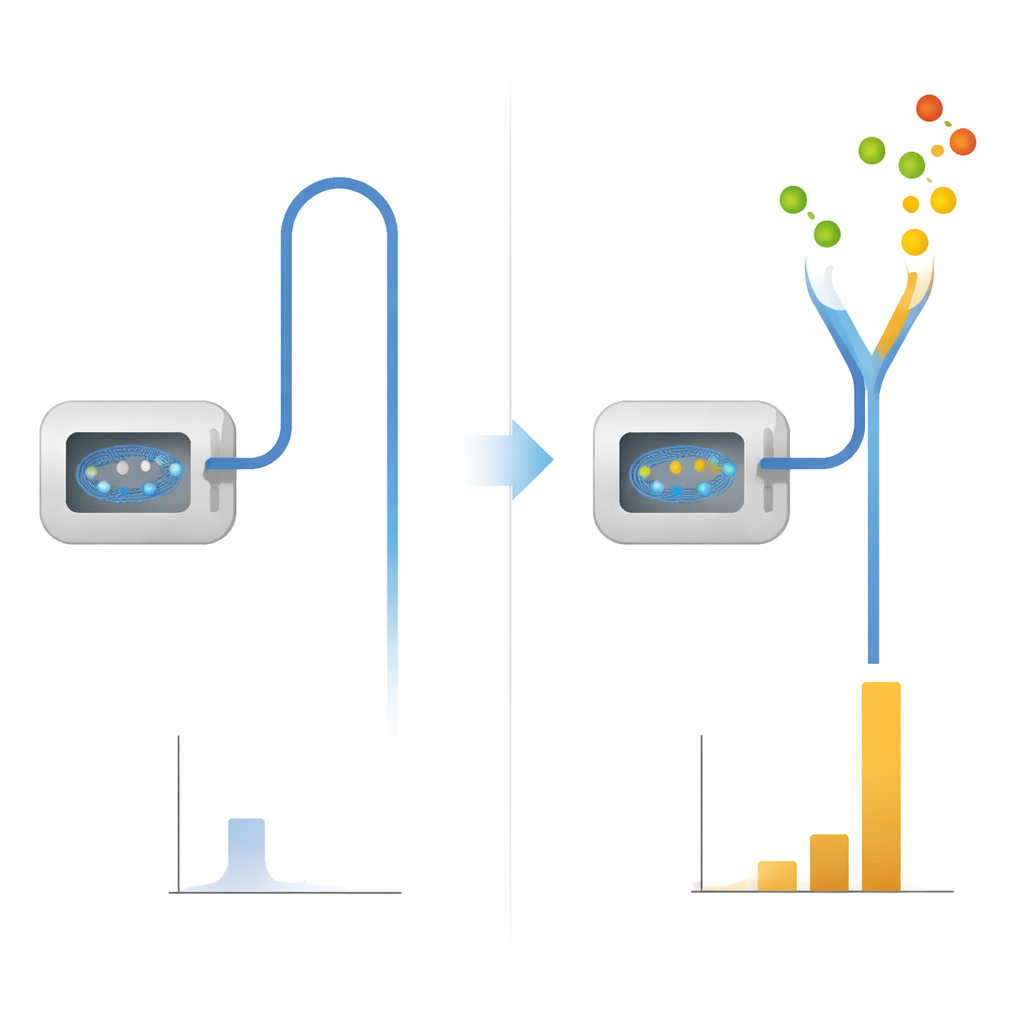
A simple tweak with a big payoff
The researchers discovered that mixing a modest amount of ethylene gas—about nine percent by volume—into nitrogen can dramatically boost signal strength in GC–MS. Under standard operating conditions, the combined nitrogen–ethylene stream produced signals roughly twenty times stronger than nitrogen alone for a range of test chemicals, including phthalate plasticizers and polycyclic aromatic hydrocarbons, both of which are tightly regulated environmental contaminants. Crucially, this gain brings performance close to what is normally achieved with helium. Just as important, the familiar fragmentation patterns produced at the usual 70-electron-volt setting remain essentially unchanged, so existing reference libraries can still be used for automatic compound identification.
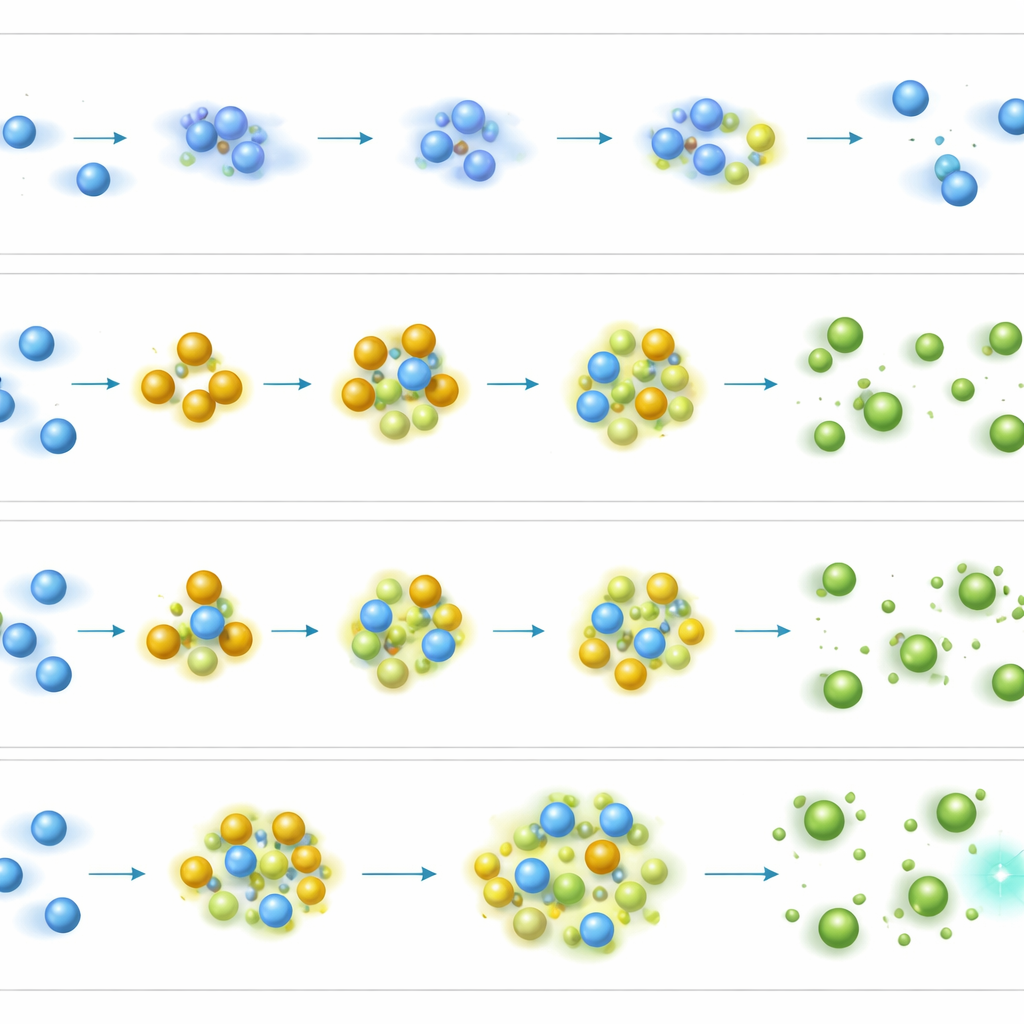
How collisions help without changing the fingerprints
At the microscopic level, the team proposes that the boost comes from a kind of energy relay among gas molecules. When electrons strike nitrogen, they form short-lived ions that normally break apart too quickly to be useful. With ethylene present, some of that energy appears to be transferred to ethylene ions that live longer and can bump into target molecules more often before they decay. Through many rapid collisions, these energized species still deliver enough punch to break molecules in the same way as standard electron ionization, preserving the characteristic fragment patterns that analysts rely on. The authors stress that this is not chemical ionization, a gentler technique that produces very different spectra; here, the spectra remain "hard," meaning they show the same rich fragmentation as classic GC–MS.
When and where the boost appears
The enhancement does not occur under all conditions. It shows up only when the gas near the ion source is dense enough that molecules frequently collide—what the authors call a collision-dominated regime. By adjusting gas flows and geometry so that the emerging jet from the column is either denser or more diffuse, they observed that the effect could be turned up, weakened, or even reversed. Under more rarefied, molecular-flow-like conditions, adding ethylene simply scatters electrons and dilutes the sample, reducing sensitivity. A computational model that tracks electron travel, collision rates, and hypothetical lifetimes of intermediate ions reproduces the observed “sweet spot,” where electron penetration and collision frequency are balanced to give the largest gain.
Practical promise and open questions
The work suggests that many labs could ease helium shortages by switching to nitrogen supplemented with a small ethylene flow, without buying new instruments or rebuilding spectral libraries. The technique restores much of the lost sensitivity for important pollutant and contaminant classes, and tests on different commercial GC–MS platforms show similar gains, pointing to a broadly applicable effect. At the same time, the authors are cautious about the underlying explanation: the exact intermediate species and their lifetimes have not yet been observed directly, and more detailed, time-resolved studies are needed to nail down the mechanism. For now, they present the improvement as a practical operating recipe and an intriguing example of how subtle gas-phase chemistry can help sustain critical analytical capabilities in a world where helium can no longer be taken for granted.
Citation: Fuse, Y., Chu, X. Nitrogen carrier gas enhancement in GC-MS via ethylene dopant improves sensitivity and preserves EI-like spectra. Commun Chem 9, 129 (2026). https://doi.org/10.1038/s42004-026-01930-x
Keywords: gas chromatography–mass spectrometry, helium shortage, nitrogen carrier gas, ethylene dopant, analytical sensitivity