Clear Sky Science · en
Exploring the stability of BOx at various inorganic supports
Cleaner fuels from a common element
Turning ordinary propane—the stuff in backyard grill tanks—into more valuable building blocks for plastics usually demands hot, energy-hungry processes that also generate a lot of carbon dioxide. This study explores how compounds of boron, a relatively abundant element, can help perform this transformation more gently and cleanly. By uncovering how boron behaves on different solid surfaces at high temperatures, the authors point to new ways of designing catalysts that make useful chemicals with less waste and lower energy use.
Why propane-to-plastics chemistry matters
Modern life relies heavily on light olefins such as propene and ethene, which are key ingredients for plastics, fibers, and countless everyday materials. Today, most of these compounds are made from oil or natural gas using energy-intensive routes that release large amounts of CO₂. An alternative process, called oxidative dehydrogenation of propane, can in principle make these olefins at lower temperatures and with fewer unwanted byproducts. Boron-based materials have recently emerged as promising catalysts for this reaction because they are highly selective: they favor turning propane into olefins instead of burning it all the way to CO₂. Yet researchers still do not fully understand what the “active” boron species look like or where exactly the reaction takes place—on the catalyst surface, in the gas phase, or both.
Surprising mobility of boron in hot reactors
The authors focused on boron oxide, a simple boron–oxygen compound often written as BOx, supported on three common inorganic materials: pure silica, pure alumina, and a mixed silica–alumina. Using a combination of techniques that track gases leaving the surface as it is heated, as well as methods that probe the local structure of atoms in solids, they showed that boron does not always stay put. On silica, boron oxide tends to form loosely bound clusters that can evaporate, creating volatile boron-containing species in the gas stream. On alumina-rich supports, by contrast, boron bonds more tightly to aluminum-linked oxygen atoms, forming a more stable, glass-like network that resists leaching into the gas phase. Simple washing tests reinforced this picture: most of the boron could be rinsed away from silica-supported samples, but much less from alumina-based ones.
Linking boron stability to catalytic behavior
These differences in boron mobility turned out to correlate closely with how the catalysts behaved in the propane reaction. Silica-supported boron oxide began converting propane to olefins at temperatures about 80 °C lower than catalysts containing alumina, even though all three systems eventually showed very similar relationships between how much propane they converted and how selectively they made olefins. Heating the samples while monitoring boron-containing fragments in the gas phase revealed that silica released far more boron oxide and related species at reaction temperatures than alumina did. This suggests that supports that let boron escape more easily can trigger the reaction sooner, because more reactive boron-containing intermediates enter the gas phase where they can start chain reactions that transform propane.
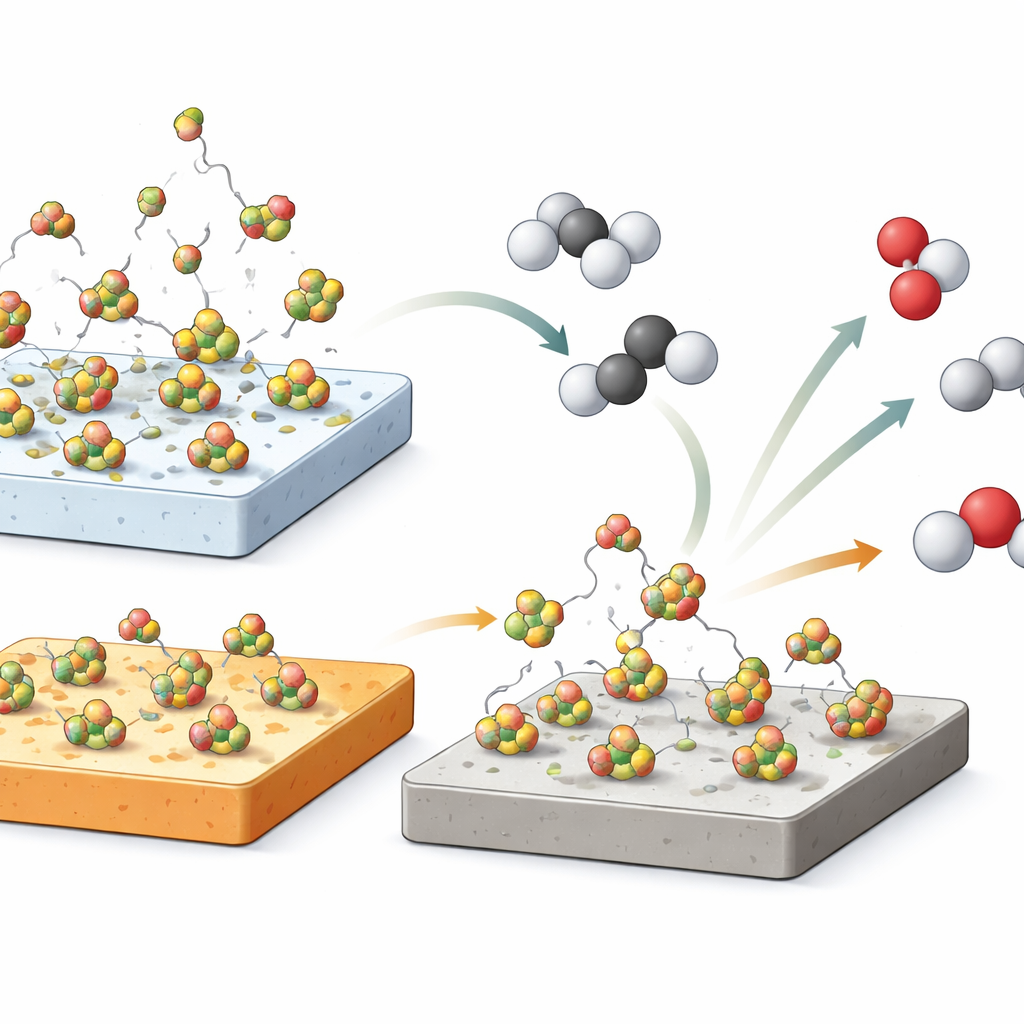
Gas-phase boron as an invisible helper
To test whether boron in the gas alone could drive the chemistry, the researchers carried out a striking experiment: they removed the solid catalyst entirely and simply injected a tiny pulse of boric acid solution, equivalent to about one-seventieth of the boron normally present on a catalyst, directly into a hot, empty reactor. As the solution rapidly decomposed to boron oxide at 500 °C, the propane conversion jumped by roughly 20%, with olefin selectivity similar to that observed over the solid catalysts. A control test with pure water produced only a small, short-lived effect. Together with the desorption measurements, this result strongly indicates that volatile boron species in the gas phase play an important role, likely by initiating radical chains that turn propane into propene and ethene.
What this means for future catalysts
For non-specialists, the main takeaway is that the solid support under a boron-based catalyst is not just an inert scaffold—it actively controls how much boron can escape into the gas and, with it, how easily the reaction starts. Supports like silica, which release boron more readily, lead to propane activation at lower temperatures, while alumina-rich supports hold onto boron more tightly and require higher temperatures, even though all end up with similar product selectivities once the reaction is underway. This insight suggests that carefully tuning how strongly boron is anchored to its support could allow chemists to design catalysts that balance stability and activity, enabling cleaner, more energy-efficient routes from simple fuels like propane to the molecules that underpin modern materials.
Citation: Johánek, V., Wróbel, M., Knotková, K. et al. Exploring the stability of BOx at various inorganic supports. Commun Chem 9, 116 (2026). https://doi.org/10.1038/s42004-026-01926-7
Keywords: boron oxide catalysts, oxidative dehydrogenation of propane, gas-phase radical chemistry, silica and alumina supports, olefin production