Clear Sky Science · en
Synergistic cation-facet effects boost alkaline hydrogen evolution kinetics on stepped Pt surfaces
Why this matters for clean energy
Producing clean hydrogen from water is a key piece of many net‑zero energy plans, but today’s most practical devices waste energy because the reaction that makes hydrogen bubbles can be surprisingly slow in alkaline (basic) solutions. This study digs down to the level of individual atoms and water molecules to explain why some platinum surfaces work better than others, and how common dissolved ions in the liquid can team up with the metal surface to speed hydrogen production.
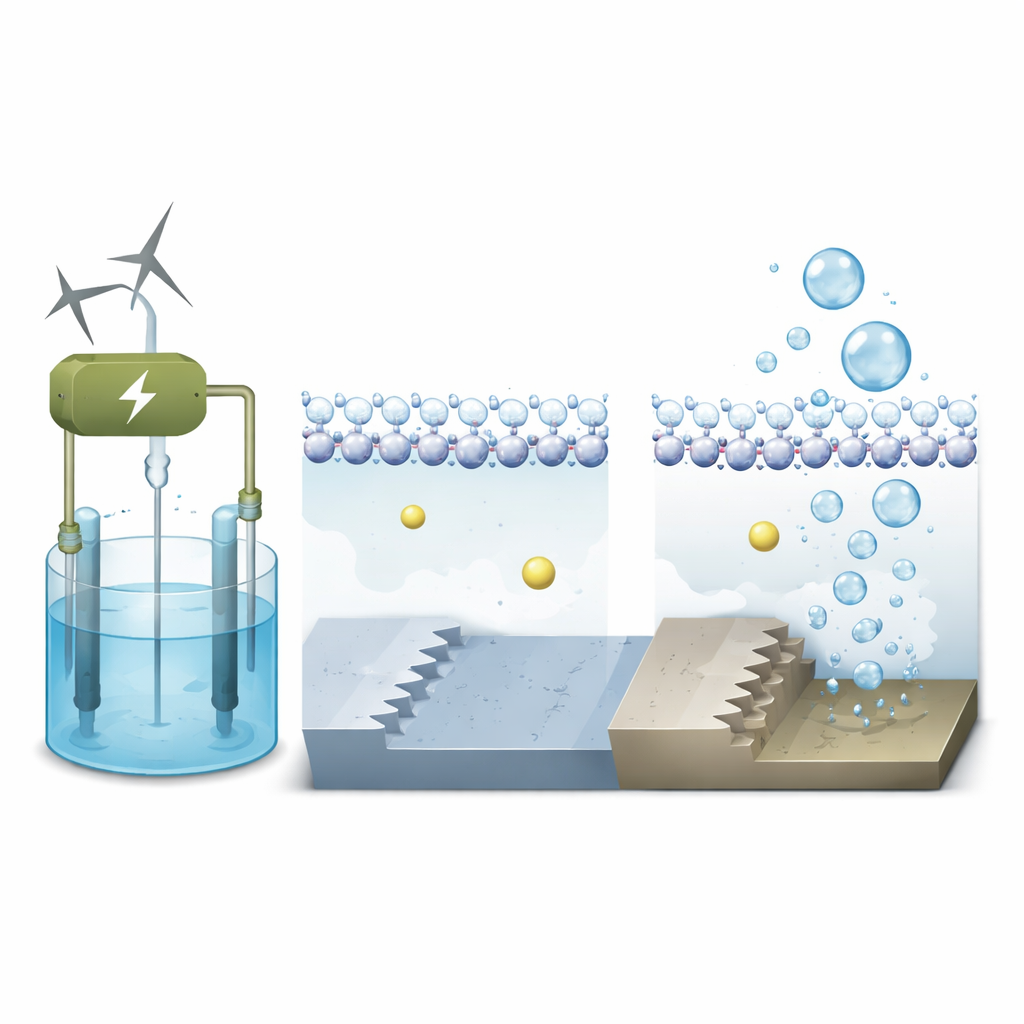
Shaping metal surfaces to guide water
The researchers focus on platinum, the benchmark material for splitting water, and compare two kinds of atomic landscapes on its surface. One is flat, like a smooth terrace (called Pt(111)); the other is stepped, like a tiny staircase (Pt(311)). Using advanced quantum‑mechanical simulations that include both electrons and moving water molecules, they model how these different surfaces behave under realistic operating voltages in an alkaline electrolyzer. The goal is to see how the local environment right next to the metal—where water, ions, and electrons meet—controls the speed of the first step in forming hydrogen.
Salt ions as hidden helpers
In alkaline electrolyzers, the liquid contains alkali metal ions such as sodium. These positively charged ions do more than just float around; they can cluster near the electrode and subtly reshape the electric field at the interface. The simulations show that on the flat platinum terrace, water forms a tight, orderly layer with hydrogen atoms pointing down toward the negatively charged surface. Sodium ions stay farther away, separated from the metal by this water sheet, so their influence on the reaction is modest. On the stepped surface, however, the under‑coordinated atoms at the step edge bind water more strongly and create a rougher, more disordered local water structure.
A special near‑surface cluster that pulls ions closer
At these step edges, the team discovers a recurring structural motif: a water molecule attached directly to the platinum step that, in turn, holds a nearby sodium ion surrounded by additional water molecules. This compact platinum–water–sodium cluster effectively drags the ion about 2.3 angstroms closer to the metal than on the flat surface. Being closer greatly strengthens the local electric field in this tiny region, which strongly polarizes neighboring water molecules. The simulations reveal that one O–H bond in these water molecules stretches more than usual, indicating that it is partway toward breaking even before the reaction formally proceeds.
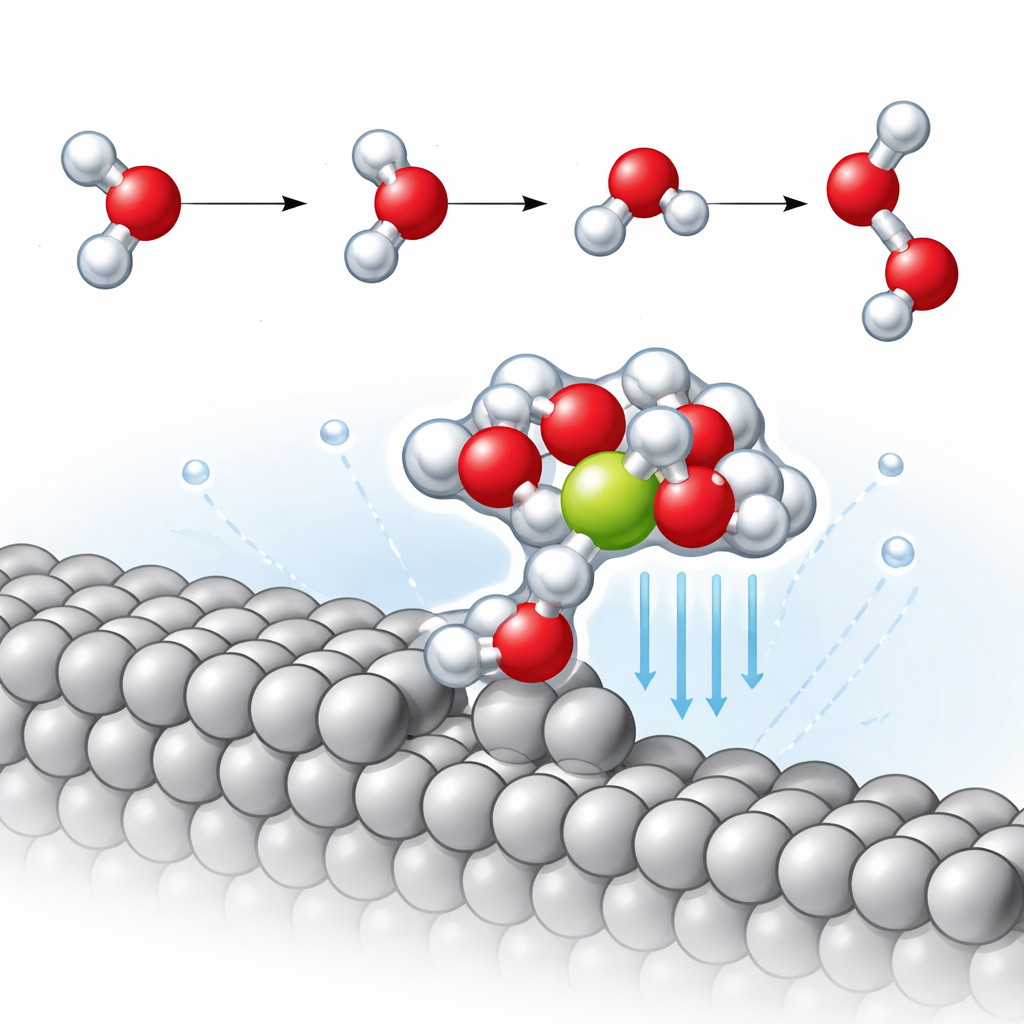
Lowering the barrier for making hydrogen
The key slow step in alkaline hydrogen evolution is known as the Volmer step, where a water molecule splits: hydrogen binds to the metal while the remaining hydroxide drifts into the liquid. By tracing the minimum‑energy reaction pathway, the authors find that on the flat platinum surface, sodium has only a small effect on the energy barrier for this step. In contrast, on the stepped surface, the nearby sodium‑stabilized cluster lowers the activation energy by about 0.14 electronvolts—roughly three times the improvement seen on the flat facet. Detailed analysis of vibrational motions shows that the O–H bond pointing toward the surface becomes significantly weaker in the presence of the close‑lying ion, making it easier to break and thereby speeding hydrogen formation.
A design rule for better hydrogen catalysts
Overall, the study concludes that the best performance in alkaline hydrogen production does not come from the metal surface or the electrolyte alone, but from their synergy. Stepped platinum sites that can anchor special water–ion clusters pull alkali cations close, intensify the local electric field, partially pre‑break water bonds, and strongly accelerate the crucial first step of hydrogen evolution. For a lay reader, the takeaway is that by carefully sculpting the microscopic shape of catalyst surfaces and choosing electrolytes that bring the right ions to the right spots, engineers can overcome some of the fundamental slowdowns of alkaline devices and design more efficient, energy‑saving systems for clean hydrogen production.
Citation: Zhang, Q., Sun, P., Li, H. et al. Synergistic cation-facet effects boost alkaline hydrogen evolution kinetics on stepped Pt surfaces. Commun Chem 9, 113 (2026). https://doi.org/10.1038/s42004-026-01924-9
Keywords: hydrogen evolution, alkaline electrolysis, platinum catalyst, interfacial water, alkali metal cations