Clear Sky Science · en
Double Z-scheme biochar-based g-C3N4/Bi2WO6/Ag3PO4 nanocomposite for efficient removal of antibiotics and synergistic mechanisms
Why cleaning antibiotics from water matters
Antibiotics have saved countless lives, but once they leave our bodies they can linger in rivers, lakes, and wastewater. In these environments they help fuel the rise of hard‑to‑treat “superbugs” and spread resistance genes through microbial communities. The paper summarized here reports a new sunlight‑driven material that can quickly break down stubborn antibiotics in water and at the same time kill harmful bacteria, pointing toward more sustainable ways to keep water safe.
A smart sponge for light and pollutants
The researchers designed a complex “photocatalyst” that acts a bit like a solar‑powered sponge. It is built from four main components: a porous charcoal‑like substance called biochar, and three different light‑sensitive solids based on carbon nitride, bismuth tungstate, and silver phosphate. The biochar, made by heating plant waste, provides a honeycomb structure with many tiny pores and a large internal surface area. This structure helps trap antibiotic molecules from water and offers plenty of space to anchor the other three ingredients as nanometer‑sized particles. Together they form a tightly connected composite, so incoming light can be efficiently converted into reactive charges that move across the whole network rather than dying out where they are created. 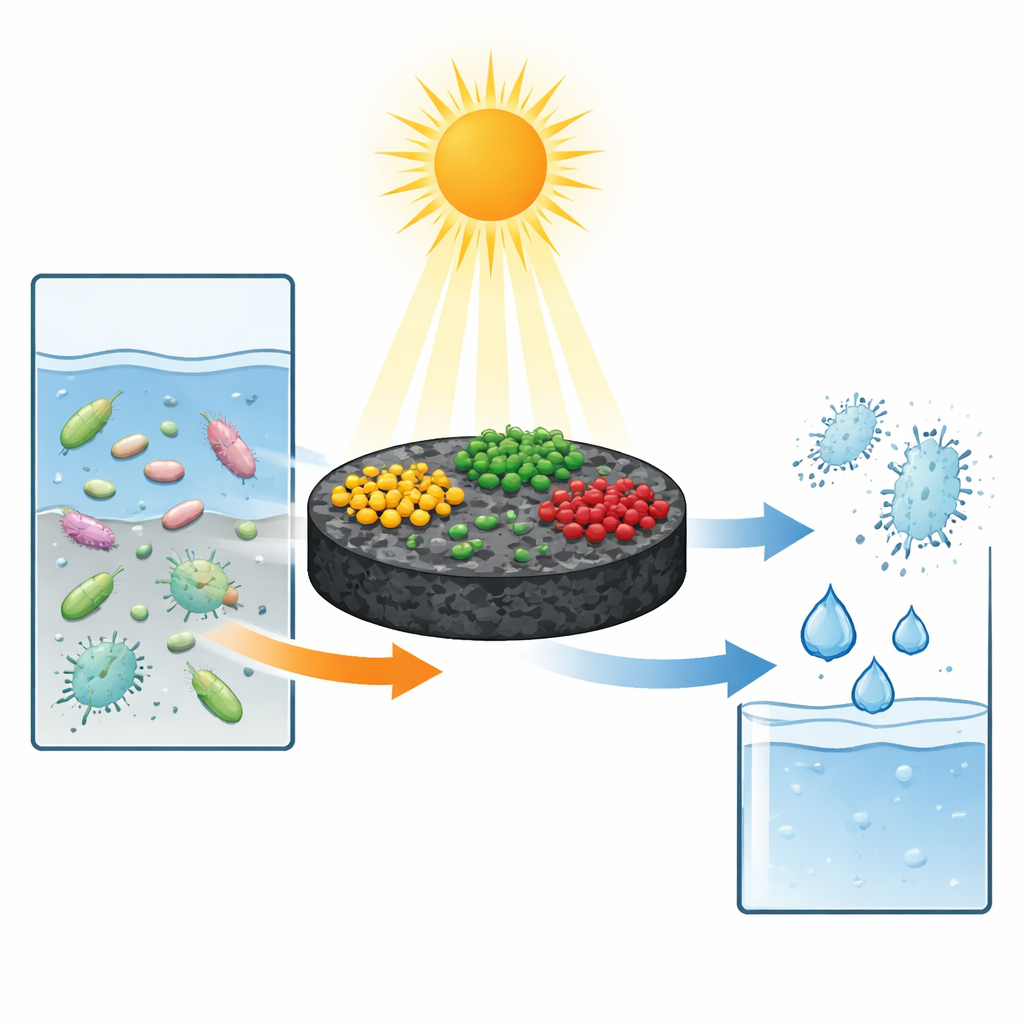
Using sunlight to tear antibiotics apart
When this composite is illuminated, it absorbs a broad range of light from ultraviolet to visible wavelengths. The energy separates electrical charges inside the material into mobile electrons and “holes” that behave like positive charges. In many photocatalysts these charges quickly find each other again and neutralize, wasting the absorbed light. Here, careful tuning of the energy levels of the three light‑absorbing components, assisted by the conductive biochar, creates what the authors call a “double Z‑scheme” pathway. In simple terms, electrons and holes are steered along two intertwined routes so that the most energetic electrons and the strongest oxidizing holes end up on different parts of the composite, greatly reducing recombination. These charges react with water and oxygen to generate highly reactive oxygen forms, including superoxide and hydroxyl radicals, which attack antibiotic molecules such as tetracycline and chop them into smaller pieces and, eventually, carbon dioxide and water.
Performance in the lab and in real wastewater
In tests with water containing a common veterinary antibiotic, tetracycline, the new composite removed almost all of a relatively high starting concentration within two hours of light exposure. Its reaction rate was roughly 9–14 times faster than any of the three light‑sensitive components used alone. Measurements of total organic carbon showed that much of the antibiotic carbon was truly mineralized rather than just converted into slightly altered by‑products. The same material also worked well on two other widely used antibiotics, norfloxacin and chloramphenicol. Importantly, when tried on actual industrial wastewater that already contained a mix of contaminants, the composite still removed more than 85 percent of tetracycline and significant portions of the other drugs, suggesting it can cope with the chemical complexity of real‑world streams.
Killing germs while limiting metal leakage
Beyond breaking down drug molecules, the material also served as a disinfectant. Under light, it eliminated about 99 percent of both Escherichia coli and Staphylococcus aureus within 48 hours. This germ‑killing effect appears to come from a combination of the same reactive oxygen forms used to degrade antibiotics and a small amount of silver ions released from the silver phosphate component. Tests over repeated cycles showed that the composite remained structurally stable and lost only a few percent of its activity, while releasing far less silver than the silver compound alone. Detailed electrical and optical measurements confirmed that the biochar not only helps capture pollutants but also improves charge transport, extends the lifetime of light‑generated charges, and boosts the creation of reactive species. 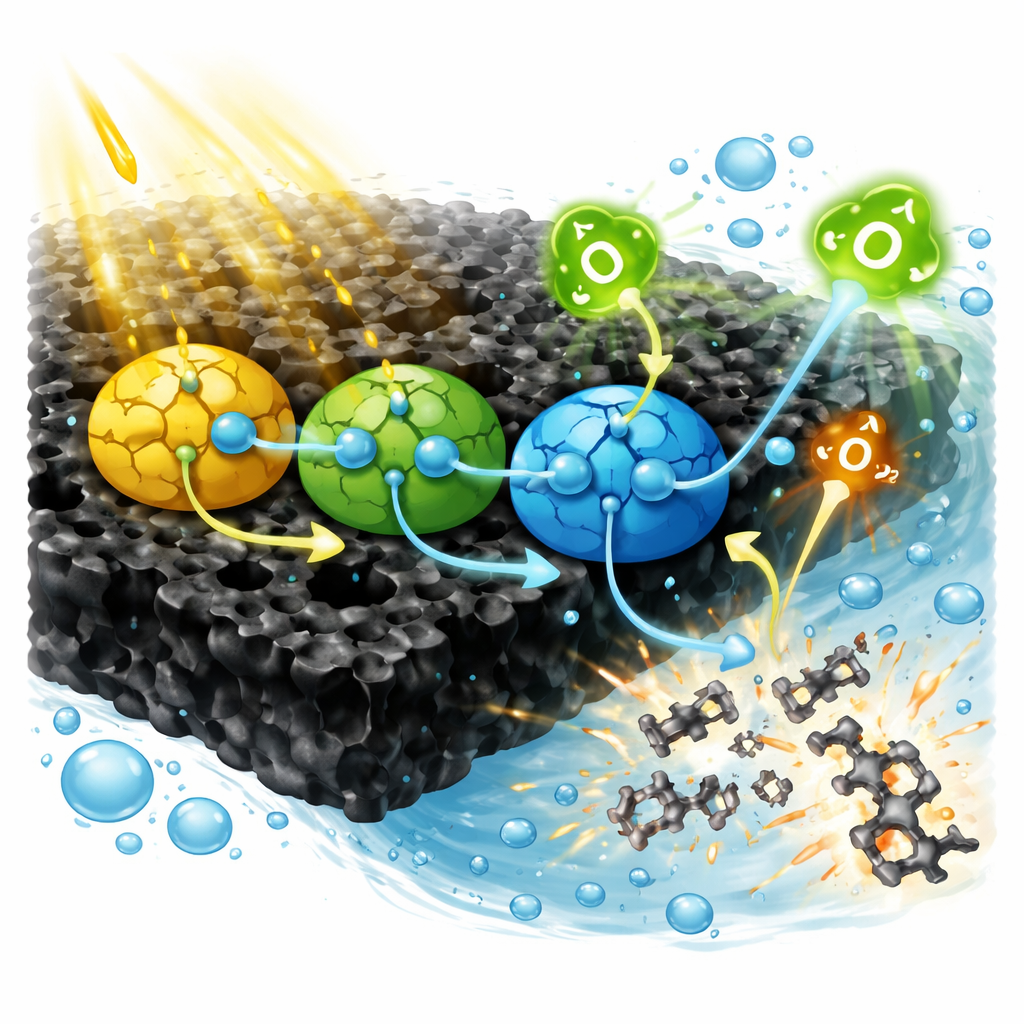
What this means for cleaner water
Put simply, the study shows that thoughtfully combining waste‑derived biochar with several complementary light‑active materials can yield a powerful, reusable water treatment tool. Under simulated sunlight, this composite can both dismantle stubborn antibiotics and kill bacteria, even in complex wastewaters, while limiting the release of heavy metals. The work offers a blueprint for next‑generation photocatalysts that use solar energy and low‑cost carbon materials to tackle emerging pollutants and disinfection in a single, integrated step.
Citation: Wang, T., Zhang, D., Shi, H. et al. Double Z-scheme biochar-based g-C3N4/Bi2WO6/Ag3PO4 nanocomposite for efficient removal of antibiotics and synergistic mechanisms. Commun Chem 9, 105 (2026). https://doi.org/10.1038/s42004-026-01923-w
Keywords: photocatalytic water treatment, antibiotic pollution, biochar composites, solar-driven disinfection, advanced oxidation processes