Clear Sky Science · en
Azobenzene-bridged ionizable amphiphilic Janus glycosides for light-controlled, single-component and organ-modulable pDNA delivery
Shining Light on Smarter Gene Delivery
Gene therapies promise to treat diseases at their roots by delivering bits of genetic code directly into our cells. But steering this fragile cargo safely through the bloodstream, into the right organs, and inside target cells remains a major hurdle. This study introduces a new class of tiny, sugar-based particles whose behavior can be switched with light before they ever enter the body, allowing researchers to redirect where a therapeutic gene ends up and how strongly it is expressed, all while keeping cells largely unharmed. 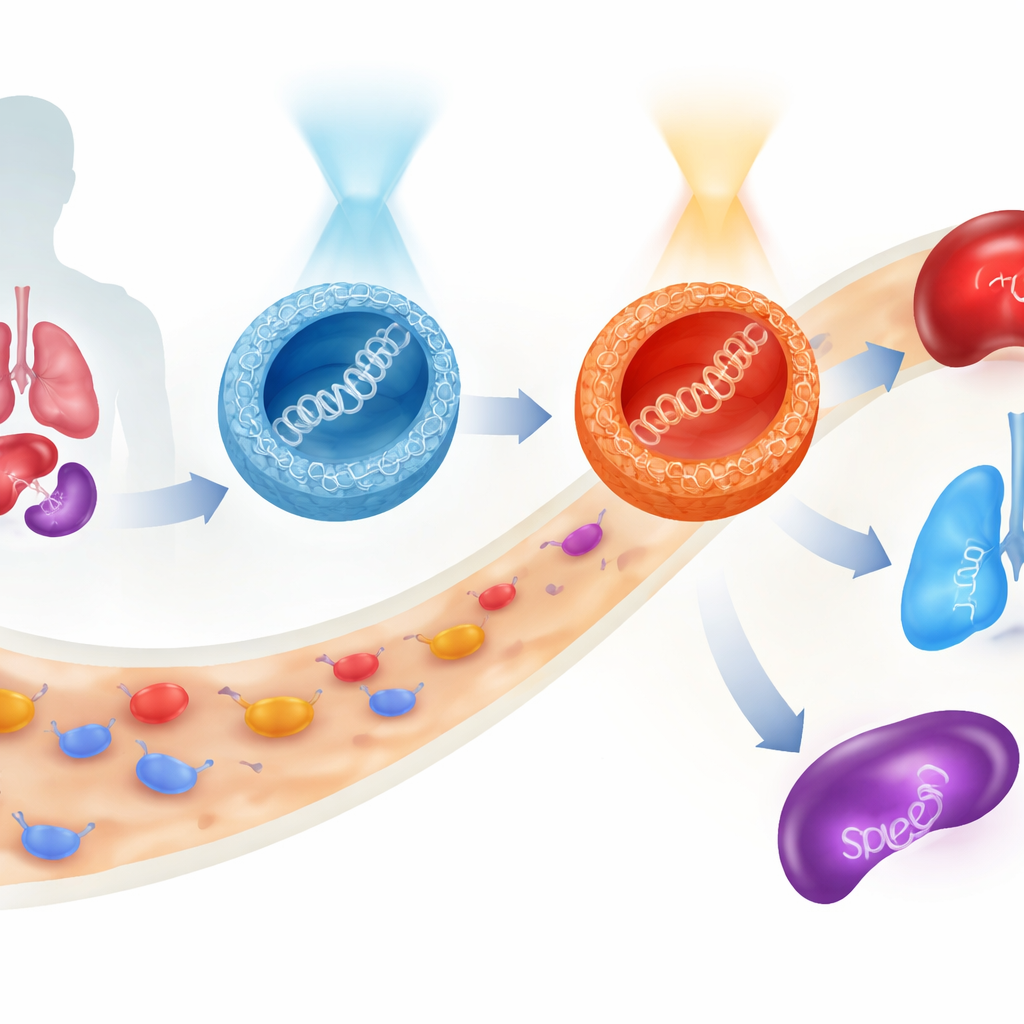
A Need for Precise Genetic Couriers
Today’s most successful gene delivery vehicles are lipid nanoparticles, which helped make mRNA COVID-19 vaccines possible. These multi-component droplets wrap and protect nucleic acids, but they tend to accumulate mainly in the liver and release only a small fraction of their cargo inside cells. Tweaking their compositions can change which organs they favor, yet the mixtures are complex and hard to control precisely. The team behind this work instead set out to design single, well-defined molecules that assemble into nanoparticles with predictable shapes and behaviors—and that can be actively tuned from the outside by a simple light trigger.
A Molecular See-Saw Controlled by Light
At the heart of the new system are twin “Janus” glycosides: molecules built from two glucose units, equipped with water-loving charged heads and fat-loving tails, and joined through a central azobenzene “hinge.” Azobenzene can flip between two mirror-like shapes, called E and Z, when exposed to different colors of light. In water with DNA present, many copies of these molecules spontaneously gather into soft, layered particles that wrap around strands of plasmid DNA. The researchers prepared two versions—one with oxygen bridges and one with sulfur bridges between the sugar and the hinge—to subtly tune stability and biological behavior. By pre-irradiating solutions, they could enrich either the E or Z form and study how each state shapes the resulting gene-delivery particles.
How Shape-Shifting Particles Behave
Measurements of particle size, surface charge, and internal structure showed that light-induced switching between E and Z states has clear physical consequences. Generally, the Z form produced smaller, more uniformly shaped nanoparticles with a more ordered, layered interior than the E form. Microscopy revealed that these internal arrangements resemble alternating sheets of lipid-like material and DNA. Importantly, both states protected DNA from enzymatic degradation. In cultured cells from kidney, liver, and immune tissue, the different isomer states produced strikingly different levels of gene expression, even though the underlying DNA was identical. Repeated light pulses applied after particles had entered cells boosted gene expression several-fold, suggesting that the mechanical jolt from switching helps particles loosen and release their cargo, and may also help them escape from intracellular compartments. 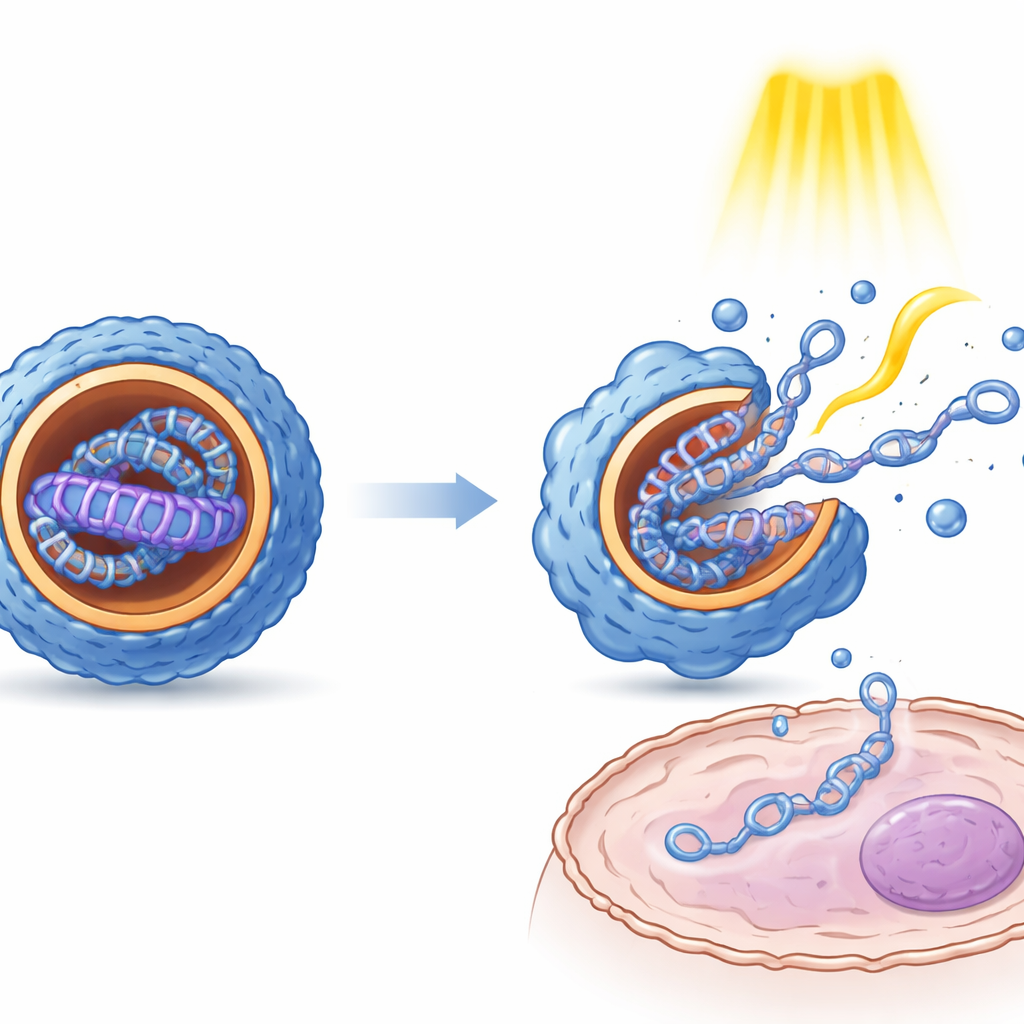
Guiding Genes to Specific Organs
The most surprising behavior appeared in mice. When the oxygen-bridged particles were given in their E-rich form, expression of a marker gene occurred mostly in the liver with some in the lungs. Pre-switching them to the Z form before injection not only increased total expression, but also shifted the balance toward the lungs. The sulfur-bridged particles told a different story: their Z form favored the spleen, a key immune organ. Thus, with the same basic molecular framework—and without changing the DNA cargo—the researchers could direct gene activity predominantly to liver, lung, or spleen simply by choosing the linkage (oxygen or sulfur) and the light-activated shape (E or Z) of the carrier. Throughout these tests, cell viability remained high, suggesting that the system is relatively gentle.
A Light-Tunable Route to Future Gene Medicines
In everyday terms, the team has built a tiny, light-switchable shuttle for DNA. Before injection, a brief light treatment “programs” the shuttles into one of two shapes. Each shape assembles into nanoparticles with characteristic size and surface feel, which in turn determines how strongly they stick to different cell types and where they end up in the body. Because the same chemical scaffold can be modified in many small ways, and because the switching is reversible and does not alter the DNA itself, this platform offers a flexible way to fine-tune where and how gene therapies act. While much work remains before clinical use, the study shows that carefully designed, single-molecule carriers controlled by light could become powerful tools for safer, more precise genetic medicines.
Citation: Wang, Z., Rivero-Barbarroja, G., Benito, J.M. et al. Azobenzene-bridged ionizable amphiphilic Janus glycosides for light-controlled, single-component and organ-modulable pDNA delivery. Commun Chem 9, 118 (2026). https://doi.org/10.1038/s42004-026-01920-z
Keywords: gene delivery, light responsive nanoparticles, azobenzene switches, targeted DNA therapy, nanomedicine