Clear Sky Science · en
Potential role of stabilized criegee intermediates in the reactivity of isocyanic acid
Hidden Danger in the Air We Breathe
Isocyanic acid is an invisible air pollutant released by wildfires, vehicle exhaust, and indoor cooking fires. Even at tiny concentrations, it has been linked to heart disease and cataracts. Scientists have long struggled to explain how this stubborn chemical is removed from the atmosphere, because it barely reacts with the usual air-cleaning agents. This study suggests that an overlooked group of short‑lived molecules, called stabilized Criegee intermediates, may play a surprisingly important role in scrubbing isocyanic acid from the air we breathe.
Why This Pollutant Is So Hard to Remove
Isocyanic acid is produced whenever nitrogen-containing materials burn, from forests and crop residues to diesel fuel and tobacco. Outdoor levels can spike near wildfires and agricultural burns, and indoor levels can soar in homes that rely on open fires for cooking or heating. Yet once released, this chemical tends to linger. It barely reacts with the main atmospheric oxidants such as hydroxyl radicals and ozone, and it does not break apart easily in sunlight. Previous research therefore concluded that isocyanic acid is mainly removed when it sticks to surfaces, dissolves in cloud droplets or raindrops, or deposits on the ground—processes that leave a large fraction of its gas‑phase behavior unexplained.
A New Player in Atmospheric Cleanup
In recent years, atmospheric chemists have become increasingly interested in Criegee intermediates, fleeting molecules formed when ozone attacks double bonds in plant‑derived and pollution‑derived hydrocarbons. Many of these intermediates lose energy in collisions with surrounding air and become “stabilized.” Although their concentrations are modest, they are highly reactive and can strongly influence the fate of sulfur dioxide, organic acids, and even some industrial chemicals. The authors of this study wondered whether these reactive intermediates might also attack isocyanic acid, providing a missing piece in our understanding of how this toxic gas is removed from the atmosphere.
Following Reactions Step by Step on a Computer
Because Criegee intermediates are short‑lived and challenging to study directly, the researchers turned to advanced quantum‑chemical calculations. They focused on the simplest and most common Criegee species, CH2OO, and a close cousin with a methyl group attached, syn‑CH3CHOO. Using a suite of high‑accuracy electronic structure methods, they mapped out how isocyanic acid and these intermediates approach each other, form weakly bound complexes, pass through transition states, and finally generate a variety of products. They also used detailed kinetic modeling to translate these energy landscapes into reaction rates under realistic atmospheric temperatures and pressures, carefully accounting for energy loss in collisions and subtle quantum effects.
How Criegee Intermediates Attack Isocyanic Acid
The calculations reveal that the most important pathway begins when isocyanic acid forms a hydrogen‑bonded complex with CH2OO. In this arrangement, the acidic hydrogen of isocyanic acid interacts with the oxygen end of the Criegee intermediate, while its nitrogen leans toward the carbon center of CH2OO. From this starting point, a concerted dance occurs: the hydrogen shifts from nitrogen to oxygen at the same time as a new bond forms between the carbon skeletons. Crucially, the energy barrier for this reaction lies below that of the separated reactants, meaning the process can proceed very readily once the molecules meet. Competing routes, such as more distant additions or simple hydrogen abstraction at other positions, are much less favorable. When the methyl‑substituted Criegee is considered, the same basic picture holds, but its extra bulk and altered bonding make it slightly less reactive overall.
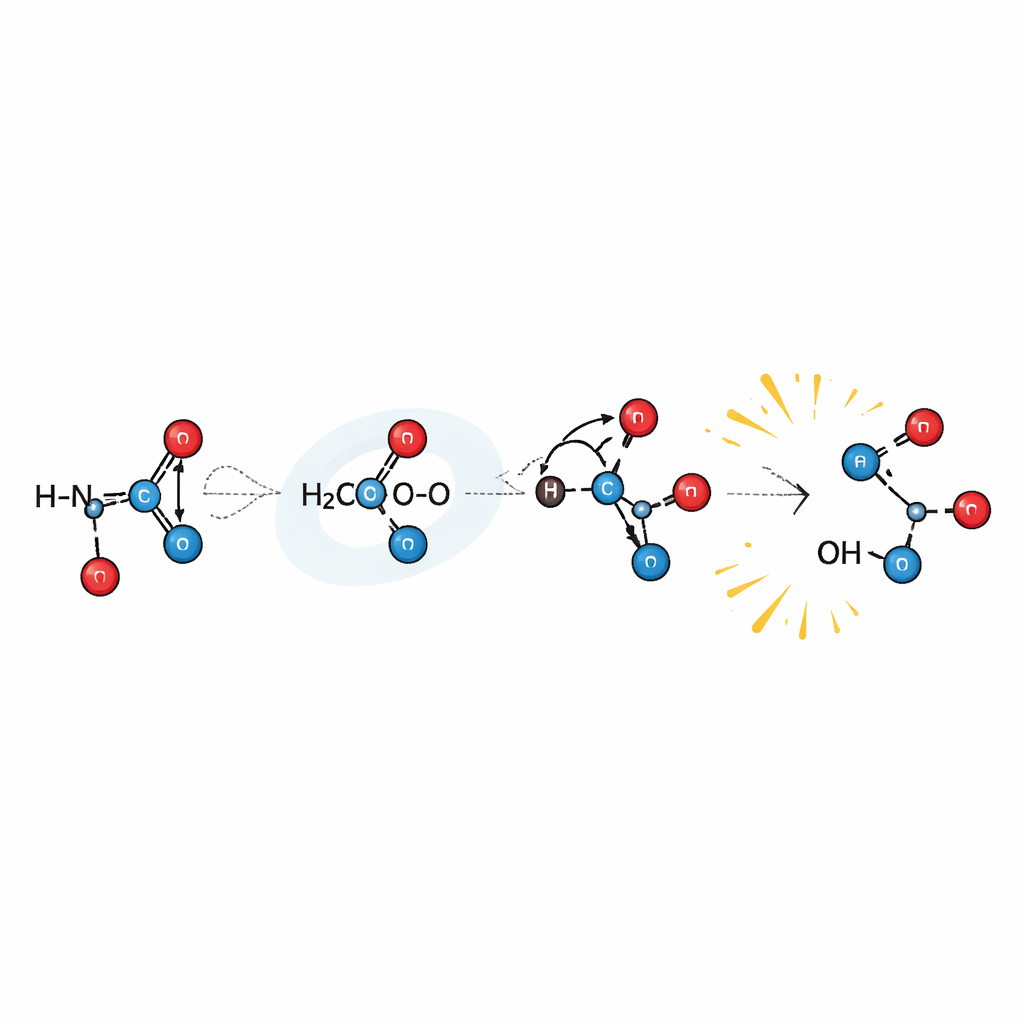
What This Means for the Lifetime of Isocyanic Acid
The modeled reaction between isocyanic acid and the simple Criegee CH2OO is astonishingly fast—roughly a thousand times quicker than the previously considered reaction with hydroxyl radicals under typical tropospheric conditions. The calculated rate also decreases as temperature rises, a behavior consistent with a reaction that proceeds through a pre‑formed, stabilized complex. One major product can further break its weak oxygen–oxygen bond to release highly reactive hydroxyl radicals and an organic fragment, hinting that these reactions may feed back into broader atmospheric oxidation cycles. However, the overall impact of this pathway depends strongly on how many stabilized Criegee intermediates are actually present in a given region and how large a fraction of them are of the CH2OO type. Current field estimates suggest that, while this chemistry may shorten the lifetime of isocyanic acid in some environments, it is unlikely to dominate its atmospheric budget everywhere.
Big Picture: A New but Subtle Atmospheric Sink
This study shows that stabilized Criegee intermediates can react rapidly and efficiently with isocyanic acid, providing a new homogeneous gas‑phase route by which this toxic pollutant can be removed from the atmosphere. Although uncertainties in Criegee concentrations mean that surface uptake and cloud processes may still be the main sinks overall, the newly identified pathway helps close an important gap in our understanding of isocyanic acid’s fate. It also suggests that these elusive intermediates influence not only traditional air pollutants, but also lesser‑known toxic species. Future laboratory measurements will be crucial to confirm the predicted reaction rates and to determine how much this chemistry truly shapes the air quality and health impacts associated with isocyanic acid.
Citation: Zhang, M., Hou, H. & Wang, B. Potential role of stabilized criegee intermediates in the reactivity of isocyanic acid. Commun Chem 9, 110 (2026). https://doi.org/10.1038/s42004-026-01919-6
Keywords: isocyanic acid, atmospheric chemistry, Criegee intermediates, air pollution, reaction kinetics