Clear Sky Science · en
Ultrafast solvent-to-solute proton transfer mediated by intermolecular coherent vibrations
How Molecules Shed Light-Induced Stress
When molecules absorb ultraviolet light, they can store more energy than is good for them. If that energy is not released quickly, it can break chemical bonds and damage materials or even DNA. This study explores how a special kind of molecule, a “photobase,” hands off a tiny hydrogen nucleus—a proton—from the surrounding liquid to protect itself within trillionths of a second. Understanding this dance between a molecule and its liquid surroundings could help scientists design better light-driven sensors, catalysts, and protective coatings.
A Tiny Proton on the Move
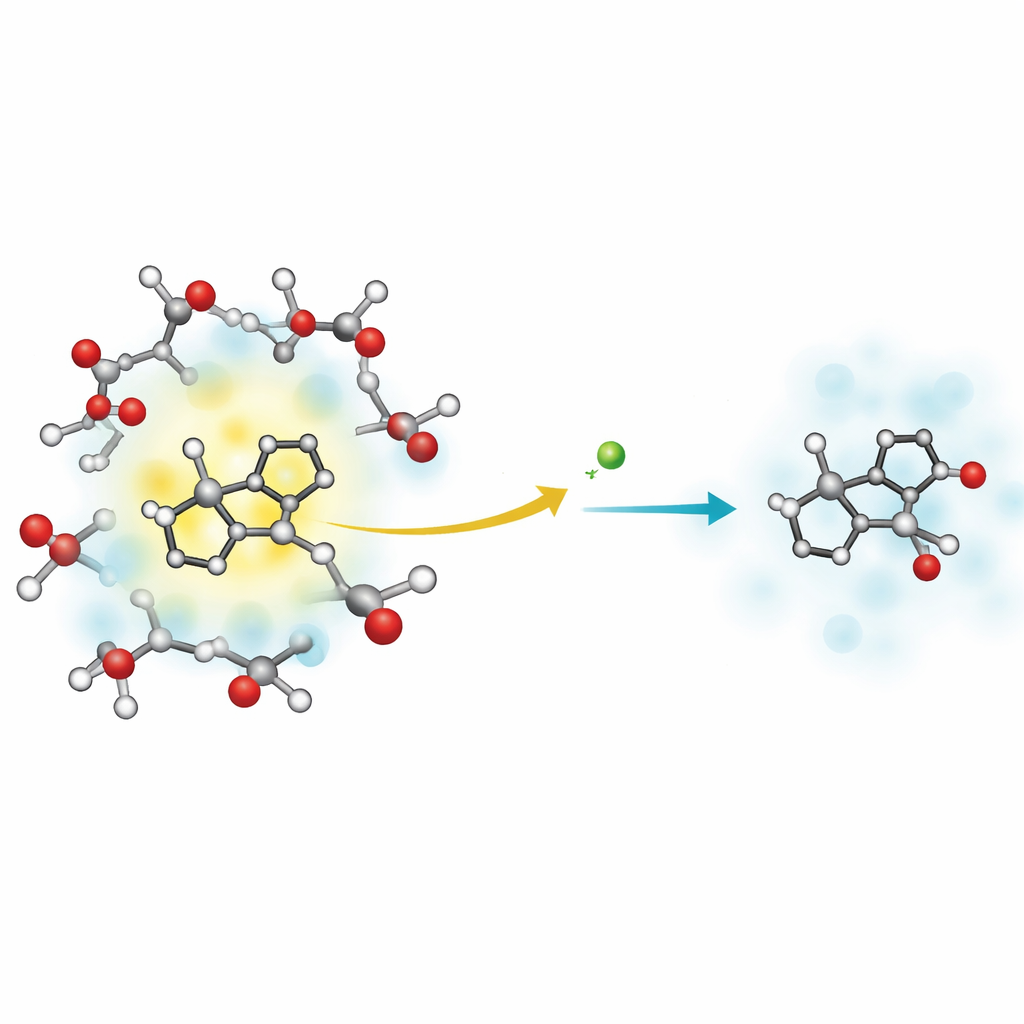
At the heart of the work is a molecule called 2-(2′-pyridyl)benzimidazole, or PBI, dissolved in methanol, a simple alcohol. PBI can grab a proton from the solvent when it is excited by light, acting like a stronger base only in its energized state. The researchers used extremely fast laser pulses to give PBI a jolt of ultraviolet light and then watched, in real time, how its absorption of light changed as it relaxed. These subtle color changes in the measurements reveal when and how protons move and how the surrounding liquid responds.
Three Stages of Energy Release
The measurements show that the energized PBI–methanol system relaxes in three distinct stages. First, within about 2.2 picoseconds (two trillionths of a second), a proton moves from a methanol molecule to a nitrogen site on PBI. This is the key solvent-to-solute proton transfer step, where the environment donates a proton to the excited molecule. Next, over roughly 31 picoseconds, the newly protonated PBI drops back down to its electronic ground state without emitting light, dumping its excess energy into vibrations instead. Finally, over about 186 picoseconds, this vibrational energy gradually leaks into the surrounding methanol, leaving both the molecule and solvent back at thermal equilibrium.
Hidden Vibrations Guiding the Reaction
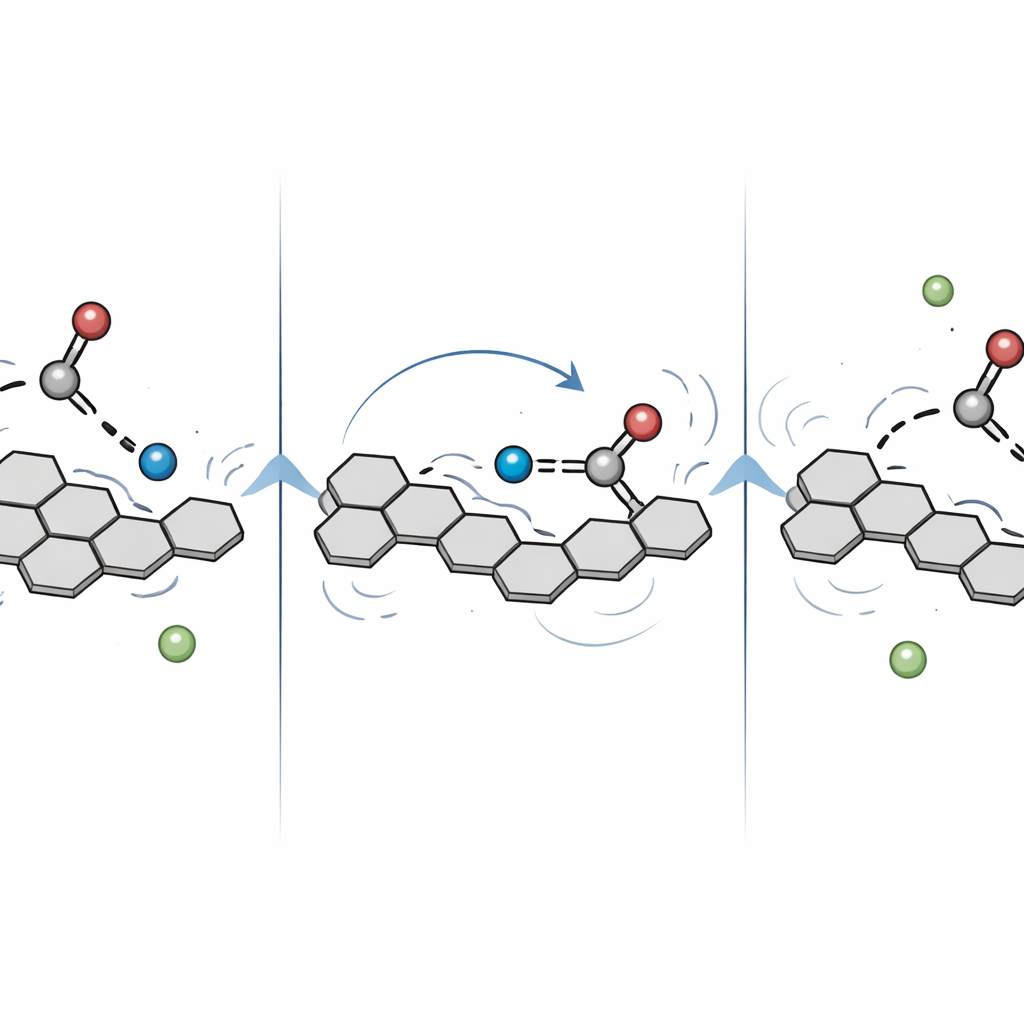
To zoom in on the earliest instants after the light pulse, the team recorded data with much finer time steps on the femtosecond scale (one millionth of a billionth of a second). After removing the overall decay trend, they found a faint but regular pattern of oscillations in the signal—evidence that the atoms in the PBI–methanol pair were vibrating in a coordinated way. Two main vibration periods emerged: about 117 femtoseconds and 340 femtoseconds. Calculations showed that these correspond to low-frequency motions that twist and bend both the PBI framework and the attached methanol molecule, constantly reshaping the hydrogen bond that connects them. These motions modulate the distance and alignment between the proton donor and acceptor, effectively steering the proton along its path. The oscillations faded in less than 300 femtoseconds, implying that the system quickly moves onto a more rugged energy landscape as it climbs toward and crosses the reaction barrier.
Why This Pathway Matters
Computer modeling backed up the experimental picture. Using quantum chemical methods, the authors calculated the energy landscape for several possible reaction routes. The pathway in which the proton moves directly from methanol to PBI was found to have a relatively low barrier and to lead to a more stable product than an alternative route where a hydrogen atom is shuttled differently. The simulated excited-state absorptions for the favored proton-transfer product matched the observed spectra, reinforcing the conclusion that straightforward proton transfer, not a more complex hydrogen-atom shift, dominates under these conditions.
What It Means for Light-Active Materials
Overall, the study shows that proton transfer from solvent to solute in this photobase is not just a simple hop but is intertwined with coordinated vibrations of both the molecule and its liquid partner. These ultrafast motions help set up the right geometry for the proton to move and shape how quickly the system can rid itself of excess energy. For a lay reader, the key takeaway is that chemists are learning to watch and understand how matter protects itself from light one proton and one vibration at a time. Insights like these could guide the design of smarter light-responsive materials—molecules that switch on or off, catalyze reactions, or protect sensitive components—by harnessing, rather than fighting, the restless motion of atoms in liquids.
Citation: Jarupula, R., Mao, Y. & Yong, H. Ultrafast solvent-to-solute proton transfer mediated by intermolecular coherent vibrations. Commun Chem 9, 111 (2026). https://doi.org/10.1038/s42004-026-01917-8
Keywords: ultrafast proton transfer, photobase, vibrational coherence, transient absorption spectroscopy, solute–solvent interactions