Clear Sky Science · en
Steric confinement-induced emission probe for monitoring protein conformations in live cells
Watching Shape-Shifting Proteins in Real Time
Inside every living cell, proteins are constantly changing shape as they carry signals, sense their surroundings, and carry out vital tasks. Many diseases arise when these shape changes go wrong, yet it has been remarkably hard to watch such motions directly in living cells, especially over both very fast and slow timescales. This paper introduces a new light-emitting probe, called BIOSCE, that turns tiny protein motions into visible flashes, allowing researchers to track individual proteins as they bend, twist, and interact in real time inside living cells. 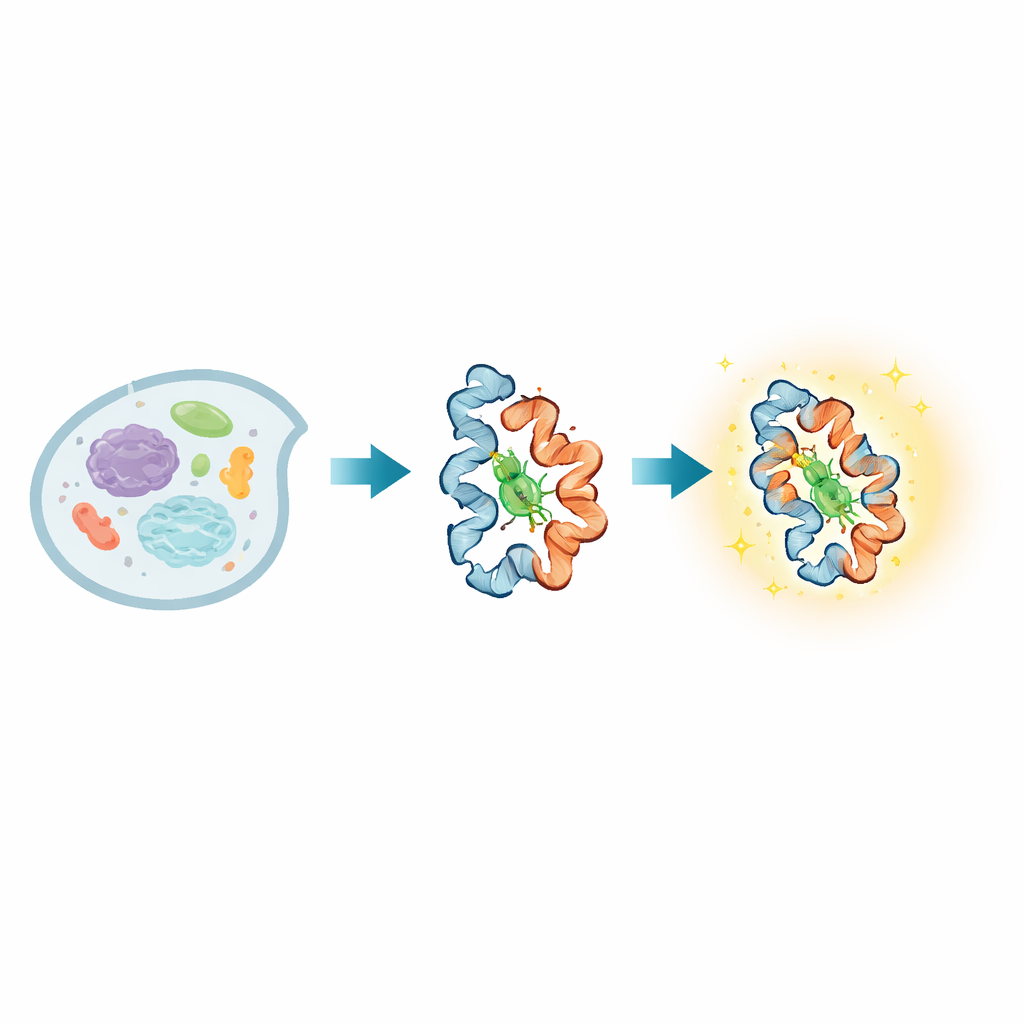
A New Light Switch for Moving Proteins
At the heart of BIOSCE is a small dye molecule, MTPABP-Cl, that becomes brighter when it is squeezed. The researchers attach this dye to a widely used protein tag called HaloTag, which can be fused genetically to almost any protein of interest. When the tagged protein changes its shape or packs more tightly with neighbors, it alters how much “room” the dye has to wiggle. In a loose environment the dye’s internal parts can rotate freely and most of the absorbed light is lost as motion; in a tighter pocket those motions are blocked, and the dye instead releases more of that energy as light. This “steric confinement-induced emission” turns subtle shifts in local crowding around a single protein into a smooth change in brightness, rather than a simple on–off signal, making the probe highly sensitive to small conformational changes.
Building and Testing the Squeeze-Sensitive Dye
The team first designed and synthesized MTPABP-Cl so that it would be dim in solution but brighten when held in place by HaloTag or by a more compact fusion protein. Careful measurements showed that the dye absorbs blue light and emits far-red light, which is favorable for imaging deep inside cells and for long-term observation. When bound only to HaloTag, its light output rose; when HaloTag was fused to a partner protein that folded around the dye, the brightness increased even further. Computer simulations confirmed that, in more compact protein arrangements, the dye experiences stronger crowding, a smaller exposed surface, and more stabilizing contacts, all of which restrict motion and enhance emission. The dye also binds rapidly and specifically to HaloTag, shows very low background in cells that lack the tag, and remains non-toxic at working concentrations, supporting its use in live-cell experiments. 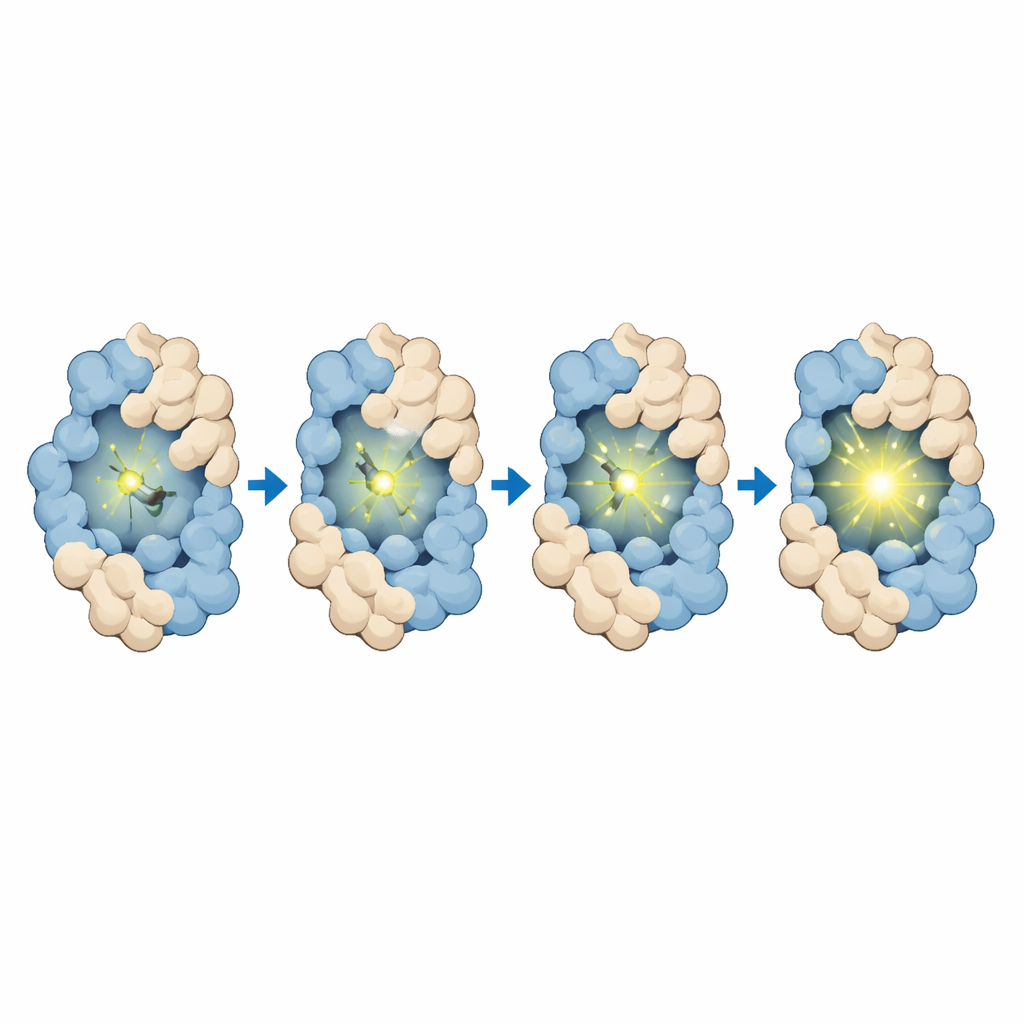
Following Fast Signals and Protein Encounters
To demonstrate what BIOSCE can do, the authors applied it to several well-known cellular processes. First, they adapted a calcium sensor based on calmodulin, a protein that changes shape when it binds calcium ions, which are central messengers in nerve firing and many other cellular events. By fusing calmodulin to HaloTag and labeling it with MTPABP-Cl, they created a chemigenetic indicator called SCECaMP. In human cells and neuron-like cells, this indicator brightened and dimmed in step with calcium spikes triggered by potassium stimulation, with response speeds comparable to the widely used GCaMP fluorescent proteins. Because the BIOSCE signal depends directly on local crowding around the dye, it can faithfully mirror calmodulin’s structural changes while also providing stable, long-lasting fluorescence suitable for extended imaging.
Seeing Drug-Driven Interactions and Toxin Damage
The researchers next asked whether BIOSCE could track when two proteins are brought together by a drug. They used a classic system in which rapamycin pulls together two partners, FKBP and FRB, in a key growth-control pathway. By attaching HaloTag to either FKBP or FRB and labeling with MTPABP-Cl, they observed rapid, millisecond-scale rises in fluorescence as rapamycin induced these proteins to approach and rearrange. The increase in brightness reflected a tighter microenvironment around the dye as the partners came into close proximity. In a more complex test, they studied SNAP25, a nerve-terminal protein crucial for the release of neurotransmitters and the main target of botulinum neurotoxin A. By placing HaloTag on either side of the toxin’s cut site and labeling with the dye, they could separately track the movements of the N-terminal and C-terminal fragments after cleavage. Single-particle tracking revealed that one fragment remained anchored near the cell membrane, while the other diffused more freely through the cytoplasm, and that the exact pattern depended on whether labeling occurred before or after toxin exposure. The probe even reported rapid, zinc-induced conformational tweaks in SNAP25 during this process.
What This Means for Biology and Medicine
Together, these results show that BIOSCE is a versatile new way to visualize how individual proteins move, fold, and interact inside living cells across a wide range of timescales. Because the method relies on a general HaloTag fusion and a single small-molecule dye, it can, in principle, be applied to many different proteins without redesigning the entire sensor each time. The continuous, confinement-dependent brightness change allows researchers to detect subtle structural shifts instead of only large, binary events. Looking ahead, the authors plan to improve probe delivery and imaging depth so that BIOSCE can be used in tissues and whole animals. If successful, this approach could become a powerful tool for linking where proteins are, how they move, and what they do in health and disease, and may ultimately feed into detailed computer models of cellular behavior.
Citation: Jia, H., Yang, L., Yang, Y. et al. Steric confinement-induced emission probe for monitoring protein conformations in live cells. Commun Chem 9, 109 (2026). https://doi.org/10.1038/s42004-026-01914-x
Keywords: protein conformational dynamics, live-cell imaging, fluorescent biosensors, HaloTag probes, botulinum neurotoxin