Clear Sky Science · en
High-pressure synthesis of U2[CO3]3 and U[CO3]2 as potential host phases for uranium in the Earth’s mantle
Hidden Heat Deep Inside Earth
Much of the Earth’s internal heat comes from the slow radioactive decay of elements like uranium. This heat drives plate tectonics, powers volcanoes, and shapes the planet over billions of years. Yet scientists still do not fully know where, and in what form, uranium is stored deep in the mantle. This study explores an unexpected possibility: that certain carbon-rich rocks far below our feet may hide uranium inside special carbon-based minerals, helping to explain how heat is generated and how elements move in Earth’s interior.
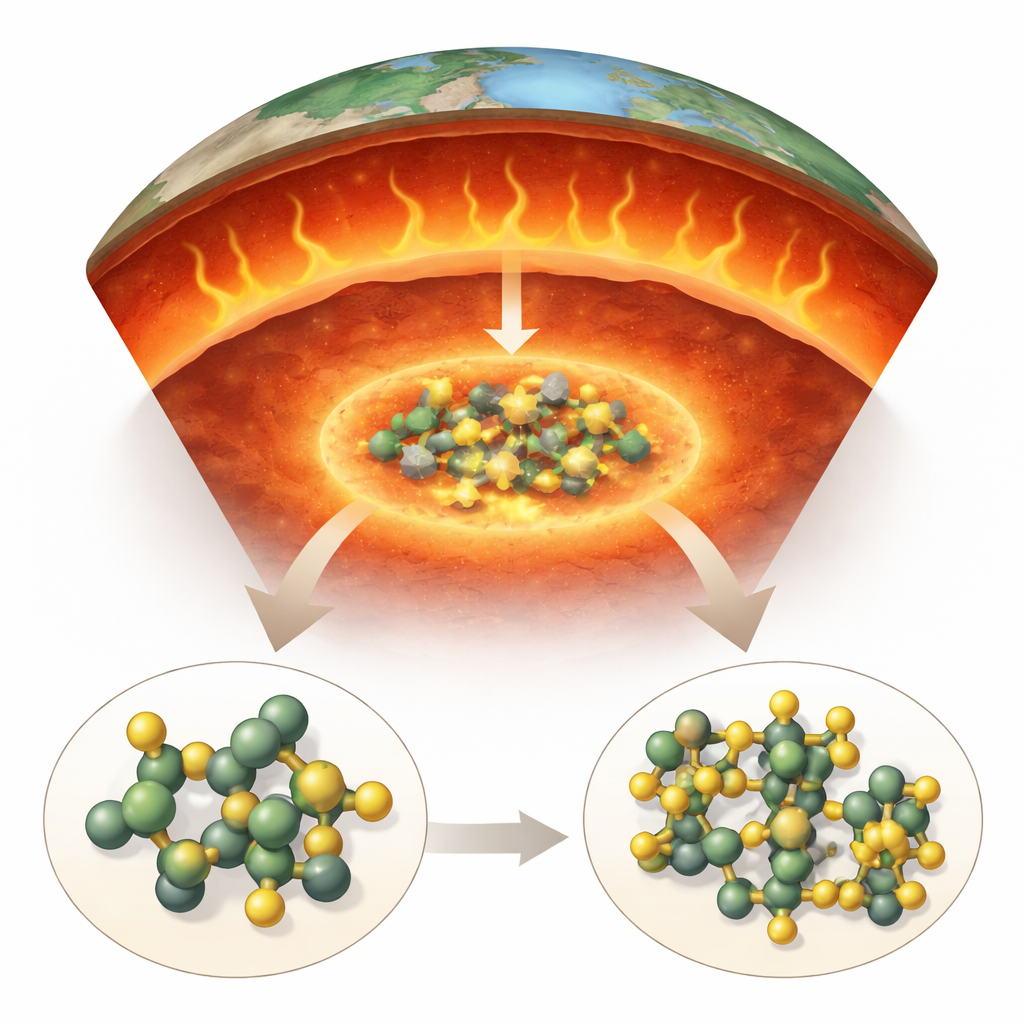
Why Uranium’s Deep Home Matters
Measurements of ghostly particles called geoneutrinos show that uranium supplies a large share of Earth’s internal heat. Near the surface, uranium is found in a variety of minerals, often bound to oxygen in well-known forms such as uraninite and uranyl carbonates. But the mantle—the vast rocky layer between crust and core—is different. The most common mantle minerals do not easily accept much uranium, so there must be other, more unusual hosts. At the same time, we know from diamonds and high-pressure experiments that parts of the deep mantle can be surprisingly rich in carbon. That raises a key question: could carbonate minerals, which are built from carbon and oxygen groups, trap uranium under the immense pressures and high temperatures found hundreds of kilometers down?
Recreating the Deep Earth in the Lab
To test this idea, the researchers recreated conditions like those in the Earth’s transition zone, roughly 600 kilometers below the surface. They used a device called a diamond anvil cell, which squeezes a tiny sample between two diamonds to reach pressures of about 20 gigapascals—more than 200,000 times atmospheric pressure. They placed a small crystal of uranium dioxide, a common uranium oxide, into this miniature pressure chamber and surrounded it with solid carbon dioxide. Then they heated the sample with a laser to around 1,800 kelvin, similar to temperatures expected in that part of the mantle. Throughout and after heating, they probed the sample using Raman spectroscopy, which senses how light interacts with atomic vibrations, and powerful synchrotron X-ray beams capable of revealing the atomic arrangement of any new crystals that formed.
Discovering New Uranium-Bearing Minerals
The experiments showed that uranium dioxide reacted with compressed carbon dioxide to form two entirely new uranium carbonates, both without water in their structure. One compound, called U2[CO3]3, contains uranium in a relatively low charge state (often described as “trivalent”), while the other, U[CO3]2, holds uranium in a slightly higher charge state (“tetravalent”). In both minerals, the carbon and oxygen form flat, triangular groups that are stacked and linked in different ways, with uranium atoms wrapped in irregular cages of oxygen atoms. Using synchrotron X-ray diffraction, the team worked out the detailed three-dimensional arrangement of atoms for each compound. They then used advanced computer calculations based on quantum mechanics to confirm that these arrangements are stable and to examine how compressible the new minerals are under pressure.
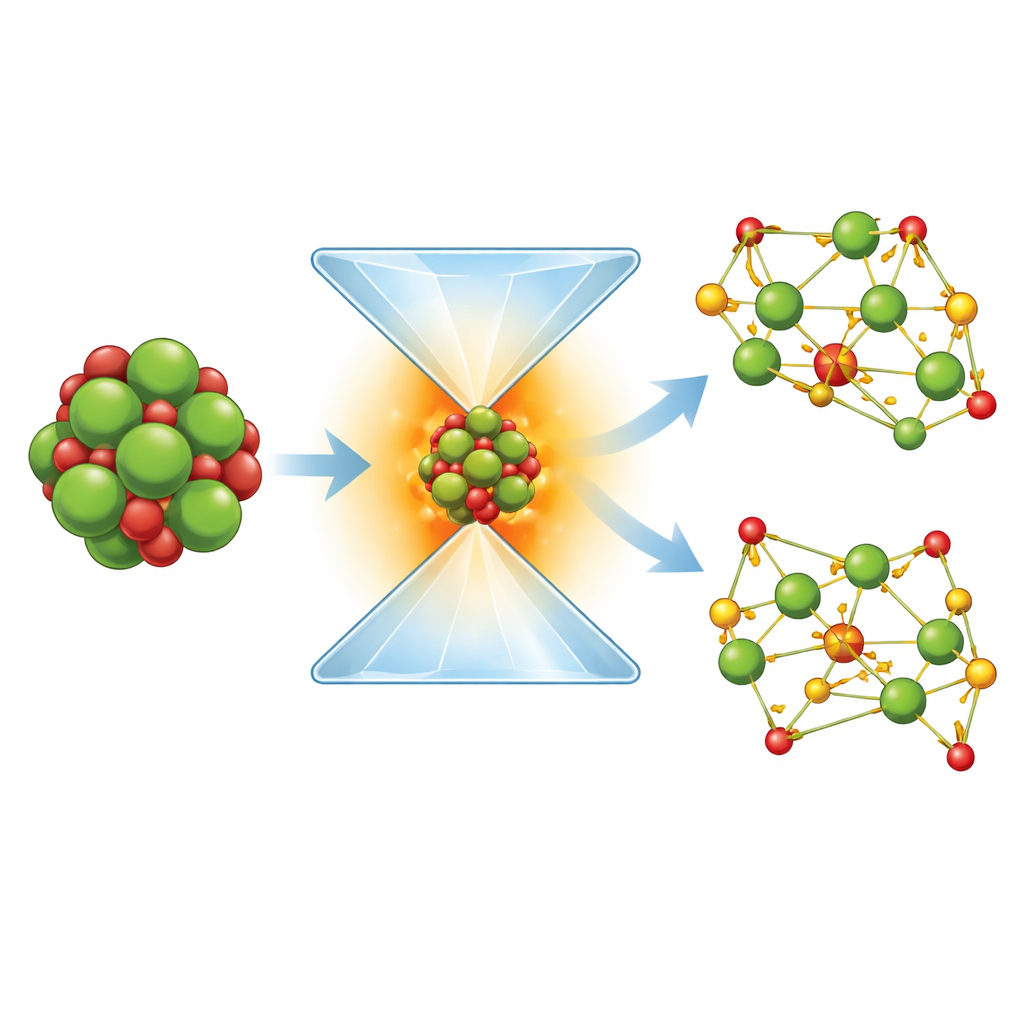
What the Atomic Structures Reveal
The structural data and calculations show that these new uranium carbonates behave much like other high-pressure carbonates that contain more common metals such as calcium or strontium. The distances between uranium and oxygen atoms, and the way the carbonate groups link together, are consistent with strong, stable bonding even at very high pressures. Importantly, the uranium is present in reduced forms compared with its usual highly charged state in near-surface uranyl minerals. This matches the more oxygen-poor, “reducing” conditions expected deeper in the mantle. The mechanical properties—the way the crystals compress—also fall in the same range as familiar mantle-relevant carbonates, suggesting that these phases could survive under realistic deep-Earth conditions.
What This Means for Earth’s Interior
By synthesizing and characterizing these two new uranium carbonates, the study shows that simple, water-free carbonate minerals can indeed host uranium at the pressures and temperatures of the deep mantle, particularly in regions rich in carbon. This provides a plausible answer to where some of Earth’s uranium might reside as tectonic plates carry surface rocks downward. If uranyl carbonates formed near the surface are dragged deep into the mantle, they may transform into reduced uranium carbonates like those discovered here, helping to store radioactive elements and their heat far below our feet. Future work on how stable these minerals are alongside other mantle rocks will further clarify how uranium is partitioned in Earth’s interior and how it contributes to the long-term thermal engine of our planet.
Citation: Spahr, D., Bayarjargal, L., Bykova, E. et al. High-pressure synthesis of U2[CO3]3 and U[CO3]2 as potential host phases for uranium in the Earth’s mantle. Commun Chem 9, 112 (2026). https://doi.org/10.1038/s42004-026-01911-0
Keywords: uranium carbonates, Earth mantle, high-pressure minerals, deep carbon cycle, radiogenic heat