Clear Sky Science · en
Direct diazotization of indoles with 2-Methoxyethyl nitrite
Turning a Problem Gas into Useful Chemistry
Nitric oxide is often viewed as a troublesome industrial gas: it is toxic, hard to handle, and usually treated as waste. Yet it is also rich in nitrogen, a key element in many medicines. This study shows how chemists can tame nitric oxide by converting it into a stable liquid reagent that, in turn, helps build complex molecules related to drug candidates. The work offers a way to turn a hazardous by-product into a tool for making valuable compounds more safely and cleanly.
A New Helper Molecule for Tricky Reactions
Chemists frequently rely on “diazo” compounds—molecules that carry a very reactive pair of nitrogen atoms—to build intricate carbon frameworks. These frameworks appear in many pharmaceuticals, especially those based on indoles, a ring system common in natural products and drugs. Traditional ways to make diazo compounds often use explosive azides or large amounts of strong acids and bases, creating safety and environmental concerns. The authors focus on a safer alternative: a liquid called 2-methoxyethyl nitrite, or MOE-ONO, which can be made directly from nitric oxide gas, oxygen, and a simple alcohol, generating only water as a by-product.
Directly Reworking an Important Drug-Like Ring
The team set out to attach diazo groups directly onto indoles, modifying them at a precise position on the ring without harsh conditions. They discovered that combining MOE-ONO with a common organic radical, TEMPO, and a small amount of a metal salt catalyst based on scandium triggers this transformation efficiently. Compared with classic mixtures of sodium nitrite and acid, or with other liquid nitric oxide donors like tert-butyl nitrite, the new combination gives higher yields, far fewer unwanted by-products, and works in much shorter times. Even indoles that had resisted older methods—such as those bearing bulky phenyl groups—could be converted cleanly using this approach.
One Method, Many Building Blocks
After optimizing the reaction, the researchers tested how broadly it could be applied. They found that a wide variety of indole derivatives tolerated the conditions, including those carrying esters, ketones, amides, and different substituents around the ring, from electron-rich alkyl and methoxy groups to halogens and cyano groups. The method also extended beyond indoles to related naphthol compounds, forming diazo derivatives that had previously required longer, multi-step procedures. Notably, the reaction still worked when run in water, even though the starting materials do not dissolve well in it. This so‑called “on‑water” behavior suggests that simply stirring the mixture in water provides a greener medium that helps the reaction proceed while reducing reliance on organic solvents.
From Reactive Intermediates to Drug Candidates
To show why these diazoindoles are valuable, the authors transformed them further into more elaborate molecules. Using a rhodium catalyst, they converted the diazo group into a highly reactive intermediate called a carbene, which then formed new bonds to build cyclopropane rings at specific positions on the indole. In another sequence, they assembled a candidate modulator for a receptor involved in blood sugar control, demonstrating the method’s relevance to medicinal chemistry. They also showed that Grignard reagents—classical carbon‑adding tools—could selectively attach at one position on the diazoindole while leaving the diazo group intact, opening the door to stepwise construction of densely substituted indole and indoline structures.
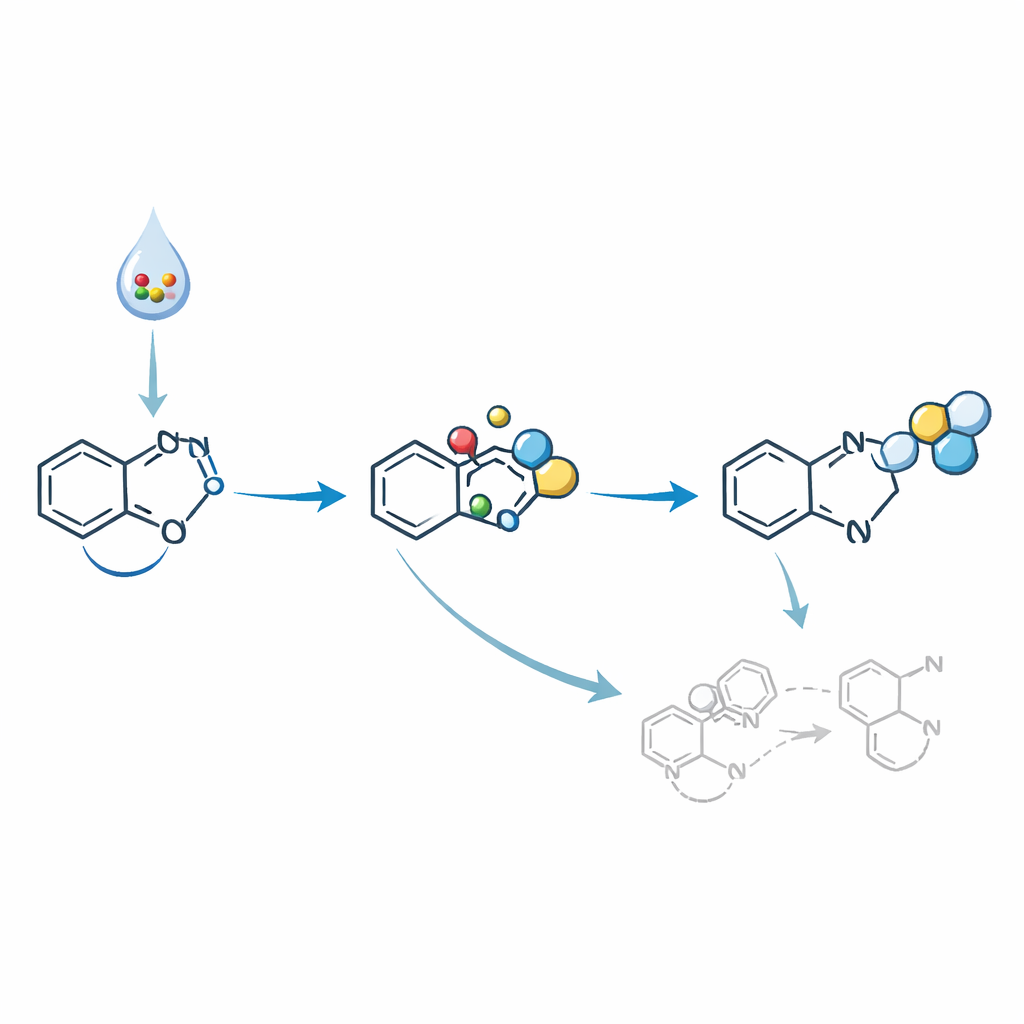
How the Reaction Stays on the Right Track
Behind the scenes, several competing pathways could lead to unwanted nitro or oxime products instead of the desired diazo compounds. Mechanistic experiments suggest that TEMPO helps steer the chemistry by trapping harmful radicals and capturing a fleeting nitroso intermediate before it can rearrange. Additional nitric oxide molecules then add in sequence, ultimately producing the diazo group and releasing harmless nitrate. The scandium salt appears to promote the breakdown of MOE-ONO into reactive species and to activate the indole core, further improving efficiency. Detecting nitrate in the final mixture supports this proposed route from nitric oxide donor to diazo product.
Safe and Sustainable Route to Complex Molecules
Overall, this work introduces a practical way to install highly useful diazo groups on indole rings without relying on explosive reagents or harsh conditions. By using a stable liquid derived from nitric oxide gas, the method both “upcycles” a problematic industrial emission and streamlines access to complex, drug‑like molecules. For non‑specialists, the key message is that clever chemistry can transform a toxic waste gas into a versatile building block for future medicines, while reducing environmental impact along the way.
Citation: Hashidoko, A., Kitanosono, T., Nakao, Y. et al. Direct diazotization of indoles with 2-Methoxyethyl nitrite. Commun Chem 9, 104 (2026). https://doi.org/10.1038/s42004-026-01910-1
Keywords: nitric oxide upcycling, diazoindoles, green organic synthesis, indole-based drugs, NO donor reagents