Clear Sky Science · en
Oncogenic PI3Kα variants reveal graded conformational spectrum with mutation-specific cryptic pockets
When One DNA Change Isn’t Enough
Cancer is often described as a disease of mutations, but this study shows that not all mutations act alone. The researchers examined a key growth-control enzyme, PI3Kα, and found that when two specific DNA changes occur together in the same copy of its gene, they can push this protein into more active shapes than either change alone. That matters because overactive PI3Kα not only drives cancers, but is also involved in benign overgrowth syndromes and brain development disorders—and it helps explain why some tumors respond poorly to current drugs.
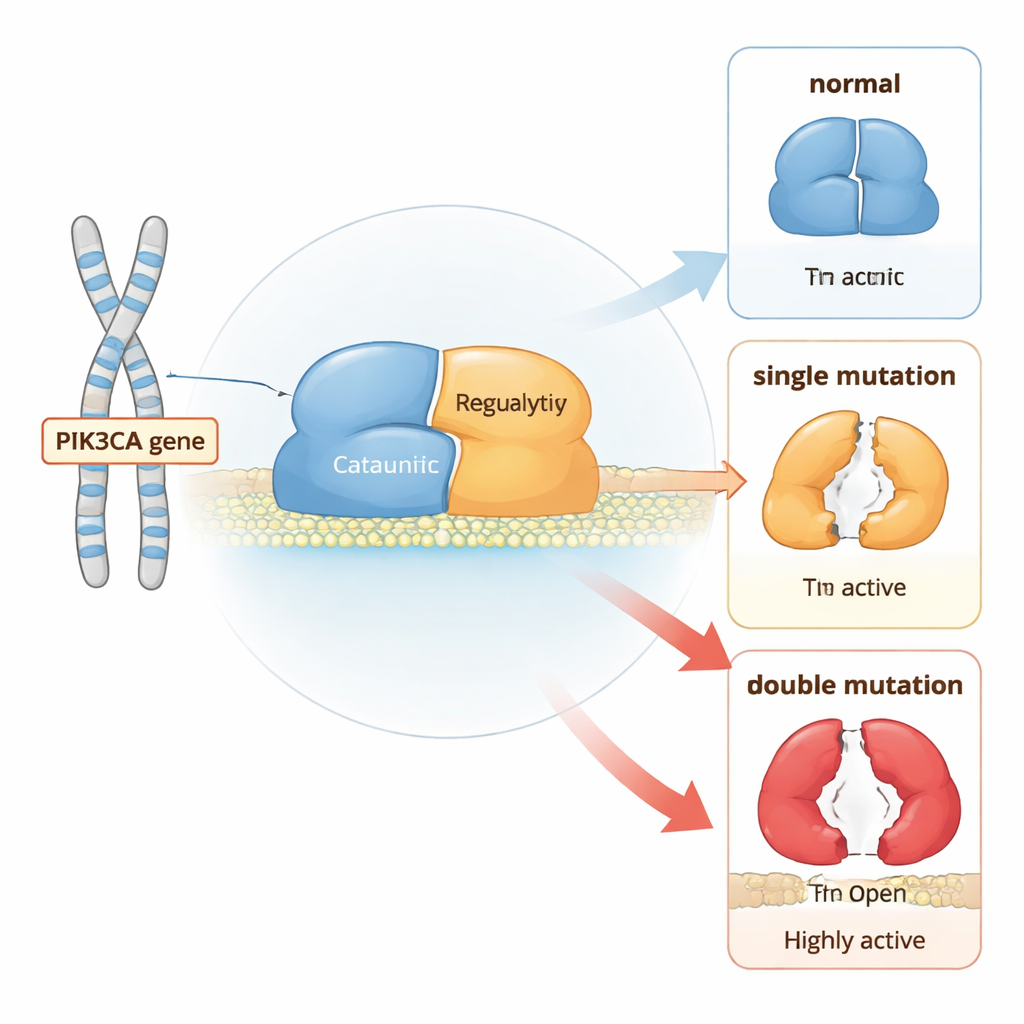
A Molecular Switch for Cell Growth
PI3Kα is a two-part protein that sits near the cell membrane and relays growth signals. Under normal conditions, it stays mostly in an “off” position until receptors on the cell surface are stimulated. Then, PI3Kα moves to the membrane and converts a lipid called PIP2 into PIP3, which tells the cell to grow and divide. To prevent runaway growth, several pieces of the protein act like built-in brakes: one region clamps the catalytic core, another holds a flexible activation loop close, and a partner subunit helps keep the whole complex in check.
How Single and Double Mutations Tilt the Balance
Using long, atom-level computer simulations, the authors compared normal PI3Kα with versions carrying either single “hotspot” cancer mutations or combinations of a hotspot with a weaker mutation in the same molecule. They found that each mutation changes how often the protein visits more open, active shapes, but double mutants push the balance much further than single ones. In particular, some pairs of mutations work together to loosen the contacts that hold a regulatory piece (called nSH2) in place and to shift another segment (iSH2) away from its docking site. This coordinated motion unlatches the catalytic core, making it easier for the protein to move to the membrane and start signaling.
Helping the Substrate Reach the Reaction Site
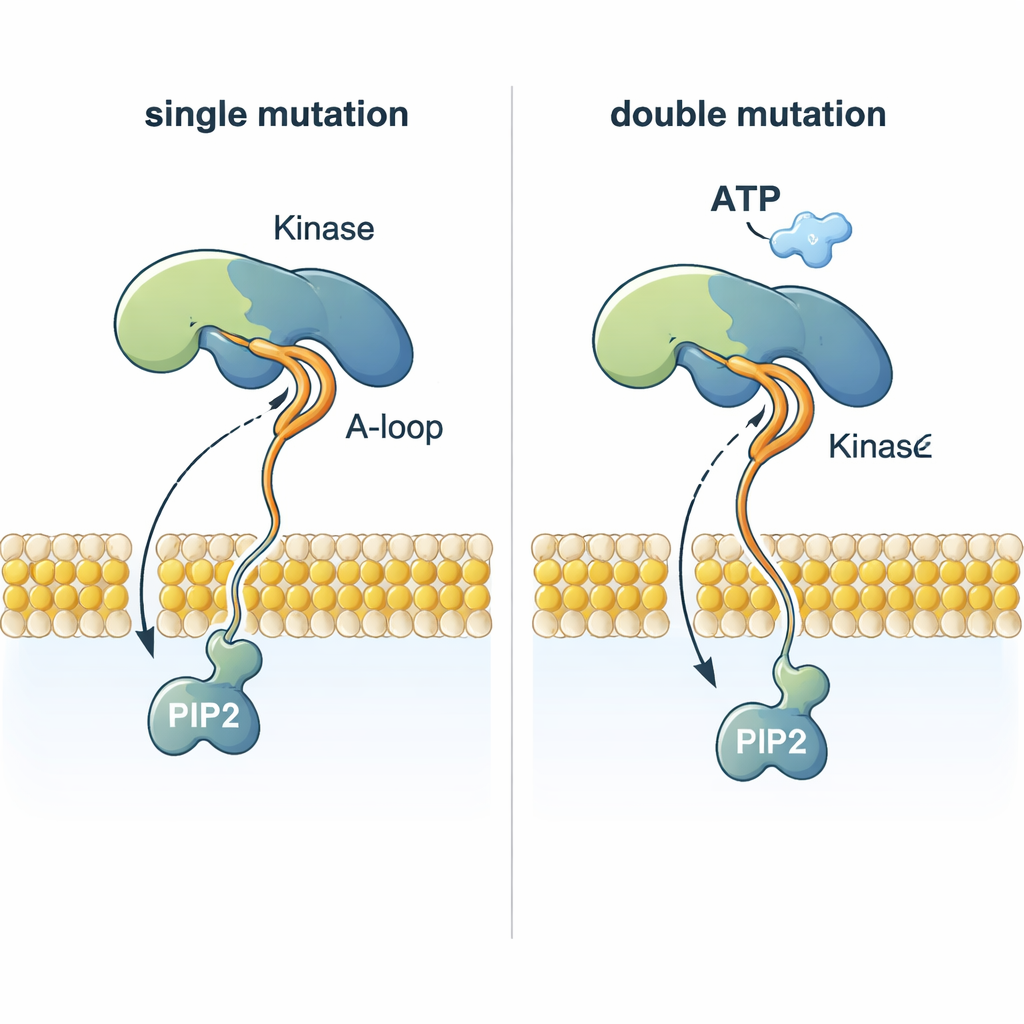
A striking puzzle is how PI3Kα can add a phosphate group from ATP—which sits nearly 2 nanometers above the membrane—to PIP2, which is embedded in the membrane. The simulations show that certain mutations reshape a flexible activation loop so that a highly positively charged stretch juts out toward the membrane. This loop can grab the negatively charged head of PIP2, pull it out of the lipid layer, and guide it up toward ATP. Double mutants that combine a membrane-anchoring mutation with one that changes this loop coordinate PIP2 much more efficiently than normal PI3Kα or single mutants, offering a structural explanation for why tumors with two PI3Kα mutations can be especially aggressive.
Hidden Drug Sites Revealed by Motion
Because PI3Kα is large and flexible, a single drug aimed at its active site often struggles to work across all mutant forms and can cause side effects such as severe changes in blood sugar. By tracking how the protein breathes and flexes over time, the researchers discovered “cryptic” pockets—temporary grooves that open only in certain mutant shapes and are invisible in static crystal structures. These pockets appear in mutation-specific locations, for example near regions that move most strongly in particular double mutants. That suggests that carefully designed allosteric drugs, which bind away from the active site, could be tailored to lock individual mutant variants back into safer, less active conformations.
Toward Smarter Combination Treatments
Overall, the work supports a “one-two punch” view of cancer genetics: a strong hotspot mutation plus a weaker partner in the same PI3Kα molecule can create a graded spectrum of more active shapes, often linked to more severe clinical outcomes. For therapy, the authors argue that instead of relying on a single inhibitor, it may be more effective to combine drugs that target different pockets—one at the active site and one or more allosteric sites that exist only in specific mutants. Such cocktails could, in principle, dial down PI3Kα activity more precisely, reduce toxic side effects, and better match treatment to the exact mutation pattern found in a patient’s tumor.
Citation: Jang, H., Yavuz, B.R., Zhang, M. et al. Oncogenic PI3Kα variants reveal graded conformational spectrum with mutation-specific cryptic pockets. Commun Chem 9, 100 (2026). https://doi.org/10.1038/s42004-026-01906-x
Keywords: PI3K alpha, oncogenic mutations, allosteric inhibitors, molecular dynamics, signal transduction