Clear Sky Science · en
Chirally locked and dynamic bis-perylene diimide macrocycles with multiple sources of chirality
Why twisted ring-shaped molecules matter
Light can do more than just illuminate; it can carry a kind of “handedness,” or twist, that is crucial in technologies ranging from advanced displays to chemical sensors. This paper explores newly designed ring-shaped organic molecules that control that twist with unusual precision. By locking twists into place and using them to influence other, otherwise neutral molecules, the researchers show how to build more reliable materials for devices that respond to or emit circularly polarized light.
Building tiny glowing rings
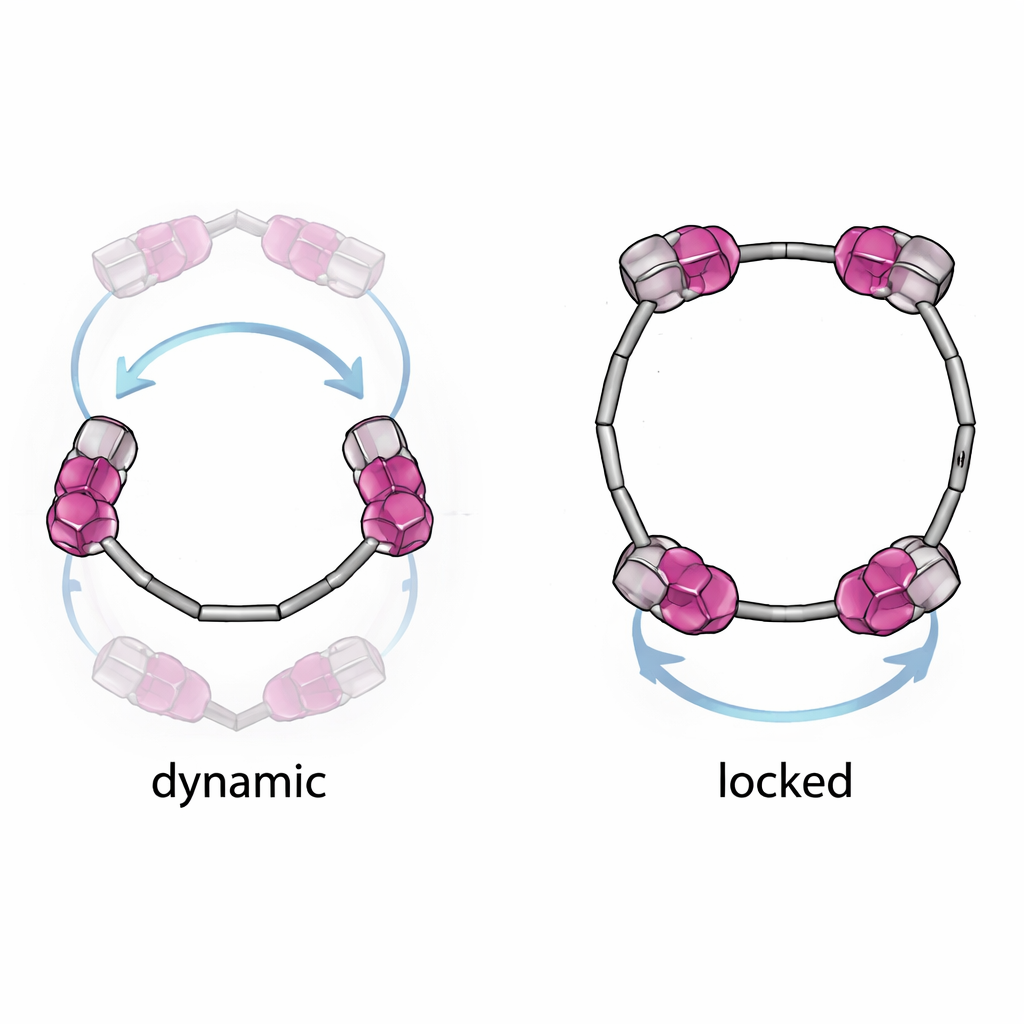
The team focuses on a family of colorful dye molecules called perylene diimides, or PDIs, well known for their stability and bright emission. Two of these PDIs are joined head-to-head to form a molecular ring, known as a macrocycle. Because of how the dyes are attached and subtly twisted, each ring can exist in several mirror-image forms, much like left- and right-handed versions of the same object. The chemists carefully tuned the size and shape of the side arms on the PDIs so they could either allow the dyes to flip between forms (a dynamic ring) or block this motion and “lock in” a specific handedness.
Locking in molecular handedness
Short, compact side arms produced a flexible macrocycle in which the two PDIs could twist and somersault through the ring’s central opening, constantly interconverting between different chiral arrangements. Longer, bulkier side arms, however, were just long enough to jam this motion like a bar across a doorway. This created three distinct, stable forms of the ring: two “homochiral” versions where both PDIs twist the same way, and one “heterochiral” version where they twist in opposite directions. Using techniques such as nuclear magnetic resonance, circular dichroism (which measures how differently a material absorbs left- versus right-circularly polarized light), and X-ray crystallography, the authors confirmed that these locked forms do not easily transform into one another, even when heated.
How twist changes light
Once the rings were in hand, the researchers examined how their different patterns of handedness affected the way they interacted with light. All of the macrocycles absorbed and emitted light in the visible range, like typical PDI dyes. However, the locked homochiral rings showed significantly stronger signatures in both circular dichroism and circularly polarized luminescence, meaning they engage much more strongly with twisted light and can emit it more efficiently. Detailed analysis showed that the dominant contribution to this behavior comes from the intrinsic helical twist of each PDI unit, rather than just from how the two dyes stack together in the ring. In other words, the built-in twist of the building blocks is crucial for amplifying chiral optical effects in the final structure.
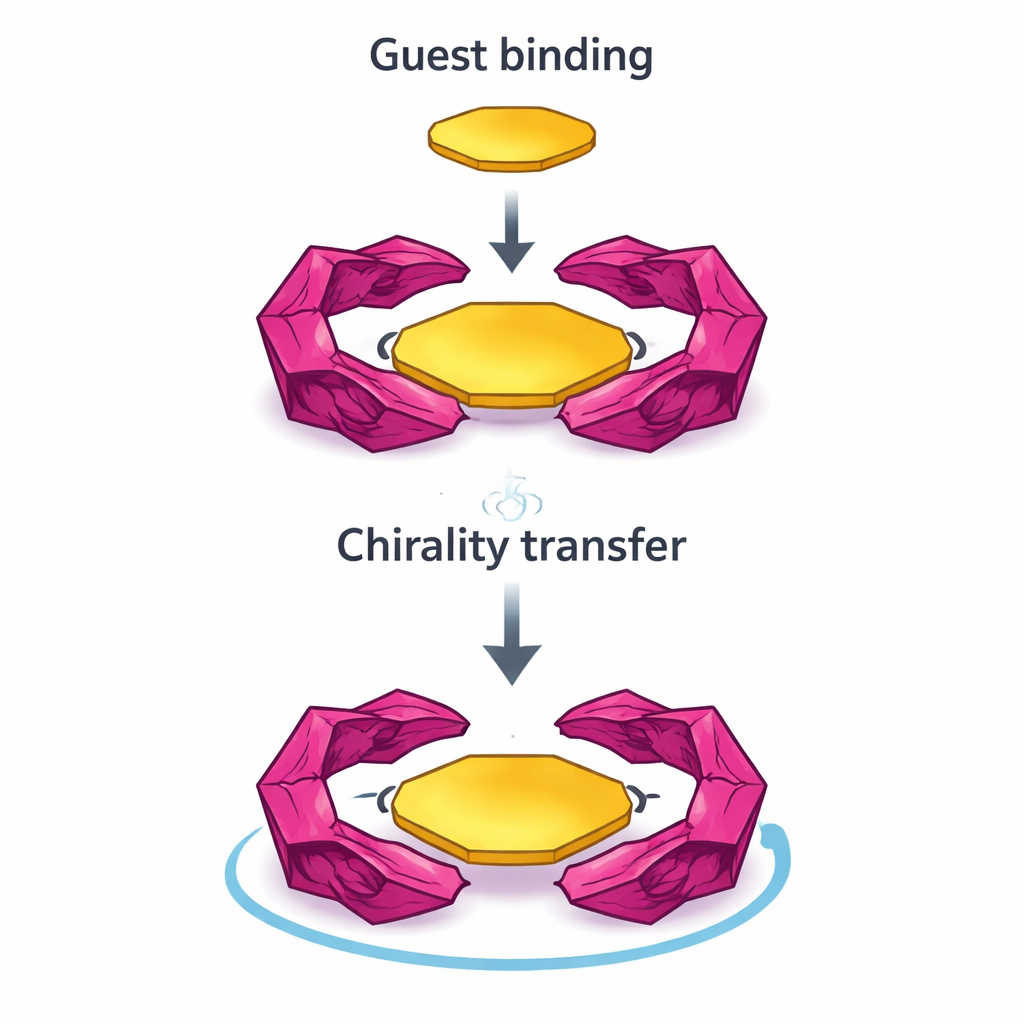
Passing chirality on to a guest
These macrocycles are not only light-responsive; they also act as hosts for flat, disk-like aromatic molecules such as coronene, which by themselves are not chiral. When such a “guest” slides into the cavity of a locked homochiral ring, the combined structure acquires a strong circular dichroism signal at wavelengths where the guest absorbs. This shows that the guest molecule has effectively “borrowed” handedness from its chiral host. The effect is clearest in the locked homochiral rings, which bind guests more strongly and maintain their circularly polarized emission even after binding. In contrast, the heterochiral ring and the flexible ring show weaker binding and a loss or near-cancellation of chiral optical signals once the guest is present, because competing twists cancel each other out.
What this means for future technologies
For non-specialists, the key message is that the authors have learned how to design small, robust rings that not only possess a controlled twist but can also pass that twist on to other molecules without losing it themselves. This level of control over molecular handedness and light emission could feed directly into better circularly polarized LEDs, more sensitive optical sensors, and new spintronic components that use electron spin rather than charge. By showing that the intrinsic twist of the dye units is the dominant factor, and that locking this twist in place enhances both light response and guest binding, the work provides a blueprint for crafting next-generation chiral materials from the bottom up.
Citation: Hartmann, D., Penty, S.E., Pal, R. et al. Chirally locked and dynamic bis-perylene diimide macrocycles with multiple sources of chirality. Commun Chem 9, 102 (2026). https://doi.org/10.1038/s42004-026-01904-z
Keywords: chiral organic materials, perylene diimide macrocycles, circularly polarized luminescence, host–guest chemistry, supramolecular chirality