Clear Sky Science · en
Sequence and length-scale dependent dynamics in biocondensates of highly charged disordered proteins
Why tiny protein droplets matter
Inside our cells, many vital reactions happen inside soft, droplet-like structures that have no surrounding membrane. These “biomolecular condensates” behave like tiny liquid compartments and are often built from floppy, highly charged proteins. Experiments have shown that molecules inside some of these droplets can move surprisingly fast, even though the droplets as a whole can be quite thick and sticky. This paper asks a simple but important question: how does the exact order of positive and negative charges along these proteins control how quickly things move inside such droplets?
Building droplets from floppy charged chains
The authors focus on intrinsically disordered proteins—long, flexible chains that do not fold into rigid shapes. Many of these chains carry both positive and negative charges, arranged in different patterns along their length. Using large-scale computer simulations, the team created model droplets formed by many copies of such proteins, along with water and small ions, to mimic the crowded interior of real condensates. They systematically changed how “blocky” the charge pattern was, from sequences where positive and negative charges alternate to sequences where all positive charges are grouped together and all negative charges form a separate block.
Water content and fluid flow shape motion
The simulations revealed that these droplets can still behave like true liquids with a very high water content—between about 55% and 84%, depending on the charge pattern. That amount of water means that fluid flow, or hydrodynamics, is not completely suppressed as many theories for dense polymer solutions would suggest. By tracking how the protein segments wander over time, the authors found motion patterns characteristic of chains whose movement is aided by the surrounding fluid, rather than slowed down by friction alone. This hydrodynamic influence persists up to and even beyond the size of an entire protein chain, especially in droplets formed from sequences with more mixed charges and therefore more water.
Local charge patterns control micro-scale flexibility
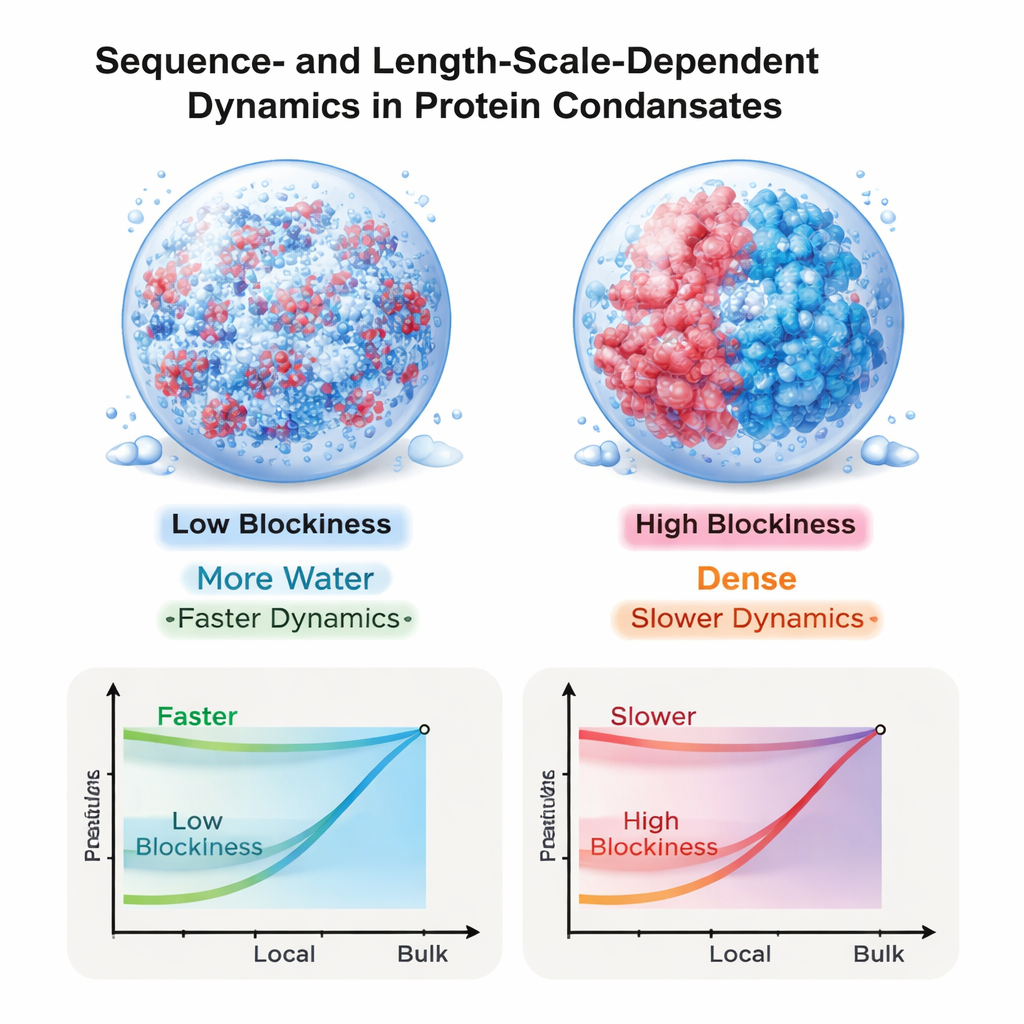
Looking closer, the authors examined how short sections of each chain relax and rearrange inside the droplet. Different portions of the same protein, containing the same number of building blocks, could move at very different rates depending on their local mix of positive and negative charges. Sections that were more balanced in charge relaxed more quickly, while stretches dominated by one charge type moved sluggishly. Surprisingly, these differences remained strong even though simple screening arguments would predict that electrostatic effects should be short-ranged in such salty, crowded conditions. The work shows that the fact the charges are tied together along a chain preserves long-ranged electrical correlations that strongly affect local motion.
Viscosity depends on the size of what you probe
The team then asked how “thick” these droplets feel at different length scales. Using a method that links tiny ripples in the fluid’s momentum to its resistance to flow, they computed a viscosity that depends on the probing length. For all sequences, the fluid appears thinner when probed at very small scales and thicker when probed across the whole droplet. This size dependence is especially dramatic for the blocky-charge sequences: for the most segregated pattern, the bulk viscosity is more than twenty times higher than the local, monomer-level viscosity. In contrast, droplets from well-mixed sequences show a much smaller gap. Overall, the bulk viscosity grows steeply with protein concentration, in line with how dense neutral polymer solutions behave, even though the proteins themselves are highly charged.
Connecting simulations to puzzling experiments
These results help resolve an apparent paradox from recent experiments: how can small molecules and protein segments diffuse so rapidly inside condensates that, measured as a whole, appear extremely viscous? The answer emerging from this work is that both the flow of water and long-ranged electrical interactions remain important in these droplets, and their impact is tightly controlled by how charges are ordered along each chain. Sequences with blocky charge patterns produce dense, highly viscous droplets that nonetheless remain relatively fluid on microscopic scales, while more mixed sequences yield uniformly more mobile environments. By demonstrating that charge sequence can tune motion differently at different length scales, the study offers a design principle for engineering synthetic protein-based materials and sheds light on how cells may program condensate behavior through the detailed sequence of their disordered proteins.
Citation: Zhou, H., Wu, Z., Jiang, L. et al. Sequence and length-scale dependent dynamics in biocondensates of highly charged disordered proteins. Commun Chem 9, 98 (2026). https://doi.org/10.1038/s42004-026-01903-0
Keywords: biomolecular condensates, intrinsically disordered proteins, charge patterning, protein dynamics, viscosity