Clear Sky Science · en
An integrated workflow for the structure elucidation of nanocrystalline powders
Why tiny crystals matter
Many modern medicines and advanced materials are made as fine powders, not nice, shining crystals. That’s a problem for scientists, because our best tool for seeing how atoms are arranged in a solid—X‑ray crystallography—needs large, perfect crystals. This article presents a new step‑by‑step strategy for figuring out the full atomic structure of stubborn nano‑sized powders, the kind often produced by greener, solvent‑saving manufacturing methods. Understanding these structures can help improve how drugs dissolve, how stable they are, and how reliably they work in the body.
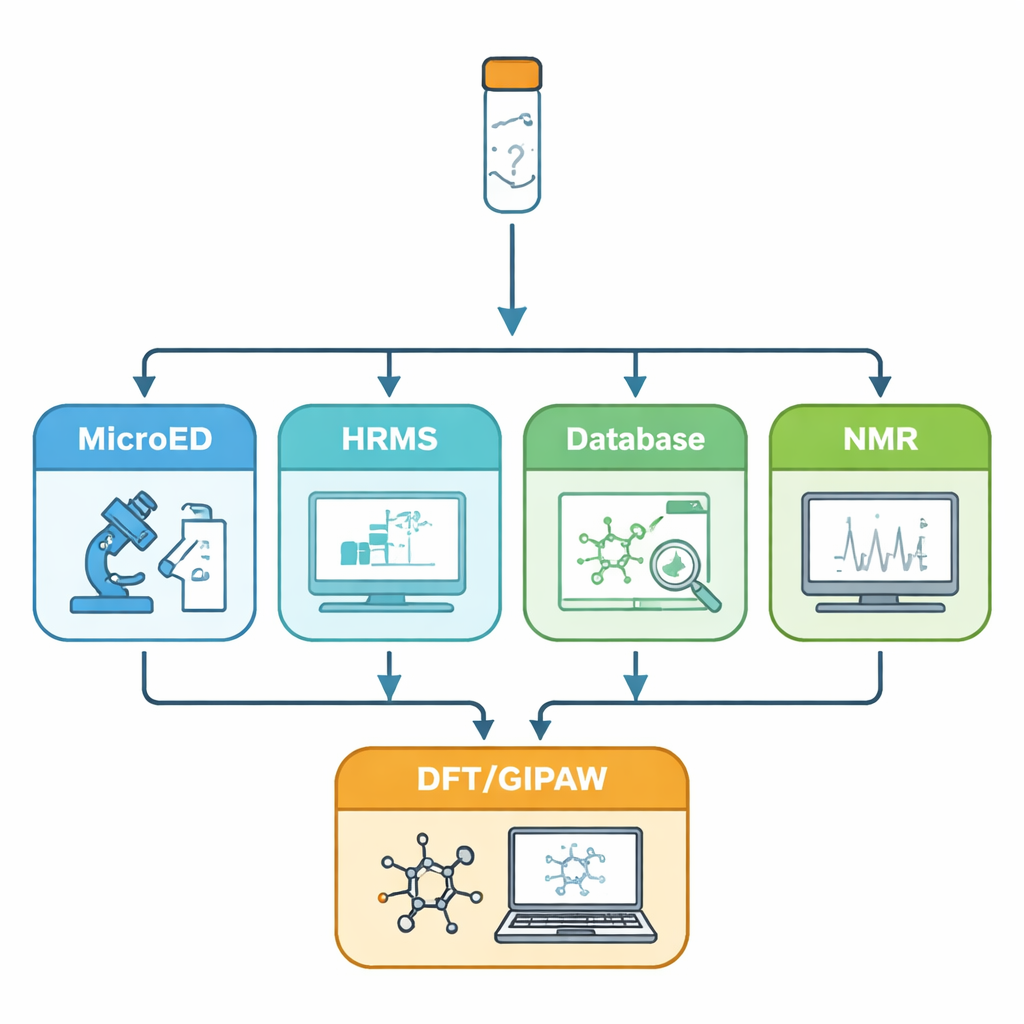
The challenge of invisible atoms
Mechanochemical methods, such as grinding chemicals together in a mill instead of dissolving them in large volumes of solvent, are increasingly popular in line with green chemistry principles. They can create new solid forms of drugs—like salts and cocrystals—that fine‑tune properties such as solubility and shelf life. But the same processes that make these methods attractive also make life hard for crystallographers: they usually yield only nano‑ or micro‑sized crystals, sometimes mixed with amorphous material. Traditional single‑crystal X‑ray diffraction cannot handle such tiny grains, while powder X‑ray diffraction often struggles with the complexity of modern pharmaceutical solids.
A toolkit for seeing the unseen
In recent years, microcrystal electron diffraction (MicroED) has emerged as a powerful way to get atomic‑level information from crystals just a few hundred nanometers across. Electrons interact with matter more strongly than X‑rays, so even these minute crystals can give sharp diffraction patterns. MicroED can reveal the basic layout—or “skeleton”—of molecules in the solid. However, it has two important blind spots: hydrogen atoms are very hard to see, and atoms like carbon, nitrogen, and oxygen can be difficult to tell apart. The authors address these weaknesses by combining MicroED with a series of complementary tools: high‑resolution mass spectrometry to pin down the exact molecular formula, database searches to suggest plausible molecular identities, solution and solid‑state NMR spectroscopy to confirm which molecules are actually present and how they interact, and quantum‑mechanical calculations to refine and validate the final structure.
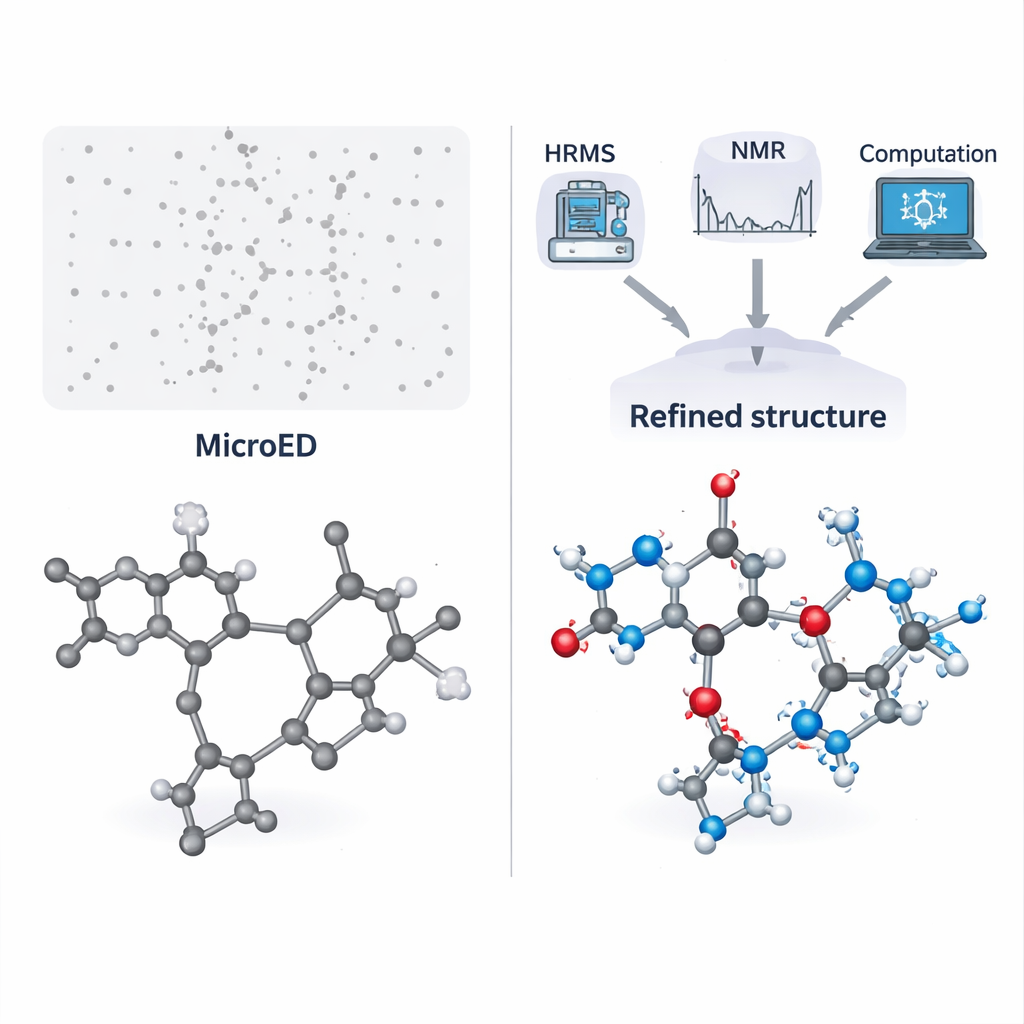
Solving two real‑world puzzles
The team first applies this workflow to a challenging salt made from pyridoxine (a form of vitamin B6) and the antioxidant N‑acetyl‑L‑cysteine. This material can only be made by dry grinding, and repeated attempts to grow suitable single crystals failed because the product is sticky. MicroED showed that the powder contained two different molecular components and even hinted at a sulfur atom, but could not reliably assign all other atoms or any hydrogens. High‑resolution mass spectrometry revealed two precise molecular weights, which were then used to search chemical databases for matching structures. By comparing these candidates with the MicroED skeleton and checking key features with solution NMR, the authors narrowed the possibilities down and confidently identified the two partners as pyridoxine and N‑acetyl‑L‑cysteine.
From rough sketch to finished picture
Once the molecular identities were known, computer‑based quantum‑chemical calculations were used to gently relax the MicroED‑derived structure and predict NMR chemical shifts. These calculated NMR signals were then compared with solid‑state NMR data from the real powder. The excellent match confirmed that the atomic positions, including most hydrogens, were correct. Additional NMR experiments focused on one crucial hydrogen shared between the two molecules, allowing the team to measure its distance to a nitrogen atom very precisely. This showed that the pair forms a true salt, not a neutral co‑crystal—an important distinction for drug behavior and regulatory classification. The same workflow was then tested on a second, quite different compound: a small tripeptide called fMLF, widely used as a model system in NMR studies but whose solid‑state structure had never been reported. Again, the integrated approach successfully delivered a fully validated structure and even shed light on fast motions of its aromatic ring.
How this helps science and medicine
By treating each unknown powder as a “blind test,” the authors show that their modular workflow can start from very limited information and still arrive at a complete, trustworthy atomic structure. MicroED supplies a first sketch, mass spectrometry and databases identify candidate molecules, NMR clarifies how many components are present and where the hydrogens sit, and quantum calculations tie everything together. For chemists and pharmaceutical scientists, this means that complex, nanocrystalline powders produced by green manufacturing routes need not remain structural mysteries. The method opens the door to routine, detailed characterization of powders—from pigments to drug formulations—even when traditional crystal growth fails, helping to design safer, more effective materials from the ground up.
Citation: Sabena, C., Bravetti, F., Miyauchi, N. et al. An integrated workflow for the structure elucidation of nanocrystalline powders. Commun Chem 9, 97 (2026). https://doi.org/10.1038/s42004-026-01902-1
Keywords: MicroED, solid-state NMR, nanocrystalline powders, mechanochemical synthesis, pharmaceutical salts