Clear Sky Science · en
Dynamics of CO photooxidation to CO2 on rutile (110)
Why Watching Molecules in a Trillionth of a Second Matters
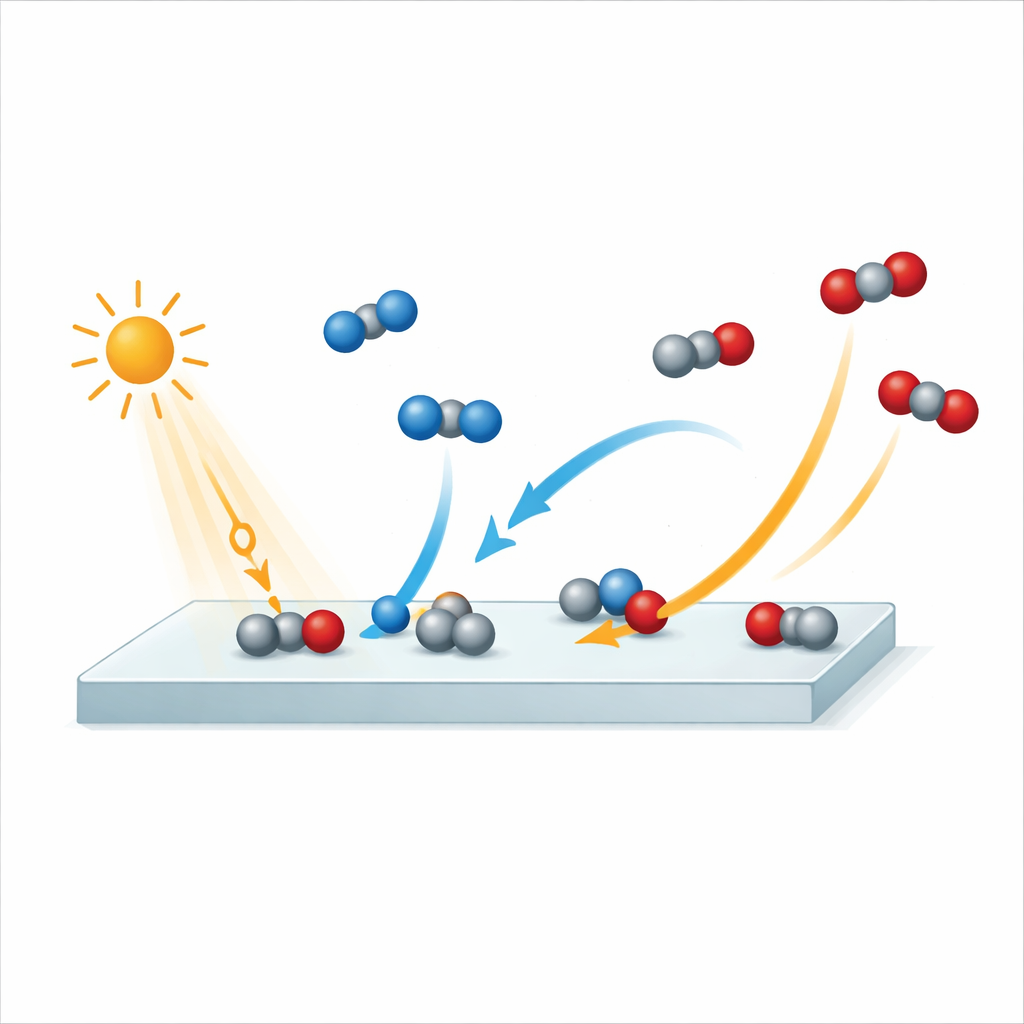
Modern life depends on chemical reactions that clean our air and water and turn sunlight into usable energy. Many of these reactions happen on the surfaces of solid materials called photocatalysts, which are powered by light. This study peers into one of those reactions—how poisonous carbon monoxide (CO) is turned into carbon dioxide (CO2) on a titanium dioxide surface—on timescales of mere quadrillionths of a second. Understanding these ultrafast steps can help scientists design smarter materials for pollution control, self-cleaning surfaces, and solar energy technologies.
A Light-Driven Clean-Up Crew
Titanium dioxide is a workhorse material found in self-cleaning windows, air-purifying coatings, and experimental solar fuels devices. It is cheap, stable, and can use light to trigger powerful cleaning reactions on its surface. But not all forms of titanium dioxide behave the same. Two common forms, called anatase and rutile, differ in how efficiently they drive reactions such as turning CO into CO2. While anatase is generally more active overall, the detailed timing of the individual reaction steps—especially how oxygen from the air is activated—has remained unclear. This study focuses on the rutile form with a specific surface orientation, known as (110), to see exactly how quickly CO is oxidized once light hits the material.
Filming a Reaction at Extreme Speed
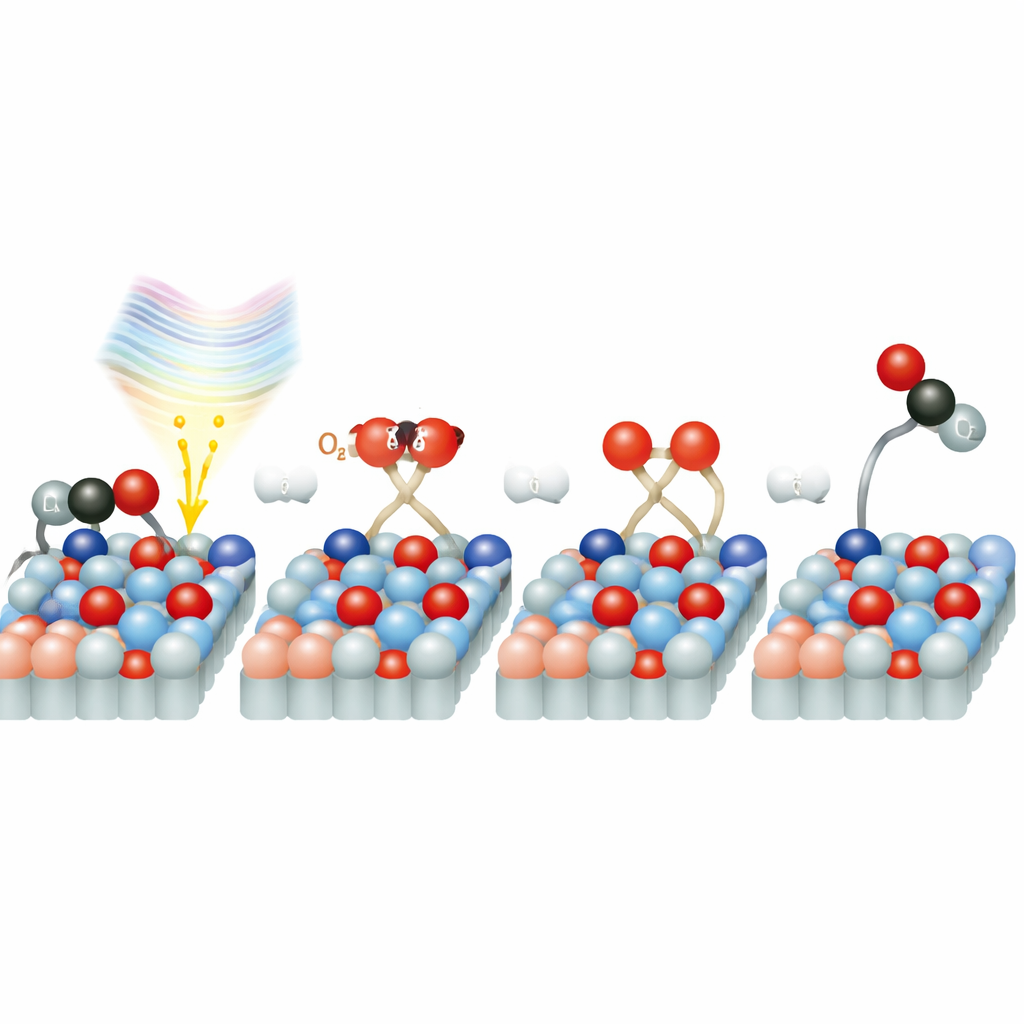
To capture this process, the researchers used a free-electron laser facility in Hamburg that produces extremely short flashes of X-rays. They first struck the rutile surface with a pulse of infrared light, playing the role of a tiny camera flash that starts the reaction. A carefully timed X-ray pulse then probed how the atoms and electrons on the surface changed. By repeating this sequence at different delays between the light and X-ray pulses, they built up a kind of “molecular movie” of the reaction with a time resolution of about 250 femtoseconds—one femtosecond is a millionth of a billionth of a second. The team monitored signals from oxygen atoms in the surface and in the gas molecules, allowing them to distinguish between CO, CO2, water, and the oxide itself as the reaction unfolded.
Catching CO2 in Less Than a Trillionth of a Second
The key observation is that new CO2 appears on the rutile surface within as little as 200 to 800 femtoseconds after the light pulse. That means that oxygen from nearby O2 molecules is activated, bonds are broken, and CO is converted to CO2 in less than one trillionth of a second. After about 0.8 picoseconds, the fresh CO2 signal fades as the product desorbs from the surface. Strikingly, this whole sequence happens faster on rutile than on anatase, where earlier work found the first CO2 appearing only after about 1.2 to 2.8 picoseconds under otherwise similar conditions. Even though anatase produces more CO2 overall and is considered the stronger photocatalyst, rutile carries out the crucial oxidation step more quickly.
A Shortcut Pathway for Oxygen
Why is the reaction so fast on rutile? Detailed computer simulations suggest that oxygen molecules sitting on the surface create special electronic states that act as a bridge between the solid and the O2. When the 770-nanometer laser light is absorbed, it can directly move an electron from the titanium dioxide into the oxygen molecule, forming a charged, activated oxygen species. This “shortcut” avoids the slower route where electrons first travel through the bulk of the solid and then drift to the surface. Once activated, the oxygen rapidly splits and one oxygen atom combines with CO to form CO2. The study also finds that traces of water on the surface can both block reaction sites and, at low levels, potentially help with this charge transfer and oxygen activation, adding another layer of complexity.
Connecting Electronic Structure to Real-World Performance
Overall, the work shows that tiny details in how electrons move between a surface and nearby molecules can dramatically change reaction speeds, even between two closely related materials. While anatase still wins in terms of total photocatalytic output, rutile shows a faster oxygen activation step linked to this special charge-transfer route. By tying together ultrafast measurements and advanced simulations, the study provides a roadmap for tailoring surfaces so that light-triggered reactions occur more efficiently. In the long run, such insights could help engineers design better coatings for air purification, more effective antibacterial surfaces, and improved materials for solar-driven fuel production and water splitting.
Citation: Gleissner, H., Wagstaffe, M., Wenthaus, L. et al. Dynamics of CO photooxidation to CO2 on rutile (110). Commun Chem 9, 127 (2026). https://doi.org/10.1038/s42004-026-01901-2
Keywords: photocatalysis, titanium dioxide, ultrafast spectroscopy, surface chemistry, carbon monoxide oxidation