Clear Sky Science · en
Development of a quadruple-conjugated carbon dot nanomodel for targeted glioma therapy
New hope for stubborn brain tumors
High-grade brain tumors like glioblastoma are among the toughest cancers to treat. Many drugs that work elsewhere in the body either cannot cross the brain’s protective blood–brain barrier or damage healthy tissue along the way. This study describes a laboratory-built “smart” nanoparticle that aims to slip into brain tumors more precisely, carry a powerful drug cocktail, and unleash it directly inside tumor cell nuclei, where it can do the most harm to cancer while sparing normal cells.
A tiny delivery truck built from carbon
At the heart of the new approach are carbon dots—nanometer-scale particles made from carbon that are cheap to produce, mix well with water, and glow under certain types of light. Because their surfaces are rich in chemical hooks, they can be decorated with multiple biological components at once. In this work, the researchers created a “quadruple-conjugated” nanomodel: each carbon dot was chemically linked, in a single one-pot reaction, to two different targeting peptides and two anticancer payloads. This modular design turns each dot into a tiny delivery truck that can recognize tumor cells, pass through cellular barriers, and carry drugs to the right intracellular address.
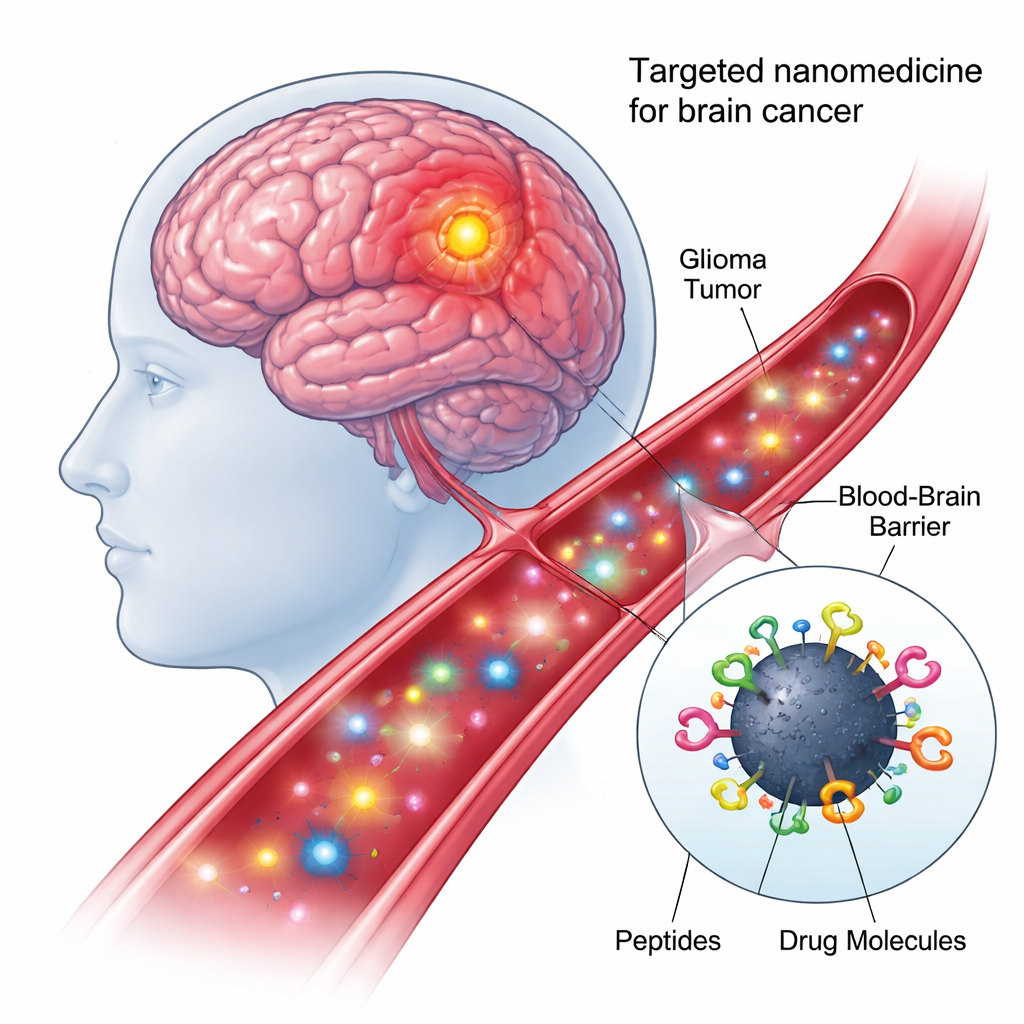
Finding and entering the right brain cells
The first peptide on the nanoparticle, called shPep-1, is designed to recognize a receptor named IL13Rα2. This receptor is found at high levels on many aggressive brain tumors—including adult and pediatric glioblastoma and diffuse intrinsic pontine glioma—but appears only weakly on normal brain cells. By binding IL13Rα2, shPep-1 helps the carbon dots home in on tumor cells and enter them through receptor-mediated uptake. The second, longer peptide, lnPep-1, contains a nuclear localization signal: a short sequence that helps cargo slip through the nuclear membrane and reach the cell’s control center, where DNA is stored. Together, these two peptides give the nanoparticle both tumor preference and the ability to reach the nucleus once inside.
Delivering a coordinated drug attack
The therapeutic payload consists of two small molecules. The first is epirubicin, a well-known chemotherapy drug that kills cancer cells by inserting itself into DNA and blocking enzymes needed to repair double-stranded breaks. The second is a metabolite called 5-aminoimidazole-4-carboxamide (AIC), produced from the brain tumor drug temozolomide at physiological pH. In earlier work, the same group showed that combining epirubicin and AIC on a carbon dot enhanced cancer cell killing compared with either drug alone. In the new design, both are co-delivered on the dual-peptide nanoparticle, aiming for a synergistic effect right in the tumor cell nucleus.
Potent in tumor cells, milder to normal cells
To test how well the nanomodel worked, the team exposed several high-grade glioma cell lines—adult glioblastoma, pediatric glioblastoma, and a diffuse intrinsic pontine glioma model—to the quadruple-conjugated carbon dots. Even at very low concentrations (as little as 50 nanomolar), tumor cell viability fell to about half of untreated controls, and at higher doses, all tested conjugates killed most tumor cells. Importantly, the quadruple nanomodel outperformed simpler “single-peptide” versions, even though it carried less epirubicin overall. When tested on noncancerous vascular smooth muscle cells, it was far less toxic: these cells required roughly 25–40 times more nanoparticle to see a similar loss of viability, suggesting a useful safety window driven by tumor targeting instead of brute-force dosing.
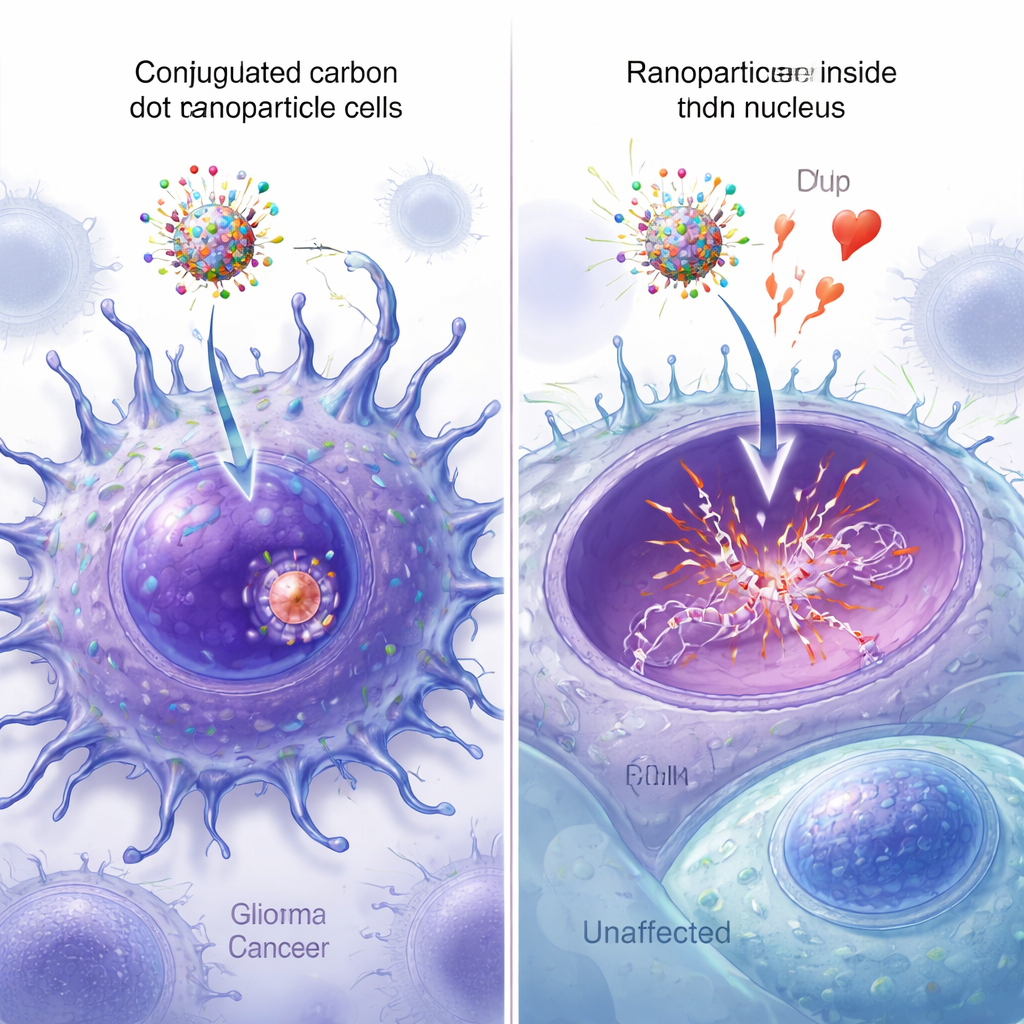
Seeing the nanoparticles reach the nucleus
Fluorescent versions of the nanoparticles allowed the researchers to watch how well each design entered cells and reached the nucleus. Under the microscope, carbon dots equipped with both peptides showed the brightest signal spread throughout the cancer cells, with strong overlap between the green nanoparticle fluorescence and blue nuclear staining. Quantitative image analysis confirmed that the dual-peptide particles had the highest nuclear colocalization, significantly exceeding that of particles carrying only one peptide or none. Additional spectroscopy, mass spectrometry, and atomic force microscopy measurements verified that all four components were successfully attached to the carbon dots and modestly increased their size, without disrupting the key DNA-binding function of epirubicin.
What this could mean for future brain cancer care
For patients, these findings are still an early-stage, cell-dish demonstration rather than a ready therapy. Yet they showcase a promising concept: a small, stable, and relatively easy-to-make carbon-based nanoparticle that can be rapidly customized with targeting peptides and drug combinations. By steering drugs specifically to tumor cells that overproduce IL13Rα2 and concentrating them in the nucleus, such nanomedicines could one day allow lower doses, fewer side effects, and more personalized treatment strategies for highly lethal brain tumors like glioblastoma and diffuse intrinsic pontine glioma.
Citation: Cilingir, E.K., Hettiarachchi, S.D., Rathee, P. et al. Development of a quadruple-conjugated carbon dot nanomodel for targeted glioma therapy. Commun Chem 9, 96 (2026). https://doi.org/10.1038/s42004-026-01900-3
Keywords: glioblastoma, nanomedicine, carbon dots, targeted drug delivery, brain tumors