Clear Sky Science · en
A generalizable assay for intracellular accumulation to profile cytosolic drug delivery in mammalian cells
Why getting medicines inside cells matters
Many of today’s most promising medicines—tiny drug molecules, short protein fragments called peptides, and full-sized proteins—are designed to act on targets inside our cells. But the cell membrane is a tough security gate. A drug can stick to the outside of a cell or get trapped in internal sacs called endosomes and still never reach the watery interior, the cytosol, where many targets live. This article describes a new laboratory test, called the CHAMP assay, that tells researchers not just whether a molecule enters a cell, but whether it truly arrives in the cytosol, where it can do its job.
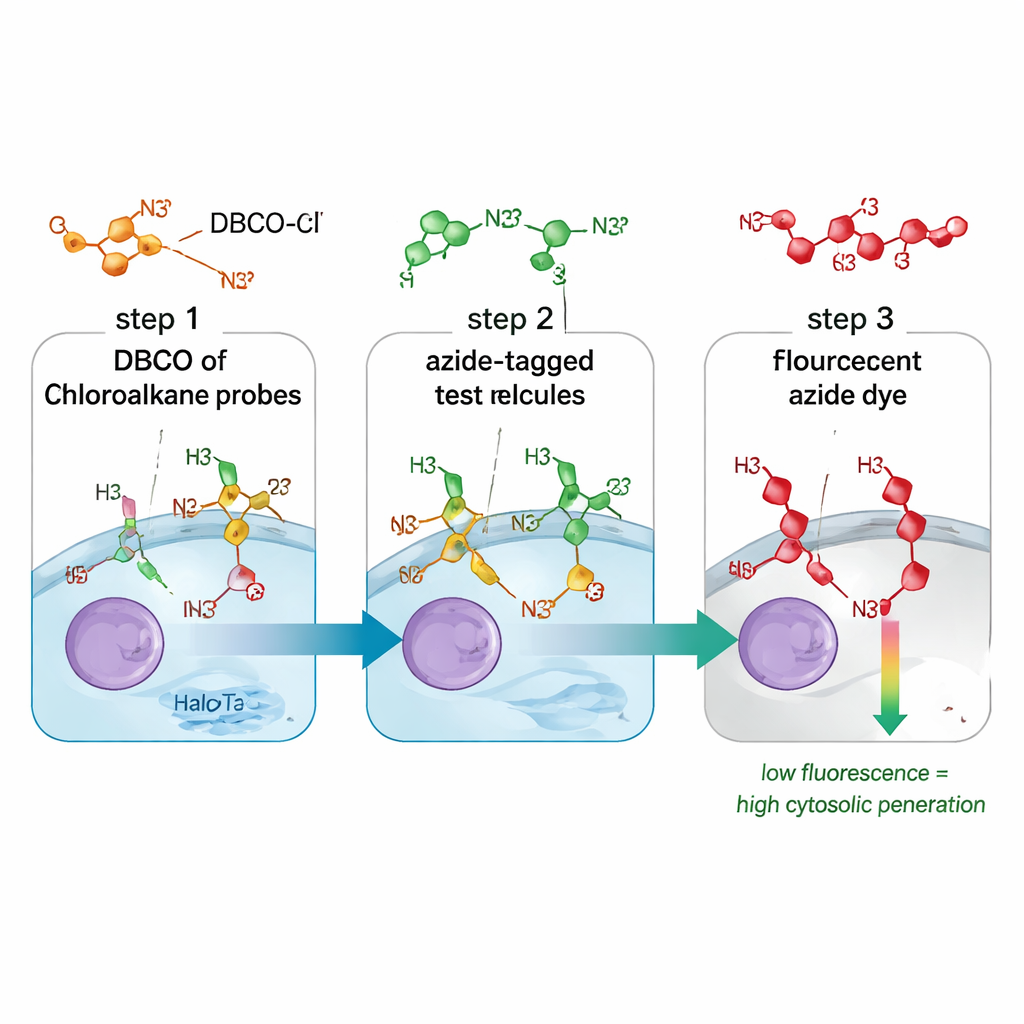
A new way to track drug entry
Traditional methods for measuring drug uptake often blur together molecules that merely touch the cell surface, sit in endosomes, or successfully reach the cytosol. They may also rely on bulky fluorescent tags that change how a molecule behaves. The CHAMP assay solves both problems by combining a small chemical “handle” called an azide with a protein marker named HaloTag, which is engineered to float freely in the cytosol of cultured human cells. First, cells are treated with a linker molecule that snaps DBCO, a strained form of alkyne, onto HaloTag. Next, scientists add their test compound, which carries the tiny azide tag. Only if that compound crosses the cell membrane into the cytosol can the azide and DBCO click together in a highly selective chemical reaction. A final fluorescent azide dye reveals how many DBCO sites are left: dim cells mean the test compound got in and occupied most sites; bright cells mean poor cytosolic access.
From concept to working assay
The authors first confirmed that HaloTag was properly made and located in the cytosol of human HeLa cells by using a standard chloroalkane dye and imaging. They then optimized each step of CHAMP: how much DBCO linker to add, how long to incubate, and which fluorescent azide dye gave a strong, specific signal without harming cells. Flow cytometry—an instrument that measures fluorescence in thousands of individual cells—showed that the assay was fast, robust, and tunable. Crucially, the team demonstrated that fluorescence really arose from specific reactions at HaloTag sites rather than from dyes sticking nonspecifically to cell components, and that the small azide tag did not noticeably disturb how molecules entered cells.
What the assay reveals about small drugs and peptides
With CHAMP in hand, the researchers challenged it with hundreds of azide-labelled small molecules and systematically varied features like charge, size, and flexibility. They observed, for example, that turning a negatively charged carboxylic acid into a neutral amide increased cytosolic accumulation, and that the degree and position of nitrogen methylation changed how well compounds got inside. By comparing reactions on free beads versus live cells, they could separate intrinsic chemical reactivity from the barrier imposed by the membrane itself. The team also applied CHAMP to a panel of common antibiotics, showing that some reached the cytosol far better than others—important information when treating bacteria that hide inside host cells.
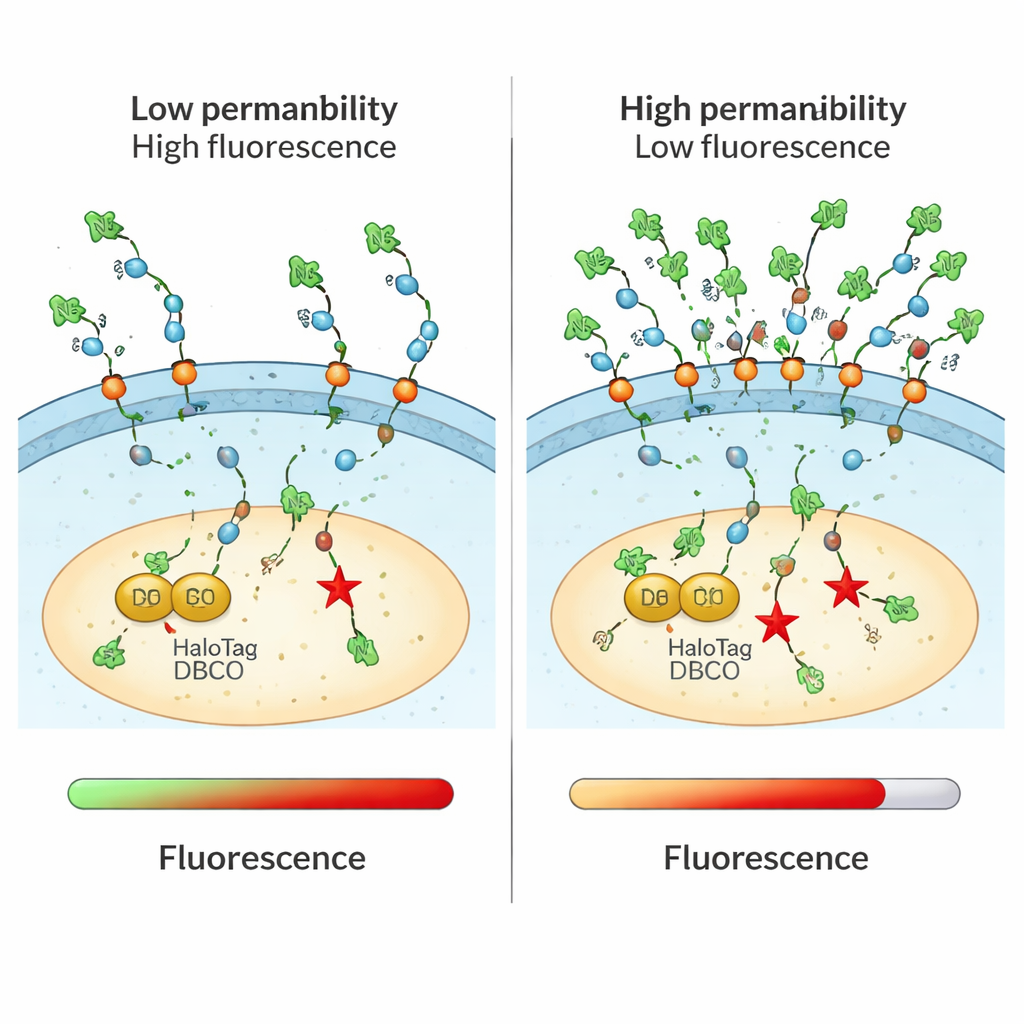
Supercharged peptides and proteins crossing the barrier
The assay was especially powerful for studying larger, highly charged molecules that are difficult to track by other means. The authors examined cell-penetrating peptides made from multiple arginine residues and confirmed a clear trend: longer, more positively charged chains delivered more material into the cytosol, up to a point. They probed how reversing the handedness (stereochemistry) of certain amino acids altered entry, finding that mirror-image versions sometimes accumulated better, hinting at subtle chiral interactions with the membrane. CHAMP also showed that “supercharged” proteins—engineered to carry many positive charges—could reach the cytosol more effectively than their normal counterparts, supporting their potential as carriers for large therapeutic cargoes.
Design rules for future intracellular medicines
By systematically testing small molecules, peptides, macrocyclic peptides, and modified backbones, the study uncovered general design principles. Macrocyclic peptides, whose backbones are closed into rings, tended to reach the cytosol more readily than flexible linear versions. Carefully placed N-methyl groups on peptide backbones modestly boosted entry, but too many could be counterproductive. These structure–permeation relationships, revealed with a single standardized assay, offer practical guidance for chemists seeking to craft molecules that not only bind their targets but can actually get to them inside cells.
How this helps drug development
In simple terms, the CHAMP assay is a finely tuned traffic counter at the cell’s front door and inner hallway. It tells scientists which experimental drugs truly make it past the membrane and into the cytosol, without being misled by molecules stuck on the surface or in internal compartments. Because it works with a wide variety of molecular shapes and sizes and uses only a tiny azide tag, CHAMP can be integrated into high-throughput discovery pipelines. Over time, this should speed the design of more effective treatments for diseases where the most important targets are locked away inside our cells.
Citation: Bhandari, S., Ongwae, G.M., Dash, R. et al. A generalizable assay for intracellular accumulation to profile cytosolic drug delivery in mammalian cells. Commun Chem 9, 94 (2026). https://doi.org/10.1038/s42004-026-01898-8
Keywords: intracellular drug delivery, cell membrane permeability, cytosolic accumulation, cell-penetrating peptides, bioorthogonal click chemistry