Clear Sky Science · en
Hydrogen-assisted dehydrochlorination of 1,1,1,2-tetrafluoro-2-chloropropane to 2,3,3,3-tetrafluoropropene over Pd-Ag/nano-MgF2 with optimized Pd isolated sites
Cleaner Coolants for a Warming World
Air conditioners and refrigerators keep us comfortable, but many of the chemicals that make them work are powerful greenhouse gases. This study tackles a crucial challenge: how to make one of the most important next‑generation refrigerants, HFO‑1234yf, more efficiently and with a smaller carbon footprint. By designing a smarter catalyst—essentially a microscopic chemical machine—the authors show a way to produce this low‑warming refrigerant at lower temperatures and with far fewer unwanted byproducts.
Why We Need Better Refrigerant Chemistry
Traditional hydrofluorocarbons (HFCs), like HFC‑134a used in car air conditioners, trap heat in the atmosphere thousands of times more strongly than carbon dioxide. International agreements such as the Kigali Amendment are phasing them down, pushing industry toward hydrofluoroolefins (HFOs), which have similar performance but much lower global warming potential (often under 10). HFO‑1234yf is the leading candidate replacement, yet current industrial methods to make it rely on high‑temperature, energy‑intensive steps that also shorten catalyst lifetimes and increase waste. A cooler, more selective route to HFO‑1234yf could therefore cut both emissions and operating costs.
A Gentler Route to a Key Molecule
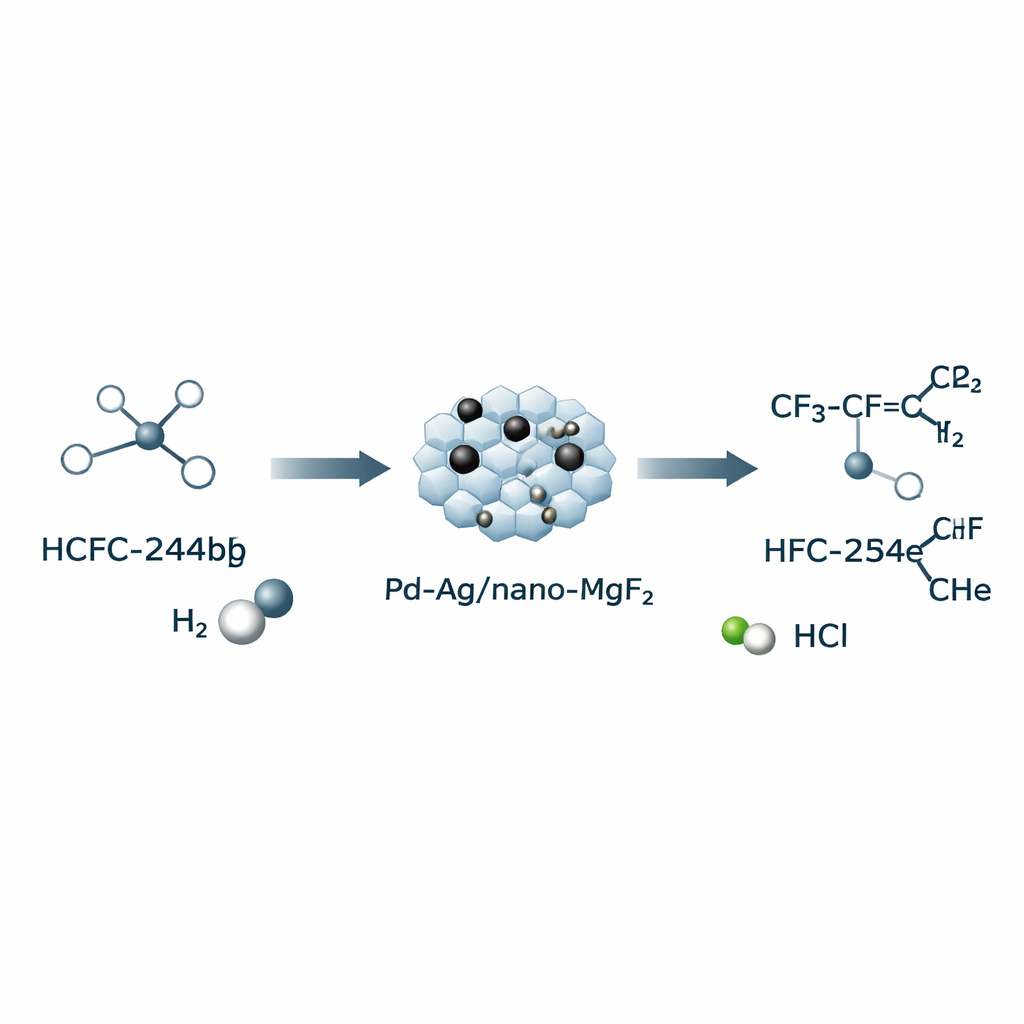
The authors focus on converting a related compound, HCFC‑244bb, into HFO‑1234yf by removing hydrogen chloride (HCl) with help from hydrogen gas. This “hydrogen‑assisted dehydrochlorination” lets the reaction proceed at a relatively mild 270 °C—far below the 600–800 °C often required by purely thermal methods. Central to the process is a tailor‑made solid catalyst: tiny hollow particles of magnesium fluoride (nano‑MgF₂) decorated with palladium (Pd) and silver (Ag). By changing the order in which Pd and Ag are added during preparation, the team can control how well the two metals mix—forming alloys of different character—and how the active Pd sites ultimately appear on the surface during the reaction.
Turning Metal Clusters into Single Atoms
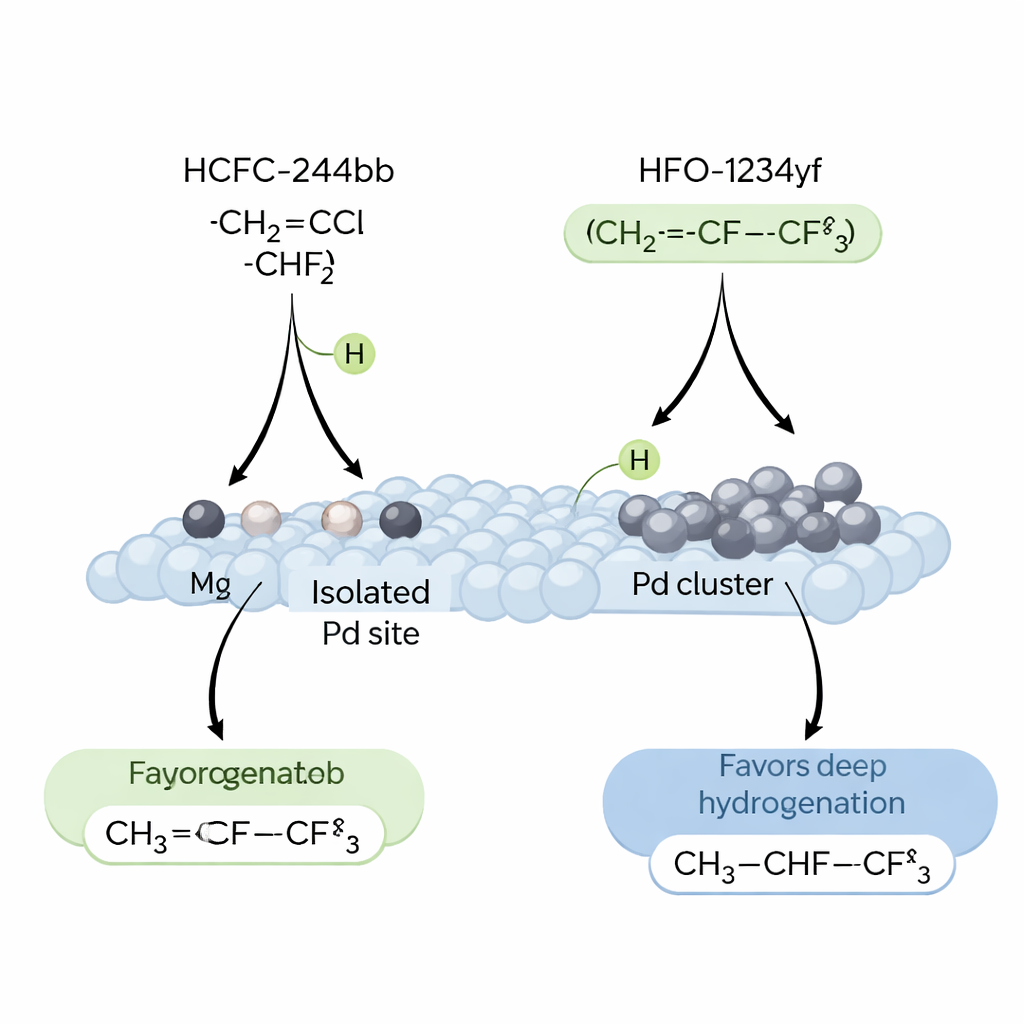
During the first several hours on stream, the catalysts show an “induction period”: the conversion of HCFC‑244bb slowly declines, while selectivity to the desired HFO‑1234yf steadily rises. Careful microscopy, spectroscopy, and surface measurements reveal why. As the reaction proceeds, hydrogen chloride formed in situ chlorinates the metal particles, breaking up larger Pd clusters and reshaping them into isolated Pd atoms anchored on the nano‑MgF₂ surface. Catalysts that start with a higher degree of Pd–Ag alloying—achieved when Pd is added first, then Ag—undergo the most extensive restructuring and end up with the greatest density of single‑atom Pd sites. This optimized material converts about 60% of HCFC‑244bb and directs roughly 82% of the product toward HFO‑1234yf, while strongly suppressing deeper hydrogenation to an unwanted saturated compound, HFC‑254eb.
How Hydrogen Behavior Controls the Outcome
To understand why isolated Pd atoms perform so well, the researchers probed how hydrogen, the starting refrigerant molecule, and the product interact with different catalyst surfaces. They found that HCFC‑244bb and hydrogen both prefer to adsorb on Pd, not Ag, but that the strength and nature of hydrogen binding are crucial. Large Pd aggregates tend to create “spillover” hydrogen that remains strongly bound at high temperatures and promotes over‑hydrogenation, turning valuable HFO‑1234yf into less useful saturated products. In contrast, single‑atom Pd sites hold hydrogen in a more reactive but less persistent form, just right for cleanly removing HCl and forming the desired carbon–carbon double bond. Computer simulations using density‑functional theory back this up, showing that these isolated Pd–fluoride environments make it easier to strip hydrogen from key intermediates on the path to HFO‑1234yf, while making secondary hydrogenation steps energetically less favorable.
From Lab Insight to Greener Cooling
In practical terms, the best Pd–Ag/nano‑MgF₂ catalyst delivers HFO‑1234yf formation rates tens of times higher than earlier solid catalysts that relied on harsh, high‑temperature dehydrochlorination. The work demonstrates that controlling metal atoms one by one—not just as nanoparticles—can dramatically steer reaction pathways, improving both efficiency and selectivity. For non‑specialists, the key message is that better catalyst design can translate directly into cleaner production of modern refrigerants. That, in turn, helps the world meet climate goals while keeping our buildings, vehicles, and food supplies cool.
Citation: Yang, C., Mao, W., Dong, X. et al. Hydrogen-assisted dehydrochlorination of 1,1,1,2-tetrafluoro-2-chloropropane to 2,3,3,3-tetrafluoropropene over Pd-Ag/nano-MgF2 with optimized Pd isolated sites. Commun Chem 9, 93 (2026). https://doi.org/10.1038/s42004-026-01896-w
Keywords: refrigerants, catalysis, palladium-silver, greenhouse gases, hydrofluoroolefins