Clear Sky Science · en
Metal surface-triggered DNAzyme catalysis for efficient DNA cleavage
Metals That Make DNA Cut Itself
Most of us think of metal as something solid and inert—good for coins, pots, and wiring, but not for chemistry in a glass of water. This study overturns that intuition by showing that bare pieces of metal can switch on tiny DNA-based catalysts, called DNAzymes, with nothing more than pure water and air. The work reveals an unexpected way that everyday materials like copper and other metals can drive complex biological-style reactions at their surfaces, hinting at new tools for sensors, medicine, and even origins-of-life chemistry. 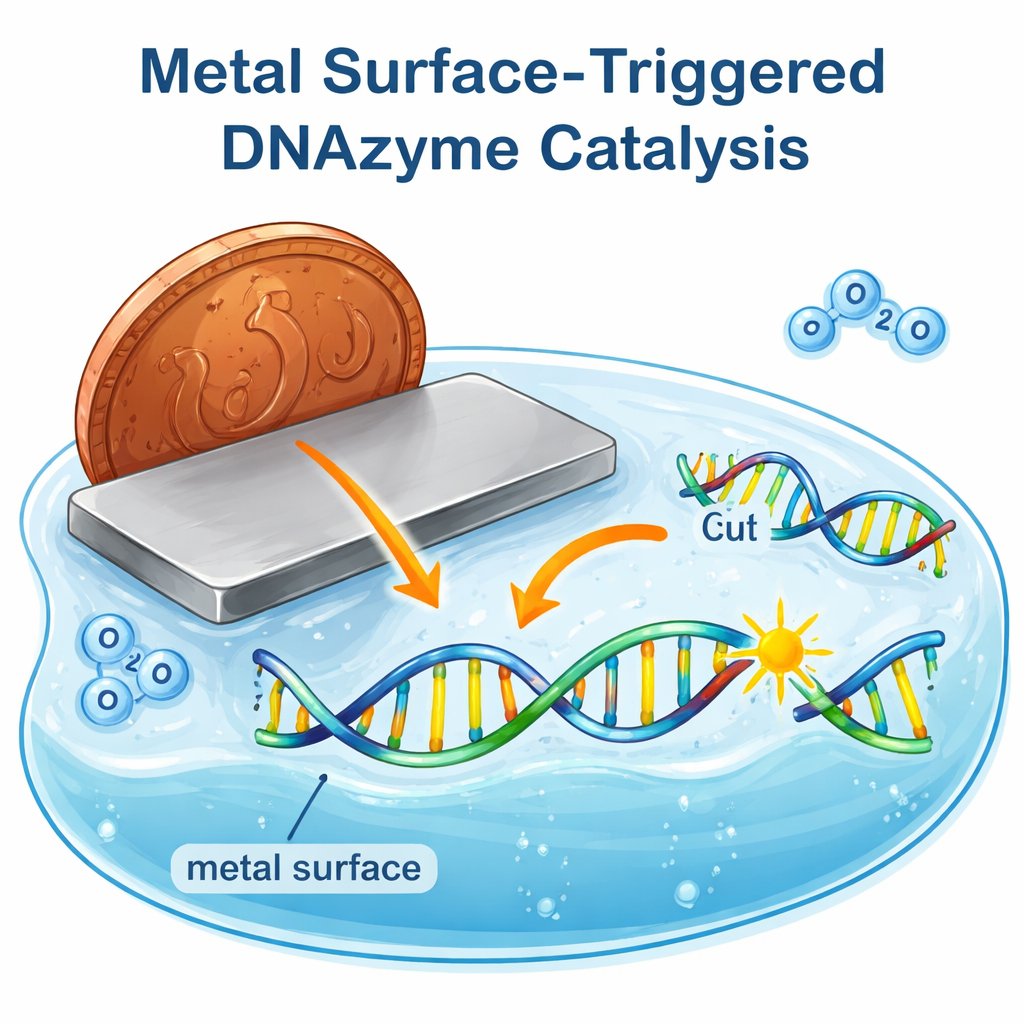
What Are DNA Scissors?
DNAzymes are short strands of DNA that fold into shapes able to speed up specific chemical reactions, much like protein enzymes do. Many known DNAzymes act as molecular scissors that cut other strands of DNA or RNA, but they almost always need dissolved metal ions—like copper, zinc, or magnesium—floating in solution to work. One such DNAzyme, called PL, is a self-cutting strand that normally relies on copper ions and certain helper molecules such as vitamin C or hydrogen peroxide. These helpers participate in redox (electron-transfer) reactions that generate highly reactive oxygen species, which then attack and break the DNA backbone at precise positions.
When a Copper Wire Becomes a Cofactor
While exploring an electrochemical setup to control PL activity, the researchers noticed something surprising: simply dipping a bare copper wire into a solution containing PL and double-distilled water led to efficient DNA cutting, even without any added copper ions, buffers, or salts. The copper surface by itself matched or outperformed traditional mixtures of copper ions plus vitamin C or hydrogen peroxide, and it cut the DNA at exactly the same site. Follow-up tests showed that many copper objects—sheets, funnels, pots, and even coins from different currencies—could trigger PL to cleave, with the extent of reaction depending on how much surface area of metal contacted the droplet. Freshly polished copper worked slightly less well than aged, oxidized copper, hinting that a thin layer formed by air exposure might actually help the reaction.
Which Surfaces Work—and Why
To see whether this was a copper-only curiosity, the team screened 24 metals and 10 non-metal materials. They found that only certain metals, such as copper, tantalum, and vanadium, strongly activated PL, while glass, plastic, wood, and other non-metals did nothing. Measurements showed that small amounts of metal ions do leach into water from active surfaces, but these ions alone were too weak to explain the strong DNA cutting. The missing ingredient turned out to be reactive oxygen derived from dissolved air. Using chemical scavengers and enzymes, the authors showed that superoxide—an energetic form of oxygen with an extra electron—is essential. When dissolved oxygen was removed by nitrogen bubbling, PL activity nearly vanished; letting oxygen back in restored cutting. Together the data support a cycle in which the metal surface and its leached ions convert dissolved oxygen into superoxide and hydrogen peroxide right at the solid–liquid interface, and these species then drive the DNA scission. 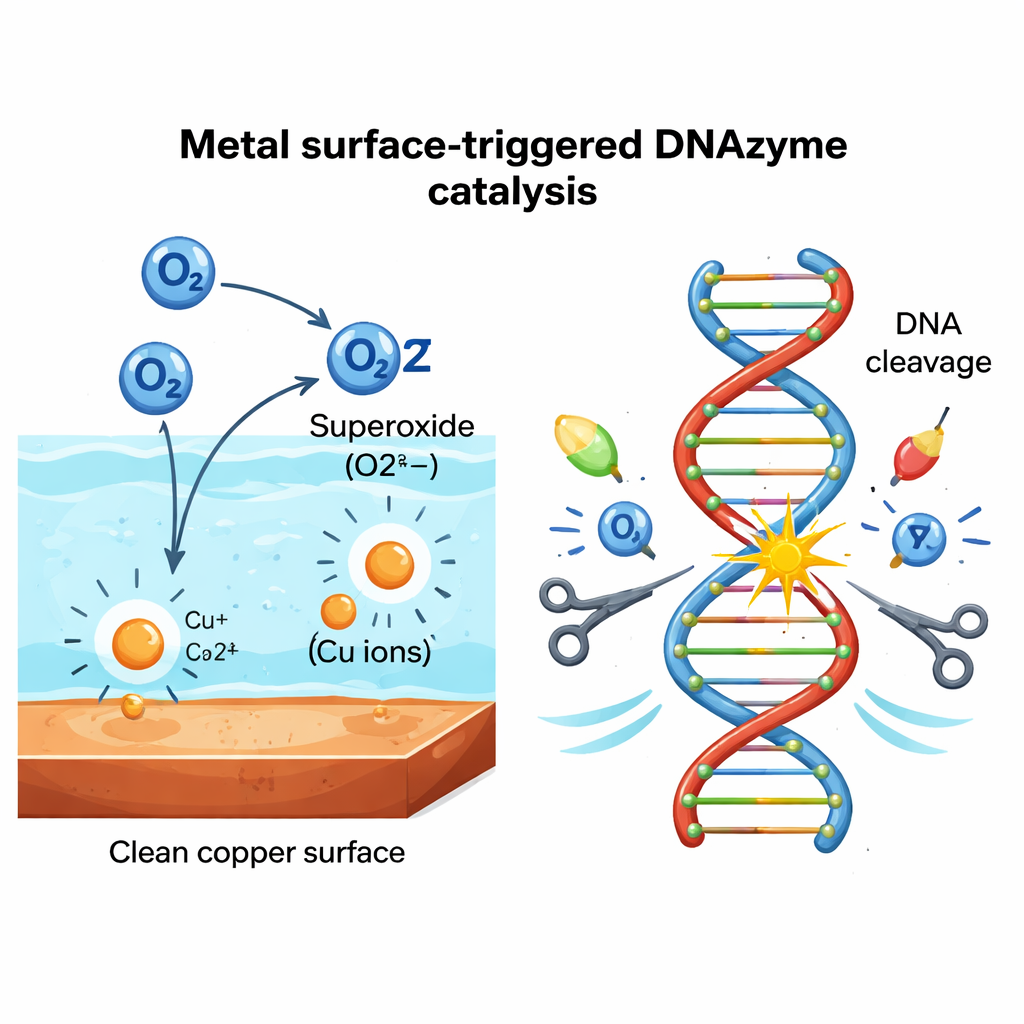
Helpers, Blockers, and Other DNA Enzymes
The surface-triggered chemistry could be dialed up or down using familiar molecules. Chelators like EDTA, which grab metal ions, slowed the reaction. Catalase, an enzyme that breaks down hydrogen peroxide, and dyes that soak up superoxide also reduced DNA cutting, confirming the role of these reactive oxygen species. In contrast, small compounds such as vitamin C, glutathione, and catechol boosted activity by feeding the redox cycle that makes superoxide near the metal surface. Remarkably, the effect was not unique to PL: other DNAzymes that cut DNA or RNA, including F-8, Ag10c, and I-R3, were also activated by the corresponding bulk metals (for example, silver metal for an Ag-dependent DNAzyme and zinc metal for a Zn-dependent one). This suggests that “metal surface-activated” DNA catalysis could be a broad phenomenon rather than a one-off oddity.
Why This Matters Beyond the Lab
For readers outside chemistry, the central message is that solid metal surfaces can act like invisible chemical partners for DNA-based catalysts, using only air and water to generate the reactive species needed to cut DNA. Instead of having to dissolve precise amounts of metal ions, one can simply touch a suitable metal surface to a DNA solution and let the interface do the work. This opens up possibilities for low-cost sensors that report on metal objects, tools to monitor or mop up harmful oxygen radicals in cells, and new ways to study how life-like reactions might occur on mineral or metal surfaces. In short, your copper coin is not just spare change—it can also be a tiny chemical factory for DNA scissors.
Citation: Jiang, F., Dong, Y., Yu, W. et al. Metal surface-triggered DNAzyme catalysis for efficient DNA cleavage. Commun Chem 9, 91 (2026). https://doi.org/10.1038/s42004-026-01893-z
Keywords: DNAzyme, metal surface catalysis, reactive oxygen species, copper interface chemistry, DNA cleavage