Clear Sky Science · en
Controlled spherulitic crystal growth from salt mixtures
Why salt crystals can look like flowers
Most of us think of crystals as sharp, faceted shapes, but in nature they often grow into spectacular spheres that resemble flowers or snowballs. These “spherulites” show up in volcanic rocks, kidney stones, and even in some diseases linked to misfolded proteins. This article explores how an everyday mineral, sodium sulfate—the same salt found in detergents and some building materials—can be coaxed into forming such intricate spherical crystals, and what this reveals about how complex structures self-assemble from simple ingredients.
From simple salts to sculpted spheres
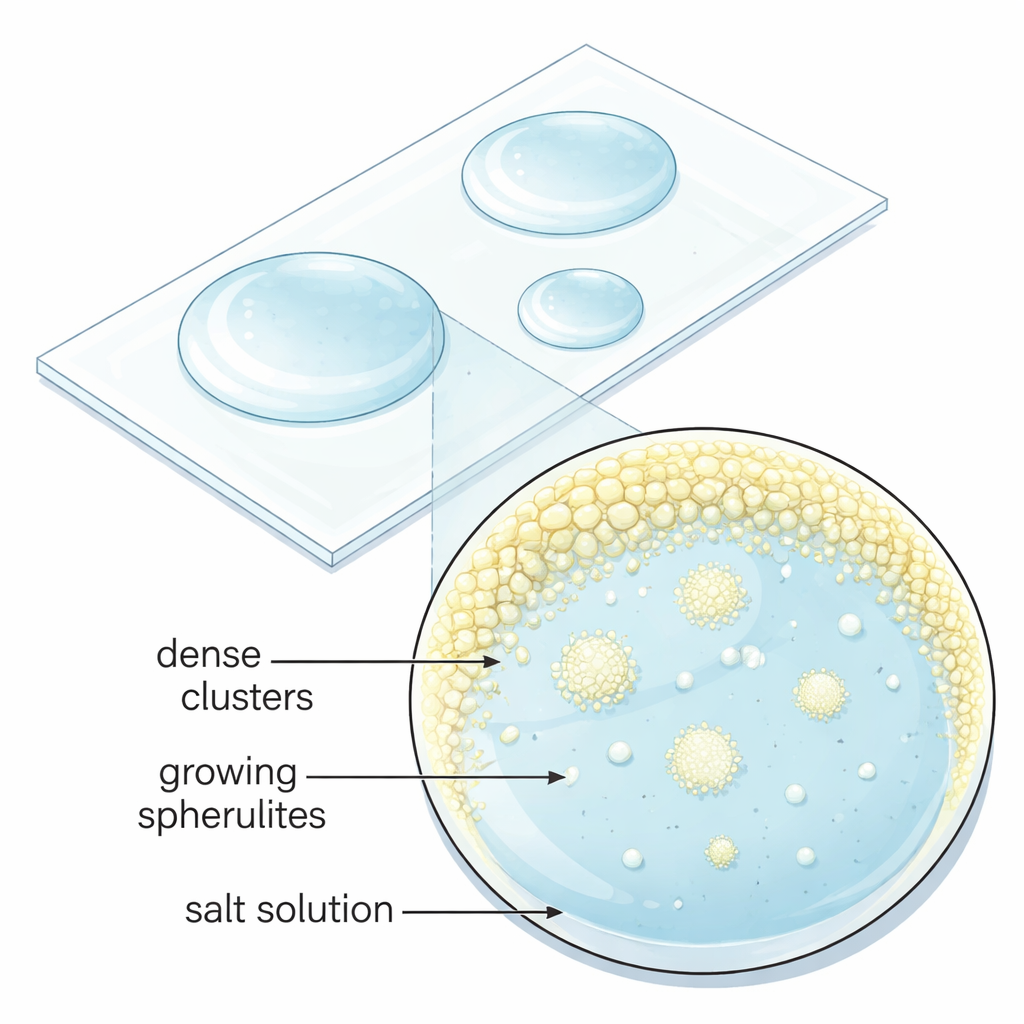
The researchers set out to understand when and how sodium sulfate grows into spherulites instead of ordinary blocky crystals. They prepared tiny droplets of water containing mixtures of sodium sulfate and other sulfate salts whose metal ions carry two positive charges, such as magnesium or iron. As these picoliter to microliter droplets slowly evaporated on glass slides at room temperature, the dissolved salts became more and more concentrated until crystals began to form. By systematically varying the mixing ratio, they found specific composition “sweet spots” where sodium sulfate consistently produced spherical, radially textured crystals. Outside these ranges, the same solution yielded either regular faceted grains or a featureless gel-like solid, showing that the spherical shapes require carefully tuned conditions.
Tiny liquid pockets that seed crystal spheres
Under the microscope, the team observed that spherulites did not appear directly from clear solution. Instead, evaporation first created small, dense liquid clusters enriched in dissolved ions near the droplet’s edge. These micron-sized pockets lingered for minutes before suddenly sprouting many spherulites at once. High-resolution electron microscope images of the dried structures revealed that each spherulite is built from countless nanometer-scale sodium sulfate crystals pointing roughly outward, which later fuse together. This behavior contradicts the textbook picture of crystal growth, where a single stable nucleus grows smoothly, and instead points to a multi-step “non-classical” pathway in which dense droplets and nanoparticles assemble and reorganize on their way to a final solid form.
When salty water turns peanut-butter thick
A crucial part of the story is how thick, or viscous, the solution becomes as water is lost. By tracking how fast spherulites expanded and by directly measuring the flow properties of related salt solutions, the authors showed that mixed sodium–magnesium or sodium–iron solutions become extraordinarily viscous—up to about 100 times thicker than honey—right when spherulites start to form. This near “peanut-butter” consistency slows down ion motion so much that diffusion, not surface chemistry, limits how fast crystals can grow. In this sluggish environment, countless tiny clusters and nanocrystals form and have time to stick together into spherical aggregates rather than growing into a few large, well-shaped crystals. The divalent metal ions are key: they bind water tightly and link into short chains or networks, which both raise the viscosity and help generate an amorphous, gel-like background around the growing spherulites.
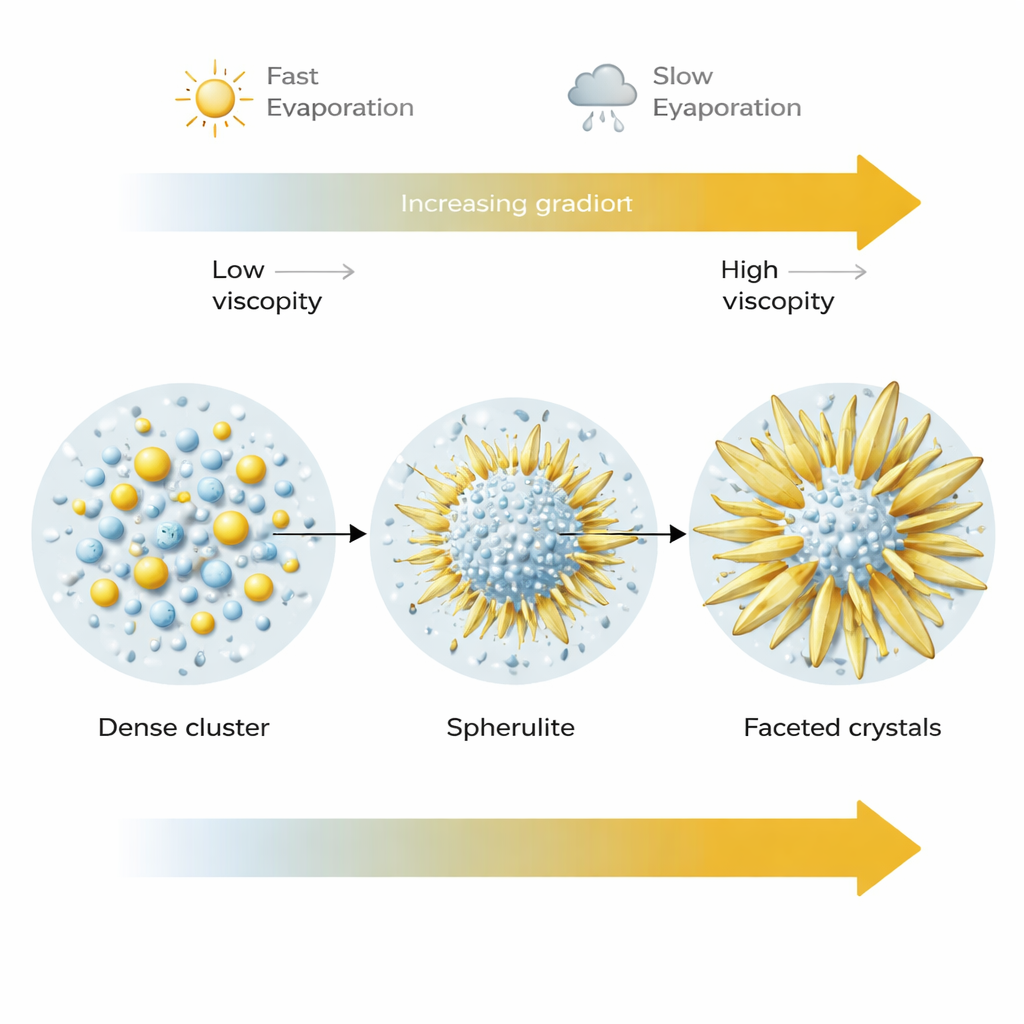
How drying speed reshapes the final crystals
Evaporation rate turned out to be another powerful control knob. At faster drying (lower humidity), many spherulites nucleated but stayed relatively small, locked into their metastable spherical form as the surrounding fluid quickly thickened. At slower drying (higher humidity), the same initial spheres had more time and access to dissolved ions. Their tiny building blocks could rearrange and merge into larger, smoother, and more faceted crystals, often sprouting blade-like outgrowths that eventually converted into the thermodynamically stable sodium sulfate form. In other words, the spherical structures act as a transient stage in a growth journey that can end with very different shapes, depending on how quickly the solution dries and how easily material can still move through it.
Why this matters beyond pretty crystals
In plain terms, the study shows that beautiful, flower-like salt spheres arise when concentrated mixtures of simple salts become thick enough to slow everything down, yet not so thick that growth stops altogether. Under these “just right” conditions, dense liquid pockets and swarms of tiny crystals self-assemble into spherical clusters that can later evolve into more stable, faceted grains. Understanding and controlling this delicate balance between composition, viscosity, and evaporation opens the door to designing custom crystal textures for applications ranging from stronger building materials and improved pharmaceuticals to better models of geological and biological crystal formation.
Citation: Heeremans, T., Lépinay, S., Le Dizès Castell, R. et al. Controlled spherulitic crystal growth from salt mixtures. Commun Chem 9, 90 (2026). https://doi.org/10.1038/s42004-026-01892-0
Keywords: spherulites, sodium sulfate, crystal growth, non-classical nucleation, salt solutions