Clear Sky Science · en
First crystal structure of an adduct formed upon reaction of a vanadium compound with human serum transferrin
Why a blood protein and a metal drug matter
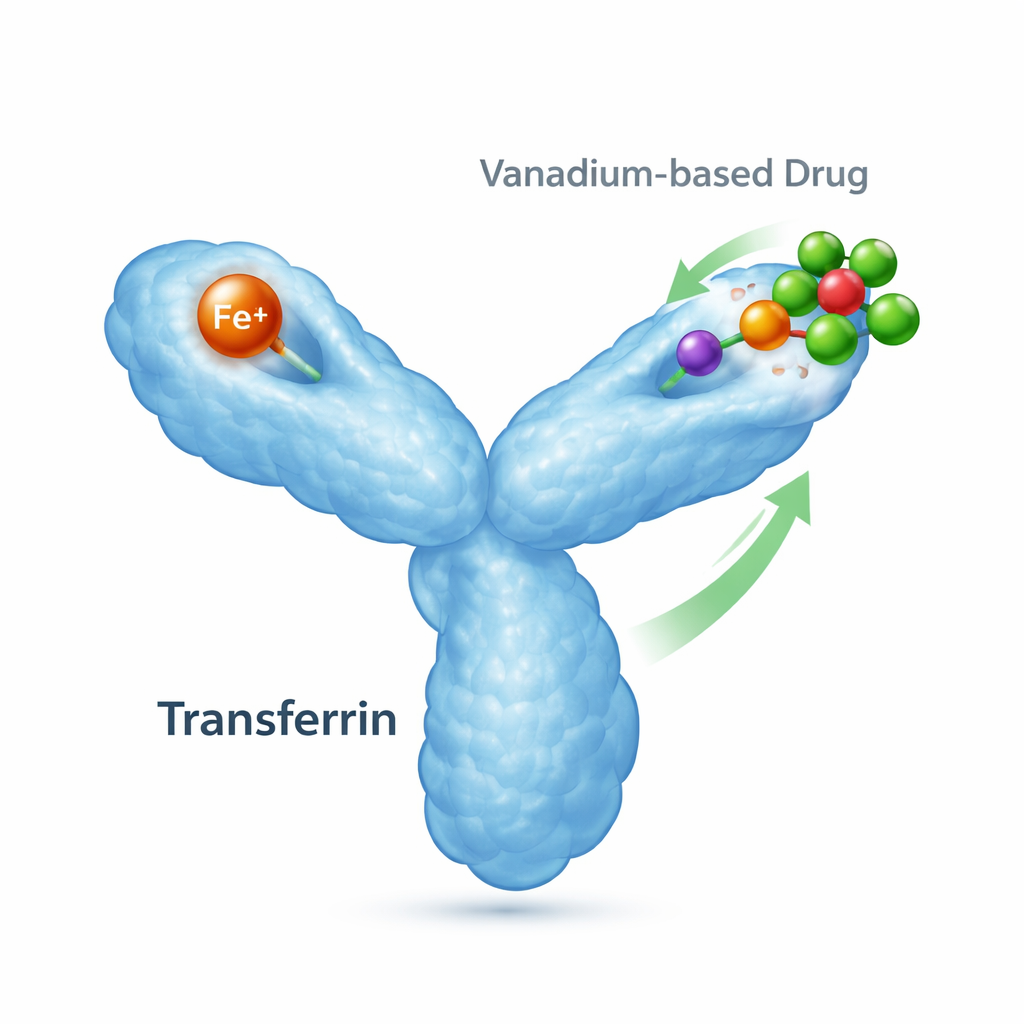
Many experimental drugs contain metal atoms that can help treat diseases ranging from diabetes to cancer. But once these metal-based medicines enter the bloodstream, they must hitch a ride on the body’s natural transport proteins. This study looks at how one such protein, human serum transferrin—the main carrier of iron in our blood—interacts with a promising vanadium compound, shedding light on how metal drugs are ferried through the body and how they might be fine‑tuned for safer, more effective therapies.
The body’s iron courier and its side jobs
Transferrin is an 80‑kilodalton protein whose day job is to grab iron and deliver it to cells that need it. It has two “lobes,” each capable of binding one iron ion, and it changes shape depending on whether iron is present. When iron binds, a lobe snaps shut; without iron, it stays open. Specialized receptors on cell surfaces recognize these iron-loaded forms and internalize them, making transferrin central to iron balance and cell health. However, transferrin can also bind other metals, including those from medical treatments, meaning it may influence where metal-based drugs travel and how they act.
A shape snapshot of vanadium on transferrin
The researchers focused on a well‑studied vanadium drug candidate called bis(acetylacetonato)oxovanadium(IV), often written as [VIVO(acac)₂]. Earlier work showed that this compound, or species derived from it in water, can attach to transferrin, but nobody had seen exactly how. Using X‑ray crystallography, the team obtained high‑resolution structures of transferrin that carries iron only in its C‑terminal lobe (the “FeC” form), both before and after exposure to the vanadium drug. In the vanadium-treated crystals, they observed not the original drug, but a transformed vanadium‑oxygen cluster bound in the iron‑free N‑terminal lobe. This gave them the first direct structural picture of a vanadium fragment attached to human transferrin.
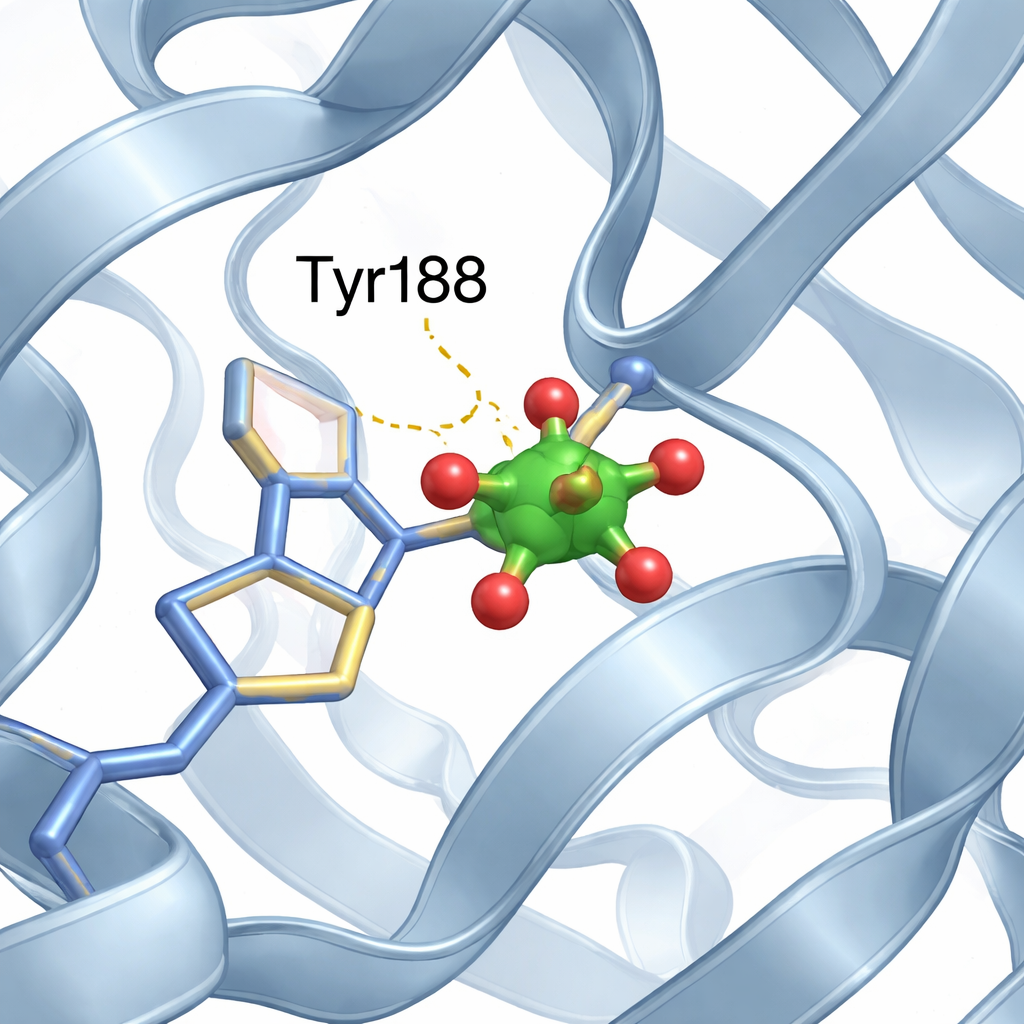
A tiny vanadium cluster finds a niche
Inside the N‑lobe’s iron site, the team identified a small cluster made of two vanadium atoms and oxygen atoms, best described as a modified “divanadate” unit. One of the cluster’s oxygen positions is taken over by the oxygen from a specific amino acid, tyrosine 188 (Tyr188), forming a direct bond between protein and metal cluster. Additional gentle interactions, such as hydrogen bonds from nearby protein segments, further stabilize the cluster in place. Importantly, despite this new metal guest occupying an iron‑binding pocket, the overall protein maintains its usual open N‑lobe and closed C‑lobe arrangement, almost identical to the vanadium‑free structure.
How transferrin reshapes the chemistry of vanadium
In solution at the pH and concentration used for crystallization, the particular two‑vanadium species seen in the crystal is normally only a minor player; larger vanadium‑oxygen clusters tend to dominate. Spectroscopic measurements and theoretical speciation calculations suggest that the original vanadium drug slowly oxidizes and falls apart in water, producing simple vanadate ions that then assemble into a variety of clusters. The crystal structure indicates that transferrin selectively stabilizes this small divanadate-like fragment over the larger clusters, essentially “choosing” one chemical form from a crowded solution mixture by providing a snug binding site at Tyr188 and a supportive hydrogen‑bond network.
Keeping its handshake with the cell receptor
Because transferrin must bind a receptor on cell surfaces to deliver iron, the researchers asked whether attaching this vanadium cluster would disrupt that crucial handshake. Using non‑denaturing gel electrophoresis and a sensitive technique called biolayer interferometry, they measured how well vanadium‑treated transferrin bound to the transferrin receptor compared with the untreated iron‑only form. Both behaved almost identically, with very strong binding in the low nanomolar range. This confirms that the vanadium cluster, while nestled in the N‑lobe, does not significantly alter transferrin’s shape where the receptor interacts, and therefore should not prevent the protein from being recognized by cells.
What this means for metal-based medicines
For non‑specialists, the takeaway is that this study provides a molecular snapshot of how a vanadium drug derivative attaches to the body’s main iron courier without derailing its normal function. Transferrin can capture a specific small vanadium‑oxygen cluster in an iron site, yet the protein’s overall shape and its ability to bind its receptor remain essentially unchanged. This helps explain how vanadium drugs might circulate attached to transferrin and highlights that different proteins can favor different vanadium cluster sizes. Such insights are key to designing smarter metal-based therapies whose behavior in the bloodstream—what forms they adopt, where they go, and how long they last—can be predicted and controlled.
Citation: Banneville, AS., Lucignano, R., Paolillo, M. et al. First crystal structure of an adduct formed upon reaction of a vanadium compound with human serum transferrin. Commun Chem 9, 89 (2026). https://doi.org/10.1038/s42004-026-01891-1
Keywords: vanadium drugs, human serum transferrin, metal-based therapeutics, protein–metal binding, structural biology