Clear Sky Science · en
Lactam enables remote boronate rearrangements to C═N bonds
A new shortcut for building drug-like molecules
Chemists are always searching for faster, cleaner ways to build the complex molecules found in medicines and advanced materials. This study introduces a clever shortcut that lets a common ring-shaped structure, called a lactam, temporarily guide a reaction and then quietly step aside—making it possible to turn simple, readily available ingredients into valuable building blocks for pharmaceuticals without using expensive metals or elaborate preparation steps.
Turning a quiet helper into a temporary guide
Many modern drugs and functional materials rely on reactions of boronic acids, a family of boron-containing compounds prized for their reliability and mild behavior. Traditionally, to coax boronic acids into forming new bonds with carbon–nitrogen double bonds (C=N), chemists must pre-install special “directing groups” on starting materials. These groups act like handles that steer the reaction but cost extra time, money, and steps—and often work only on very reactive partners. The authors realized that lactams, ring-shaped relatives of the familiar amide bond found in proteins, could serve as built‑in, temporary guides. By coordinating to the boron atom, the lactam oxygen helps form a highly organized, four‑connected boron center that can shuffle an attached fragment to a distant position along the molecule, setting up new carbon–nitrogen bonds without any permanent directing group.
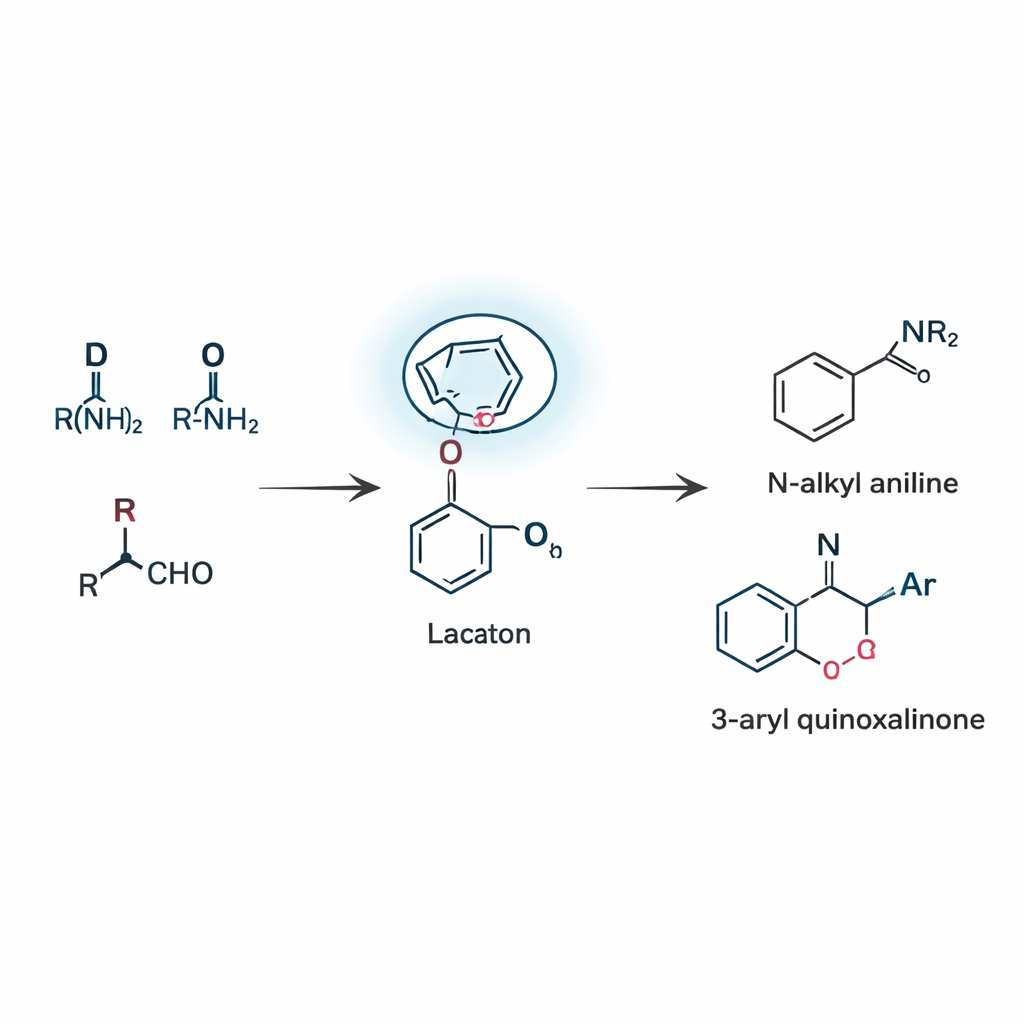
Building two families of useful products in one strategy
Using this concept, the team developed two related reactions that both rely on this lactam-guided reshuffling, known as a remote boronate rearrangement. In the first, three simple pieces—an aldehyde, an amine, and a boronic acid—come together to form N‑alkyl anilines, a core motif that appears in countless drug candidates and dyes. The reaction proceeds through a rare 1,5‑shift, in which an aryl group on boron hops over five atoms to reach the C=N bond. By carefully tuning the catalyst, solvent, and temperature, the researchers achieved respectable yields and showed that many different substituents, including halogens, alkyl groups, and heterocycles, are tolerated. In the second reaction mode, the same guiding idea is applied to quinoxalinones, a class of nitrogen-rich rings common in medicinal chemistry. Here, the reaction uses no added metal catalyst at all and still delivers 3‑aryl quinoxalinones efficiently, thanks to the lactam’s ability to engage boron and stabilize the key intermediate.
Green conditions and late-stage editing of medicines
Beyond demonstrating broad scope, the authors show that this strategy is practical for real-world molecules. Because the 1,4‑rearrangement on quinoxalinones proceeds under metal-free conditions in a specialized alcohol solvent, it avoids the need for expensive or toxic transition metals. The team applied the method to complex fragments derived from marketed drugs such as ibuprofen and other therapeutics that contain the quinoxalinone framework. In each case, the reaction installed new aryl groups at a specific position without disturbing other sensitive features. This kind of “late-stage functionalization” lets chemists rapidly decorate existing drug cores with new side chains, speeding up the search for improved potency, safety, or physical properties.
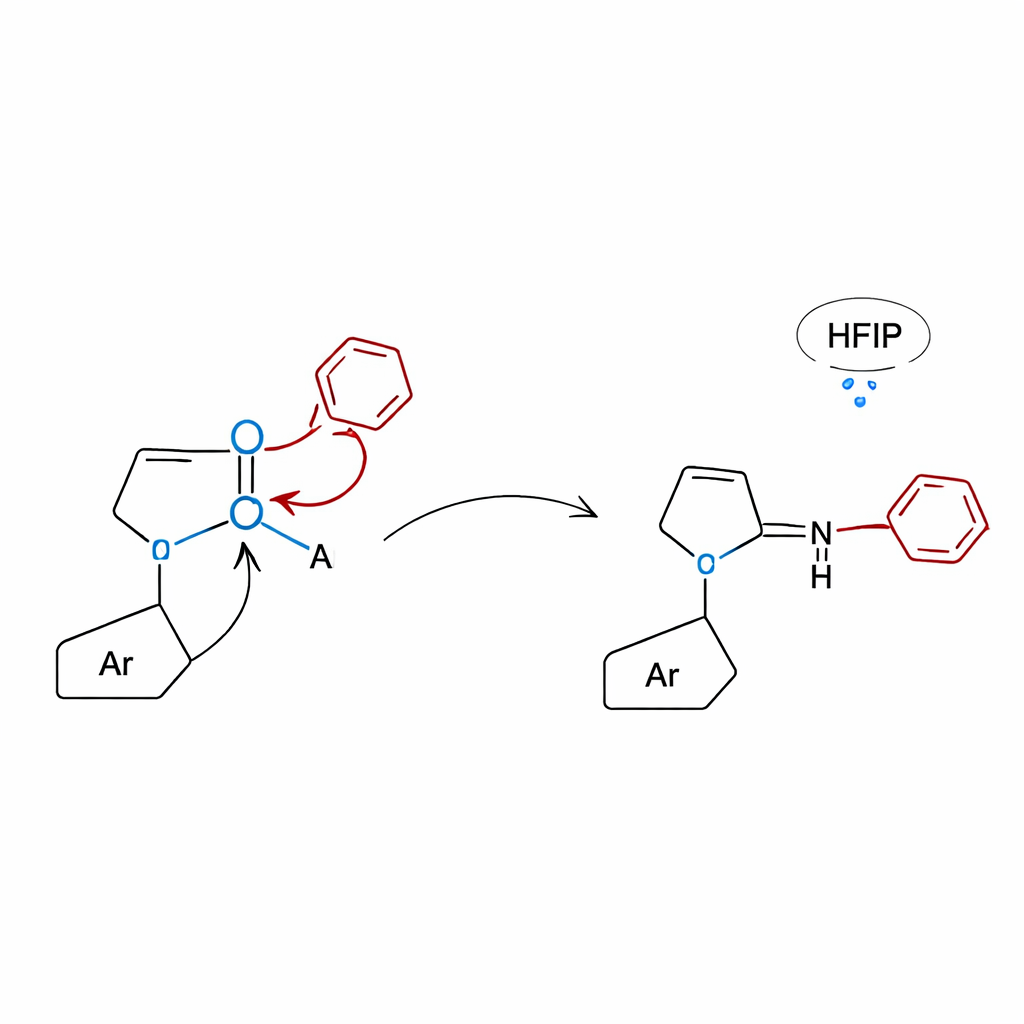
Peeking under the hood with theory and control tests
To understand why the lactam approach works so well, the researchers combined carefully designed control experiments with computer simulations. When molecules lacking the lactam moiety were tested, the reactions essentially shut down, confirming that the lactam ring is essential for activating the boronic acid and steering the rearrangement. Quantum chemical calculations revealed that the lactam oxygen binds to boron to form a compact, four‑connected structure from which the aryl group can migrate either over five atoms (1,5‑shift) to give N‑alkyl anilines or over four atoms (1,4‑shift) to give 3‑aryl quinoxalinones. In the quinoxalinone case, the hydrogen‑bonding network provided by the HFIP solvent further lowers the energy barrier, helping the reaction proceed under relatively mild thermal conditions without any added catalyst.
Why this matters for future medicines
Overall, the work shows that a simple lactam ring can act as a dynamic, recyclable guide that unlocks previously inaccessible bond‑forming patterns in boronic acid chemistry. For nonspecialists, the key message is that chemists now have a more direct, efficient way to turn basic building blocks into two important classes of nitrogen-containing molecules that underpin many drugs. Because the method avoids precious metals, tolerates a wide range of functional groups, and works on advanced drug-like structures, it is poised to help streamline the design and optimization of future pharmaceuticals and potentially new agrochemicals as well.
Citation: Lei, J., Xu, J., Li, X. et al. Lactam enables remote boronate rearrangements to C═N bonds. Commun Chem 9, 88 (2026). https://doi.org/10.1038/s42004-026-01890-2
Keywords: boronic acids, lactam chemistry, C–N bond formation, quinoxalinones, medicinal chemistry