Clear Sky Science · en
ETV7 promotes 5-FU resistance and malignant progression through CXCL1-induced NETs formation in colorectal cancer
Why this research matters
Many people with colorectal cancer are treated with the long‑standing chemotherapy drug 5‑fluorouracil (5‑FU). For some, the drug works well at first, only to lose its punch as the tumor finds ways to fight back. This study uncovers a hidden partnership between cancer cells and the body’s own immune cells that helps tumors withstand 5‑FU, and it points to new ways doctors might break that alliance and restore the drug’s power.
A cancer “switch” turned up too high
The researchers focused on a protein called ETV7, a gene‑control switch found in many cell types. By examining public cancer databases and tissue samples from patients, they found that ETV7 levels are much higher in colorectal tumors than in nearby healthy tissue. Patients whose tumors carried more ETV7 tended to have poorer overall and disease‑free survival, suggesting that this protein is linked to more aggressive disease. In laboratory-grown cancer cells, dialing ETV7 up made cells divide, move, and invade more readily, while dialing it down curbed these malignant behaviors.
When the lab dish misleads
Curiously, changing ETV7 levels did not make cancer cells clearly more or less sensitive to 5‑FU when they were grown alone in simple plastic dishes. Yet in mice, tumors engineered to produce extra ETV7 kept growing despite 5‑FU treatment. This mismatch hinted that a key piece of the story was missing in the dish but present in the living body: the surrounding support system of blood vessels, connective tissue, and immune cells known as the tumor microenvironment.
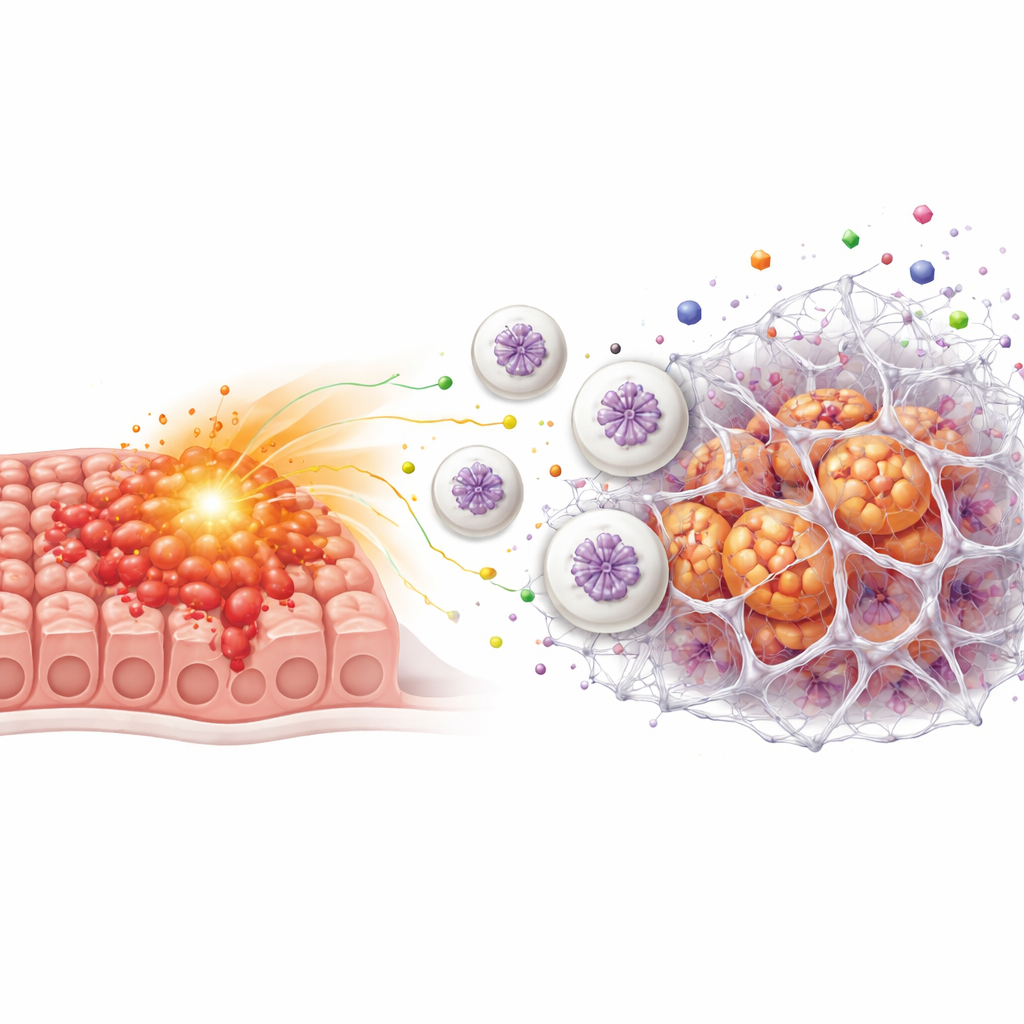
Recruiting first‑responder cells to build sticky webs
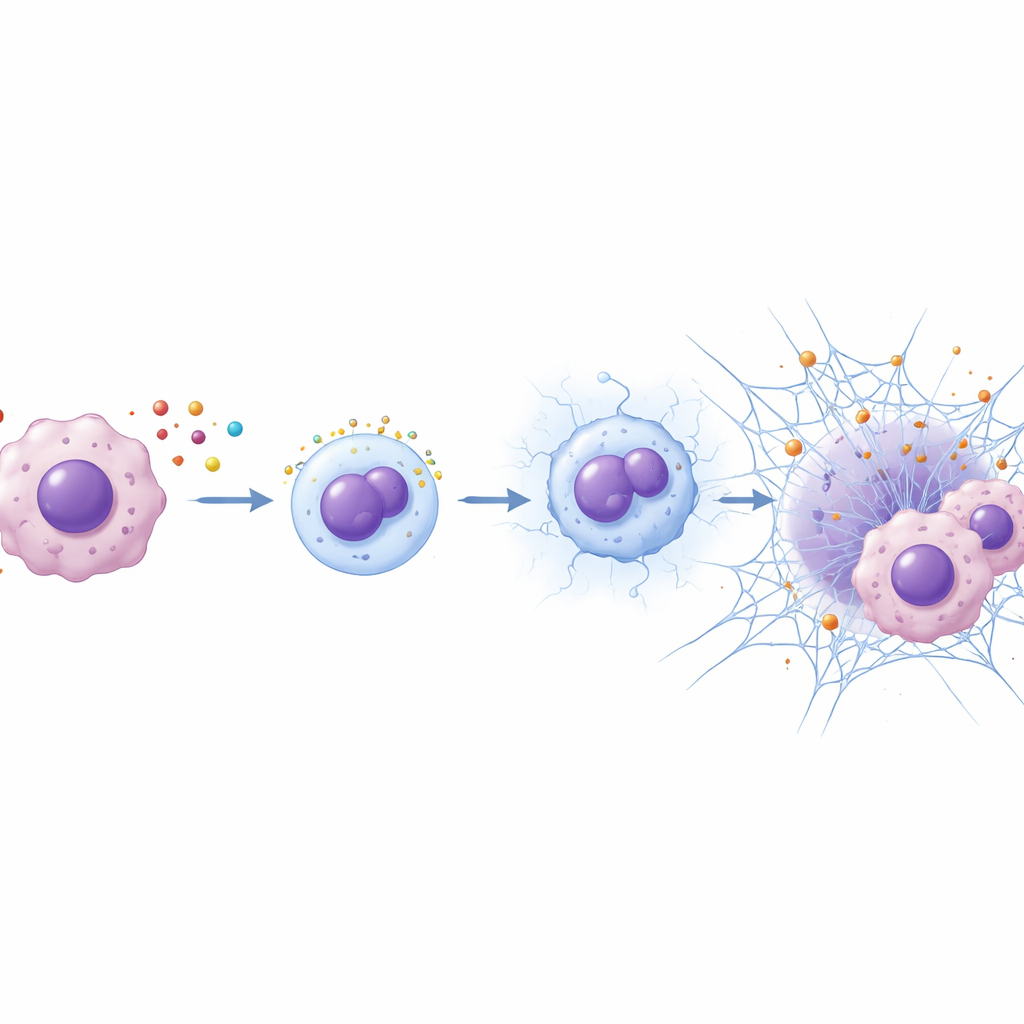
The team next asked whether ETV7 helps tumors reshape their microenvironment. Data mining linked high ETV7 to greater infiltration of neutrophils, a type of white blood cell best known for fighting infections. In mouse tumors rich in ETV7, more neutrophils clustered around the cancer. When cancer cells with high ETV7 were co‑cultured with neutrophils, the neutrophils dramatically boosted the cancer cells’ growth, invasiveness, and resistance to 5‑FU. The researchers traced this effect to web‑like structures called neutrophil extracellular traps, or NETs—tangles of DNA and proteins that neutrophils normally cast out to ensnare microbes. Tumors with high ETV7 contained more NETs, and fluid from ETV7‑high cancer cells spurred neutrophils to release these sticky webs in the lab.
A signaling chain from tumor to neutrophil
How does ETV7 convince neutrophils to arrive and spin these traps? Gene analyses pointed to CXCL1, a small, soluble signaling molecule that acts as a beacon for neutrophils. The study showed that ETV7 binds directly to the control region of the CXCL1 gene in colorectal cancer cells and turns its production up. As CXCL1 levels rise, more neutrophils are drawn toward the tumor and become activated. When CXCL1 was blocked or reduced, NET formation dropped and the tumor‑boosting, drug‑resisting effects of ETV7 largely faded—especially when neutrophils were present. In mice treated with 5‑FU, either lowering CXCL1, neutralizing it with an antibody, or enzymatically breaking down NETs all shrank ETV7‑driven tumors, and combining CXCL1 blockade with NET breakdown worked best.
Breaking the tumor’s protective shield
Altogether, the work reveals an ETV7–CXCL1–NETs axis in colorectal cancer: ETV7 in tumor cells increases CXCL1, CXCL1 summons and activates neutrophils, and those neutrophils cast NETs that form a protective, pro‑growth niche around the tumor. This shield helps cancer cells survive and spread under 5‑FU treatment. For patients, the findings suggest that measuring ETV7 or CXCL1 might help identify tumors primed for resistance, and that drugs aimed at CXCL1 signaling or NET formation could be paired with standard chemotherapy to improve its effectiveness.
Citation: Mo, S., Xia, P., Lv, Y. et al. ETV7 promotes 5-FU resistance and malignant progression through CXCL1-induced NETs formation in colorectal cancer. Commun Biol 9, 472 (2026). https://doi.org/10.1038/s42003-026-09976-2
Keywords: colorectal cancer, chemotherapy resistance, tumor microenvironment, neutrophil extracellular traps, CXCL1 signaling