Clear Sky Science · en
PACS1 syndrome mutation disrupts dynein-mediated cargo transport via HDAC6 and BICD2
Why this matters for brain development
PACS1 syndrome is a rare genetic condition that causes intellectual disability, seizures, and distinctive facial features. Families and clinicians know the symptoms, but until recently, it was unclear what was actually going wrong inside patients’ cells. This study peels back that mystery, showing how a single-letter change in the PACS1 gene scrambles the cell’s internal transport system, especially in neurons, and how that links PACS1 syndrome to a broader family of “traffic” diseases of the nervous system. 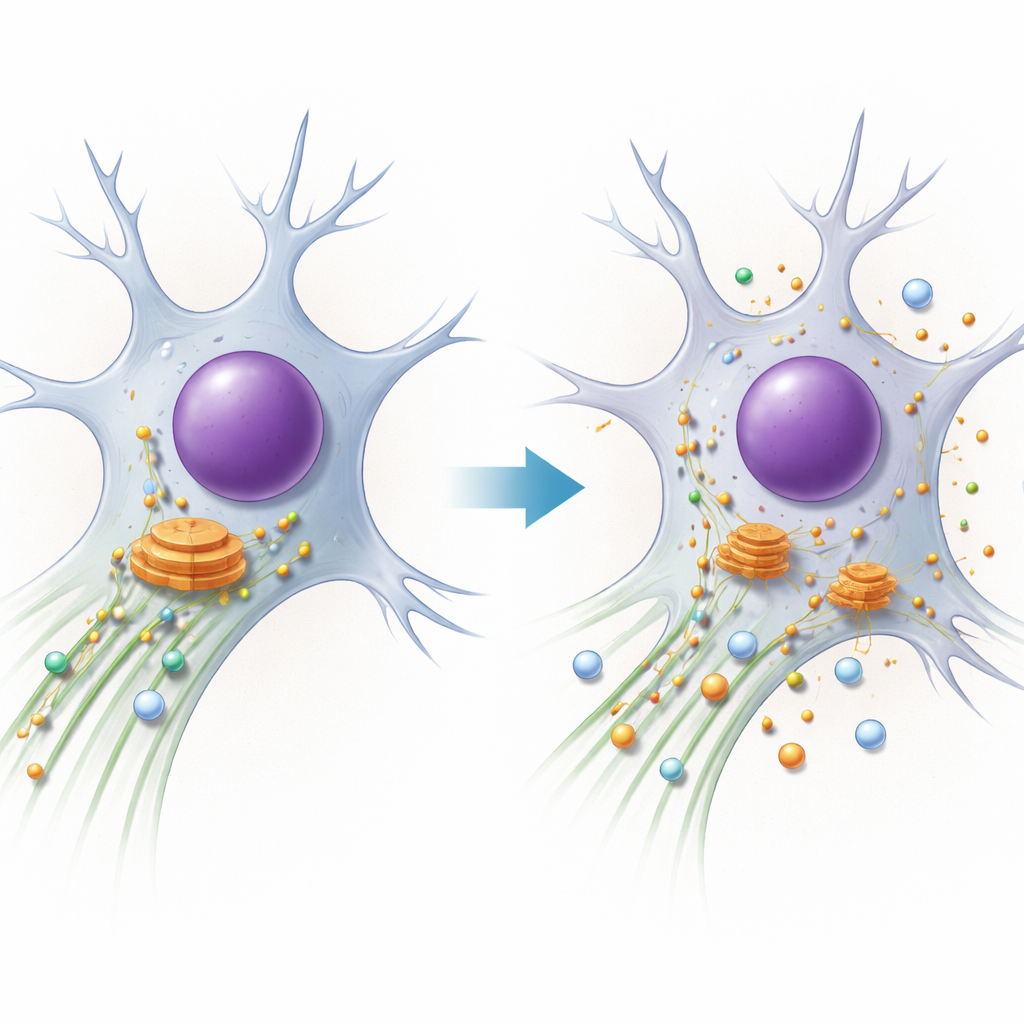
The cell’s delivery highways
Every cell relies on microscopic highways made of protein tubes called microtubules. Molecular motors run along these tracks, hauling cargo such as enzymes and membrane compartments to where they are needed. One motor, dynein, is responsible for long-distance “return trips,” pulling cargo from the outer cell back toward the center, where a major sorting station called the Golgi apparatus sits. The authors focus on PACS1, a protein that helps choose which cargo rides dynein and also tunes the state of the tracks through another protein, HDAC6, which controls a chemical tag (acetylation) on the microtubules. Neurons, with their very long extensions, are especially sensitive to even small glitches in this system.
A mutant coordinator that grabs too tightly
PACS1 syndrome is caused by the recurring R203W mutation in PACS1. The team found that both normal and mutant PACS1 physically bind dynein’s heavy chain, but the mutant form latches on more strongly. Using patient skin cells and engineered cell lines, they showed that this over‑engagement produces the same outcome as partially losing dynein’s function: the Golgi breaks apart into scattered mini‑stacks, and an important enzyme called furin, normally parked at the Golgi’s trans‑side, is misrouted into other compartments. By dissecting PACS1’s structure, they pinpointed a short beta‑strand “patch” that specifically contacts dynein. When they altered this patch without disturbing other PACS1 roles, furin again lost its proper address, confirming that the dynein–PACS1 handshake is crucial for correct cargo placement. 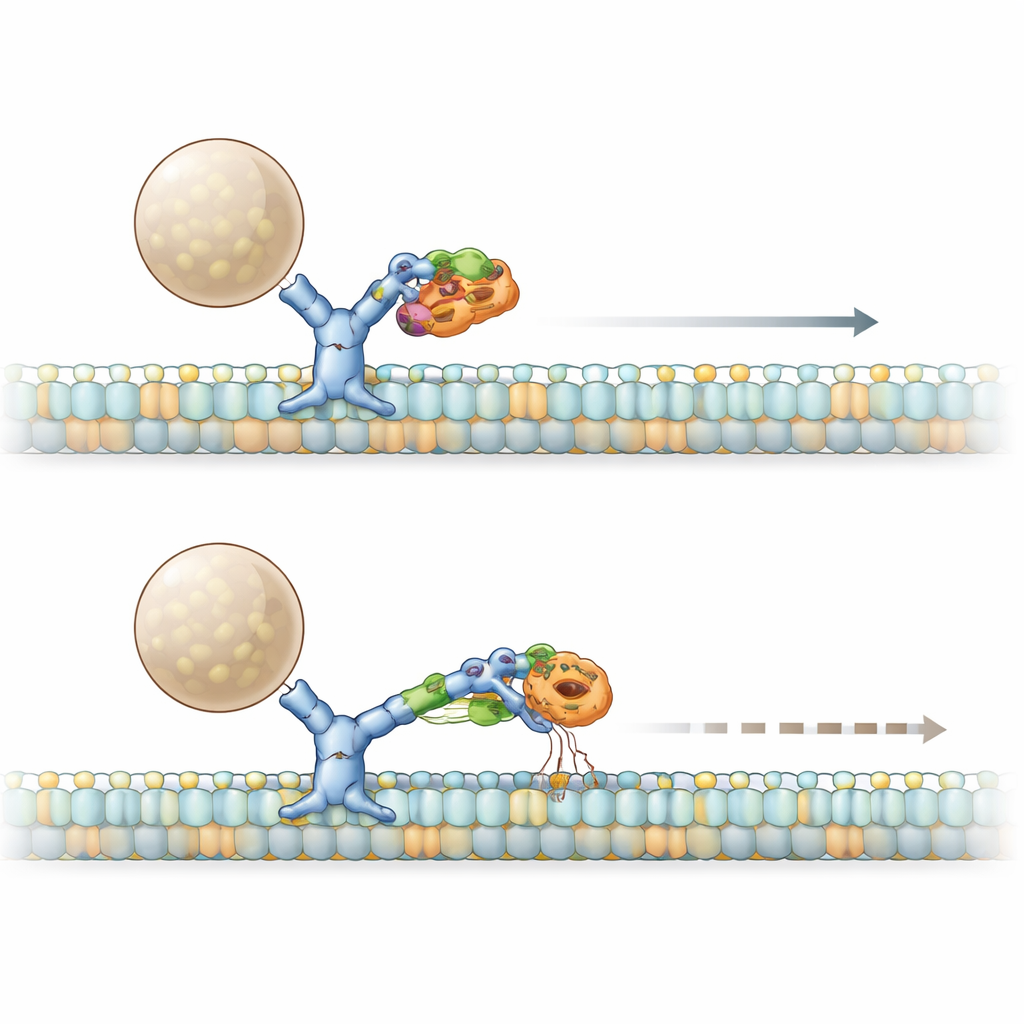
How a three‑protein alliance jams the motor
The story deepens with two additional players: HDAC6 and an adaptor called BICD2, which helps assemble active dynein complexes. The mutant PACS1 not only heightens HDAC6 activity, reducing acetylation on microtubules, but also forms an enlarged complex with HDAC6 and BICD2. Biochemical tests revealed that, together, mutant PACS1 and HDAC6 pry BICD2 away from dynein while leaving its partnership with a forward‑moving motor (kinesin) intact. In living cells, an artificial cargo that normally zips briskly along dynein slowed down and fewer cargoes moved at all when mutant PACS1 was present. Blocking HDAC6’s activity restored both speed and the number of moving particles, showing that the mutant PACS1–HDAC6–BICD2 assembly acts like a brake on dynein’s ability to start and sustain transport.
Rescuing traffic with a natural dynein helper
The researchers next tested whether dynein could be “re‑activated” despite the mutant complex. They turned to Lis1, a known dynein helper that stabilizes active motor assemblies. In patient cells, Lis1 levels were somewhat reduced. When the team added extra Lis1, two things improved: the Golgi re‑clustered near the cell center, and lysosomes, another class of organelles that had drifted outward, snapped back into place. In the same motility assay, Lis1 boosted both the frequency and speed of dynein‑driven cargo movement in the presence of mutant PACS1. These results show that the problem is not that dynein is missing, but that it is trapped in a poorly active state—a state that can be partially corrected by either dampening HDAC6 or reinforcing dynein activation.
Connecting a rare syndrome to a wider set of disorders
By combining structural analysis, cell biology, and live imaging, the authors propose a clear model: PACS1 normally links selected cargo to dynein and fine‑tunes motor performance through HDAC6 and microtubule acetylation. The R203W mutation opens up PACS1’s interaction surface, over‑recruiting HDAC6 and BICD2 into a complex that undercuts dynein’s ability to grab microtubules and move efficiently. The result is widespread misplacement of Golgi, lysosomes, and other cargoes, particularly in neurons where long‑range transport is vital. This mechanism helps explain why lowering PACS1 or HDAC6 with antisense therapies corrects brain defects in mouse models and is now being explored in patients. More broadly, it places PACS1 syndrome within the growing spectrum of microtubule trafficking disorders, alongside conditions caused by mutations in dynein, BICD2, and related transport factors.
Citation: Yang, Y., Thomas, L., Chen, K. et al. PACS1 syndrome mutation disrupts dynein-mediated cargo transport via HDAC6 and BICD2. Commun Biol 9, 450 (2026). https://doi.org/10.1038/s42003-026-09924-0
Keywords: PACS1 syndrome, dynein transport, microtubule trafficking, Golgi organization, neuronal development