Clear Sky Science · en
The secretory protein, CLCF1, improves cholestatic liver disease by inhibiting hepatic bile acid synthesis and promoting bile acid excretion
Why this research matters for liver health
Cholestatic liver diseases are conditions in which bile, a digestive fluid made by the liver, stops flowing properly and instead builds up, slowly poisoning the liver from within. Current medications help only a fraction of patients, and many ultimately need a liver transplant. This study uncovers a naturally produced protein, called CLCF1, that the body seems to switch on as a defense system during cholestatic liver disease. Understanding how this protein works could point the way toward new treatments that protect the liver by both reducing the production of harmful bile components and helping the body get rid of them more efficiently.
A hidden helper in sick livers
The researchers began by examining liver tissue from patients with primary biliary cholangitis and primary sclerosing cholangitis, two major forms of long-lasting cholestatic liver disease. They compared people with early disease to those with more advanced cholestasis, and also analyzed existing public gene datasets. Across these independent human data sources, one secreted protein stood out: CLCF1 was consistently among the most strongly increased genes in cholestatic livers. Higher CLCF1 levels tracked with worse blood markers of disease severity, suggesting that the liver ramps up this protein as stress and bile buildup worsen. In early-stage patients treated with the standard drug ursodeoxycholic acid, those who responded well tended to have more CLCF1 in their livers, hinting that this protein may mark a more adaptable, treatment-sensitive liver.
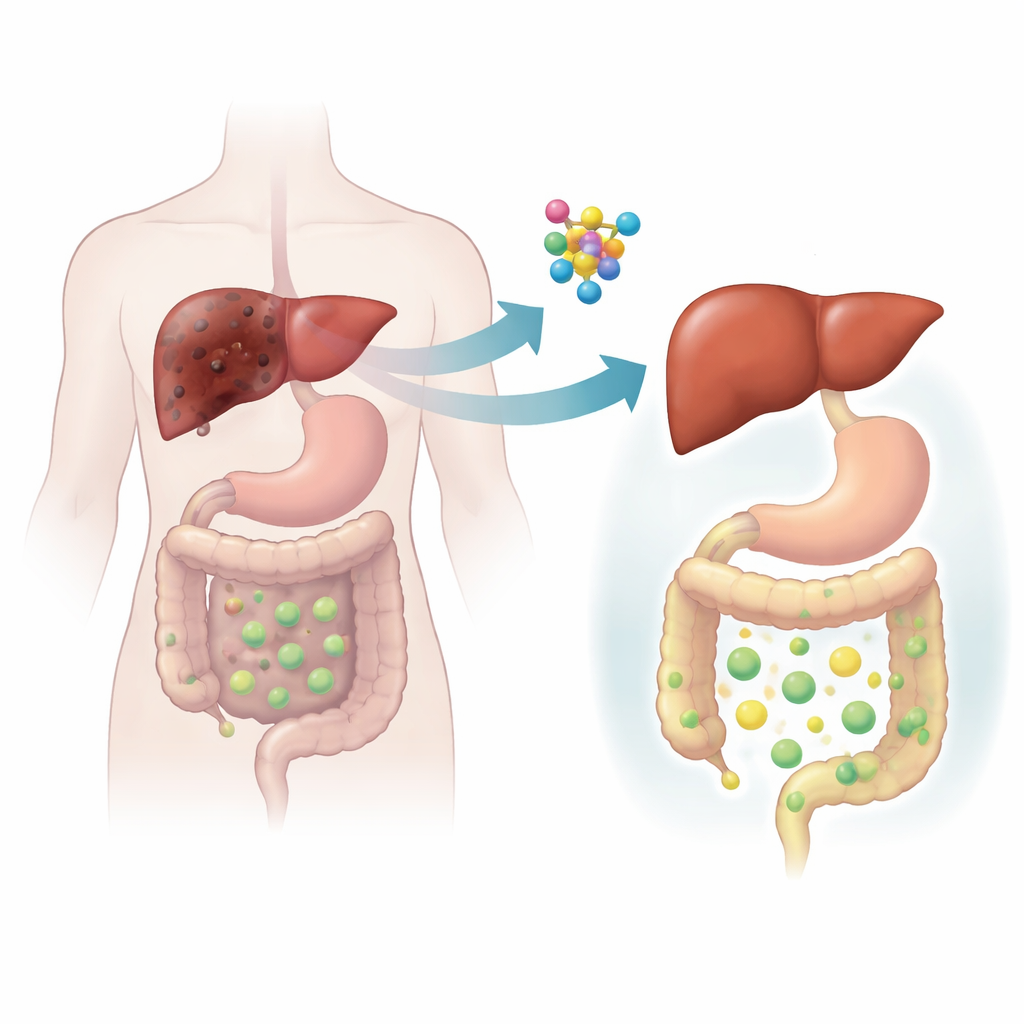
Testing CLCF1 in animal models
To move beyond human correlations, the team turned to mice and used both genetic and dietary tricks to mimic cholestatic liver disease. In two different mouse models, cholestasis strongly increased the liver’s production of Clcf1, mirroring the human findings. When the scientists specifically removed the receptor for CLCF1 from liver cells, cholestatic injury became much worse: bile ducts overgrew, liver tissue died, inflammation rose, scar tissue accumulated, and toxic bile acids built up. The reverse experiment told a more hopeful story. When the team used a harmless virus to boost Clcf1 levels only in the liver, the mice were protected. Their livers looked healthier, blood tests improved, scarring was reduced, and the amount of bile acids trapped in the liver dropped markedly.
Turning down the tap and opening the drain
How does CLCF1 lower bile acid overload so effectively? The researchers found two complementary actions. First, CLCF1 directly calmed the liver’s own bile-making machinery. In multiple mouse models and in cultured liver cells, extra CLCF1 consistently switched off key enzymes that convert cholesterol into bile acids, effectively turning down the tap on new bile production. This occurred without activating the usual liver switch for bile control, a nuclear receptor called FXR, suggesting CLCF1 uses an alternate route inside liver cells to slow bile synthesis. Second, CLCF1 influenced how bile was handled further downstream in the body. It reshaped the gut’s microbial community toward bacteria that are particularly good at processing bile acids and increased the amount of bile acids leaving the body in stool, acting like a stronger drain.
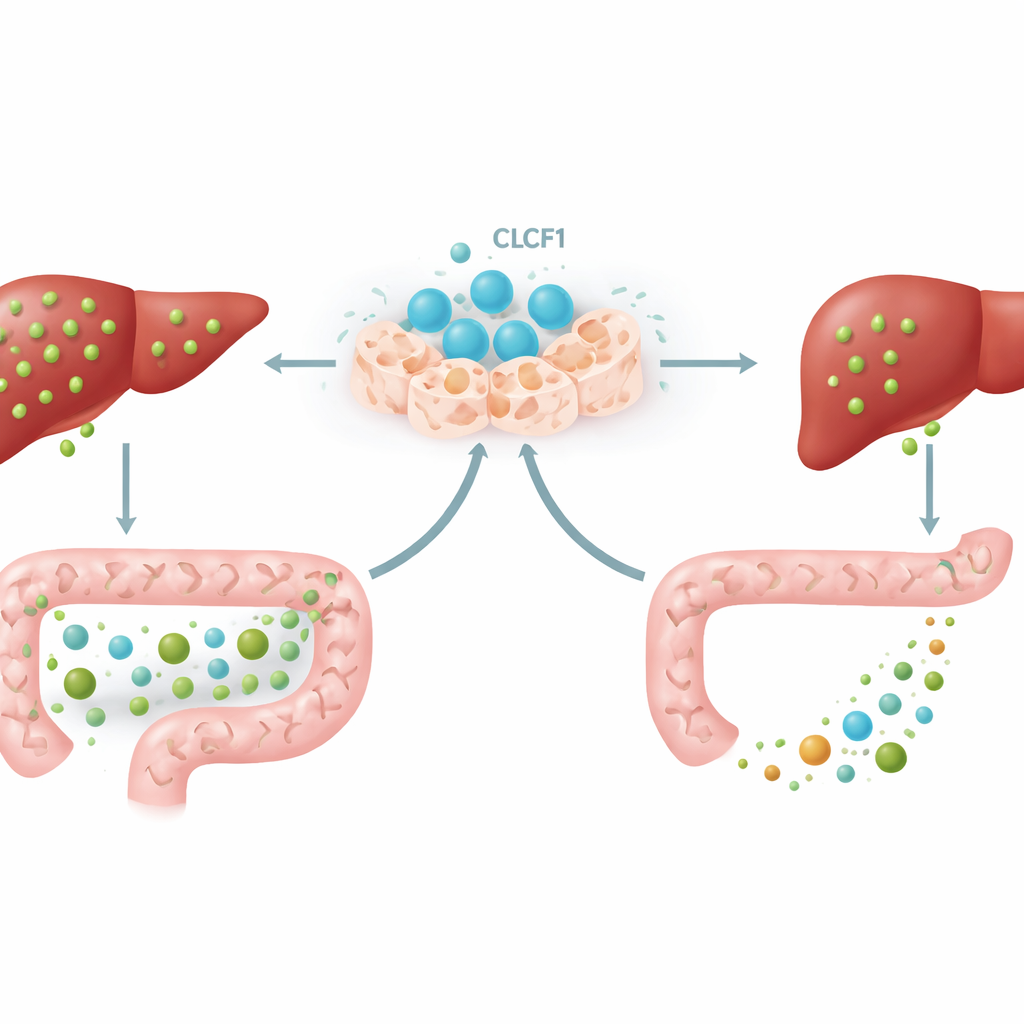
The gut–liver conversation
A key piece of the puzzle involved a signaling loop between the intestine and the liver. Certain bile acids in the gut activate a sensor called FXR in intestinal cells, which then release a messenger hormone known as FGF15 in mice. This hormone travels through the blood to the liver and tells it to slow bile acid production. With extra CLCF1, mice accumulated specific types of bile acids in their intestines—particularly forms known to be strong FXR activators. As a result, intestinal FXR switched on, FGF15 levels rose in both gut and liver, and the liver further dampened its bile acid production. When scientists blocked intestinal FXR with a gut-limited drug, much of CLCF1’s protective effect faded, demonstrating that this gut–liver conversation is an essential part of the protein’s action.
What this could mean for future treatments
Together, these findings portray CLCF1 as a built-in safety valve for cholestatic liver disease. The protein cuts bile acid levels from both ends: it slows their creation inside the liver and encourages their removal through the intestine, aided by friendly gut microbes and a hormonal feedback signal. Unlike existing drugs that activate FXR throughout the body and can cause side effects such as itching and abnormal blood fats, CLCF1 seems to steer bile control more gently and selectively, especially through the intestine. While this work is still in mice and laboratory samples, it suggests that boosting CLCF1 or mimicking its pathways could form the basis of new therapies—and that measuring this protein, or the bile acids it enriches in the gut, might help identify patients who would benefit most.
Citation: Liu, M., Su, Y., Hu, Y. et al. The secretory protein, CLCF1, improves cholestatic liver disease by inhibiting hepatic bile acid synthesis and promoting bile acid excretion. Commun Biol 9, 370 (2026). https://doi.org/10.1038/s42003-026-09847-w
Keywords: cholestatic liver disease, bile acids, gut–liver axis, CLCF1, gut microbiota