Clear Sky Science · en
Regulatory T cells in axial spondyloarthritis
When the Body’s Peacekeepers Go Awry
Axial spondyloarthritis is a form of inflammatory arthritis that targets the spine and sacroiliac joints, causing chronic pain and stiffness, often in young adults. This review explores a surprising player in the disease: the body’s own immune “brakes,” called regulatory T cells. These cells normally keep inflammation in check, but in axial spondyloarthritis they may be overrun, weakened, or even turn into culprits themselves. Understanding how and where these peacekeeping cells fail could open the door to more precise, longer-lasting treatments.
Guardians of Balance in the Immune System
Regulatory T cells are a specialized branch of white blood cells that develop from precursor cells in the thymus and in the body’s tissues. Their job is to restrain aggressive immune cells that fight infections so that these attackers do not start damaging the body’s own joints, skin, or gut. They do this by direct contact with other cells, by soaking up growth signals, and by releasing calming messenger molecules. In healthy situations, after an infection is cleared, inflammatory cells quiet down and regulatory cells help restore peace. In axial spondyloarthritis, however, inflammation driven by so‑called type 3 immunity—cells that produce the molecule IL‑17A—persists, suggesting this balance has been disturbed.
How Peacekeepers Lose Their Stability
The review highlights that regulatory T cells are remarkably adaptable. They can adjust their behavior to match the type of immune response they are trying to control, even borrowing features from the very inflammatory cells they police. This flexibility is controlled by a network of genes and chemical switches, centered on a master control protein called FOXP3. In axial spondyloarthritis, several lines of evidence suggest this control system is unstable. Patients show changes in how FOXP3 is turned on and chemically marked, reduced signaling through growth factors that normally sustain regulatory cells, and genetic variants that nudge these cells toward fragility. Under intense or prolonged inflammatory pressure, some regulatory cells appear to lose FOXP3, shedding their calming identity and drifting toward aggressive, IL‑17A‑ or interferon‑producing states.
Different Tissues, Different Stories
Regulatory T cells do not behave the same way everywhere in the body. In the gut, where bacteria and food constantly stimulate the immune system, these cells are abundant and often extremely effective. In people with axial spondyloarthritis, sections of the intestine can show an expansion of regulatory cells and anti‑inflammatory signals, hinting that they may successfully contain gut inflammation. By contrast, in joints and at tendon attachment sites, regulatory cells seem fewer, less stable, or outcompeted by inflammatory forces. Studies in human joint fluid and animal models suggest that local signals such as high levels of tumor necrosis factor, low oxygen, and products from neutrophils can blunt regulatory activity, push them toward inflammatory behavior, or favor tissue pathways that bypass them altogether. These site‑specific differences may help explain why some patients develop gut disease, psoriasis, or new bone growth in addition to spinal inflammation.
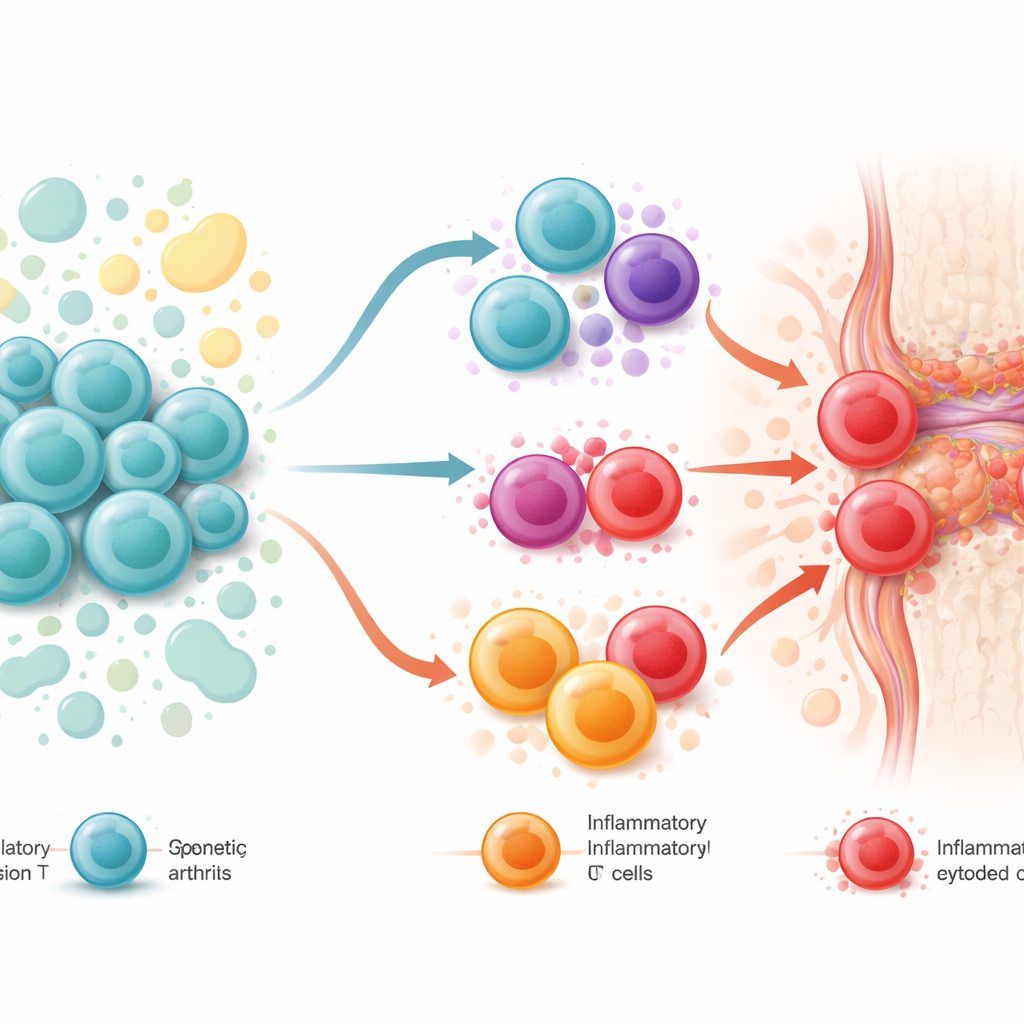
Three Ways the System Can Fail
Pulling together genetic data, cell studies, and animal experiments, the authors propose three overlapping scenarios for how regulatory T cells might contribute to axial spondyloarthritis. First, they may be “innocent bystanders”: basically functional but overwhelmed by the sheer volume of inflammatory signals. Second, they may be “good cells gone bad,” weakened by inherited risk variants and environmental stresses so that they lose their calming powers and begin to resemble the very inflammatory cells they should restrain. Third, they may act as “bad actors from the start,” using their killing abilities in an unhelpful way—through specialized regulatory subsets that can destroy other immune cells or tissue cells and thereby feed into autoimmune damage. These states likely form a continuum that shifts as disease progresses and as treatments are introduced.
What This Means for Future Treatment
To a non‑specialist, the key message is that axial spondyloarthritis may not be driven only by overactive inflammatory cells, but also by failure or misdirection of the body’s own internal brakes. This insight suggests that future therapies might not just block single inflammatory molecules but also restore or replace healthy regulatory T cells at the right time and in the right tissues. Approaches under consideration include drugs that stabilize FOXP3, fine‑tune cellular metabolism, or deliver new regulatory cells or their vesicles back into patients. As new tools allow researchers to study small samples of spinal and tendon tissue in detail, these ideas can be tested and refined, bringing the prospect of more tailored, stage‑specific treatments closer to reality.
Citation: Pacheco, A., Tavasolian, F., Lim, M. et al. Regulatory T cells in axial spondyloarthritis. Commun Biol 9, 473 (2026). https://doi.org/10.1038/s42003-026-09829-y
Keywords: axial spondyloarthritis, regulatory T cells, immune tolerance, IL-17 inflammation, autoimmune arthritis