Clear Sky Science · en
The type VI secretion system of Acinetobacter: mechanisms, biology and therapeutic potential
Hidden weapons in hospital germs
Many people know that some hospital germs can shrug off antibiotics, but fewer realize that these microbes also fight fierce battles with one another. This review looks at a microscopic “spring‑loaded spear” inside Acinetobacter, a group of bacteria that includes the notorious hospital bug Acinetobacter baumannii. Understanding how this built‑in weapon works, when it is switched on, and how it helps infections take hold could open new paths for diagnosing and disarming life‑threatening, drug‑resistant infections.
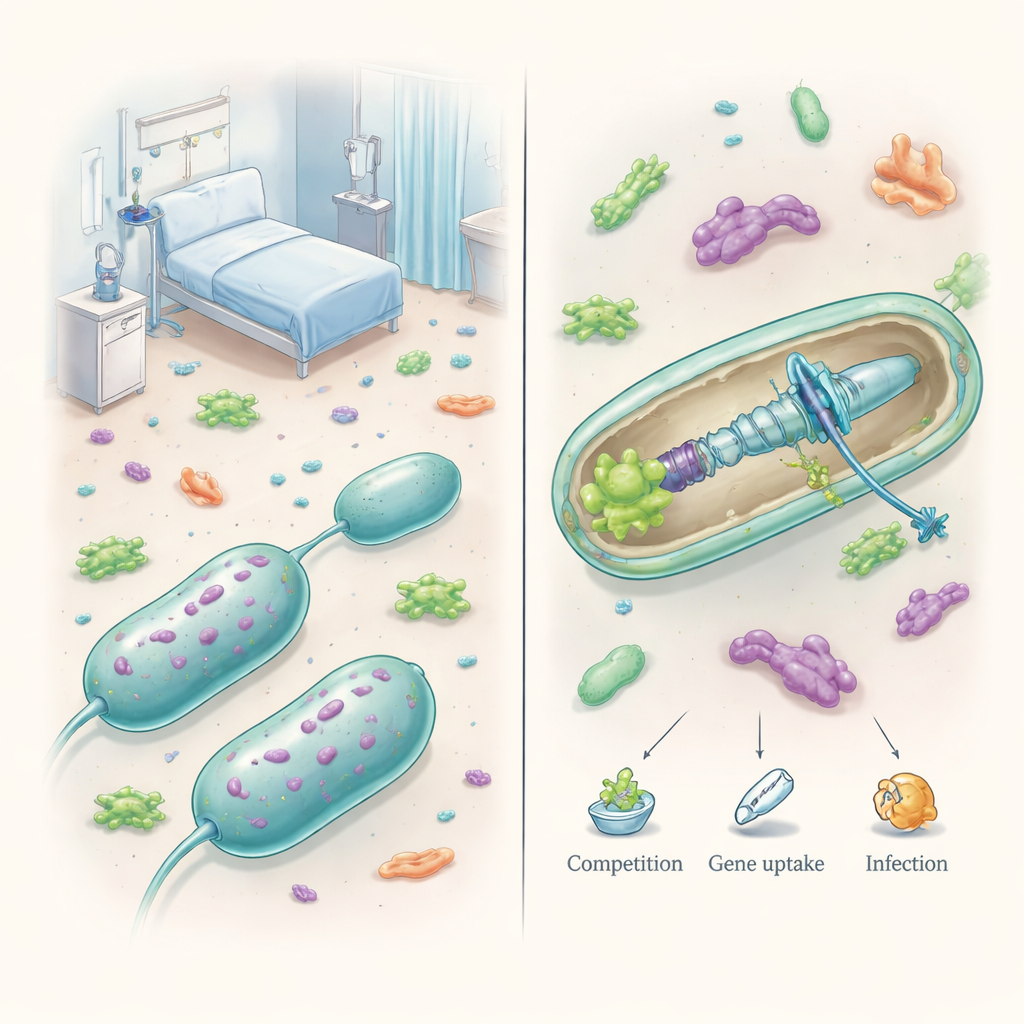
A survival machine, not just a poison dart
Acinetobacter species live in soil, water, and on our skin, but some have become highly successful hospital pathogens. A key tool in their arsenal is the Type VI secretion system, or T6SS, a tiny contractile machine anchored in the cell envelope. When triggered, it fires a needle‑like spike loaded with toxic proteins into neighboring cells, often killing competing bacteria on contact. Unlike some other germs that carry several versions of this system, pathogenic Acinetobacter usually has only one T6SS, yet it has rewired this single platform to serve many purposes, from warding off rivals to shaping infections.
Rebuilt hardware: an unusual launch platform
Most bacteria build their T6SS around a standard set of parts, but Acinetobacter has remodeled several key pieces. It lacks a common outer‑membrane anchor called TssJ and instead relies on a trio of specialized helpers: TsmK forms a foundation in the inner membrane, TslA stabilizes a long “tunnel” protein (TssM) as it crosses the cell wall, and TagX locally cuts the wall to make way for the weapon. On top of this, a particular spike protein, VgrG1, must be almost perfectly shaped for the system to fire at all—single amino‑acid changes can disable it. Together, these tweaks show how evolution can replace missing parts with new solutions while preserving the basic firing principle.
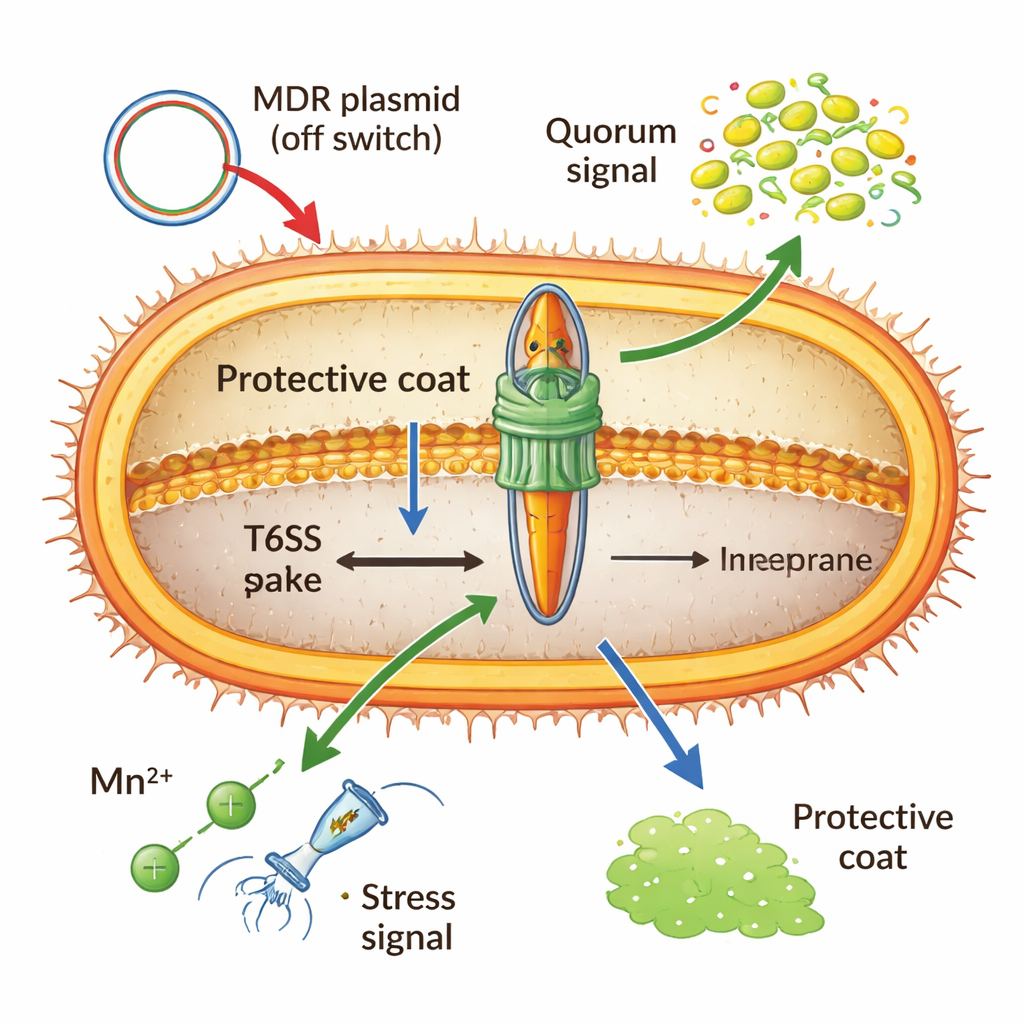
Smart controls: when attack mode makes sense
Building and firing this nanomachine is expensive, so Acinetobacter keeps it under tight control. A global DNA‑binding protein (H‑NS) and special repressors carried on multidrug‑resistant plasmids can lock the system in the “off” position, especially when resistance genes are being shared between cells. At high cell density, chemical “quorum” signals can switch it on, preparing the bacteria for close‑quarters competition. Metal ions also play a role: under oxidative stress, imported manganese activates a small RNA that causes key T6SS messages to be destroyed, dialing down the weapon to favor survival inside the host. Even the capsule—a sugary outer coat—acts as both shield and brake, blocking incoming attacks but also physically dampening Acinetobacter’s own firing.
A diverse toolkit of microscopic blows
When Acinetobacter does pull the trigger, it can deliver a wide range of toxins. Some enzymes chew through the tough cell wall of rivals; others quietly erode cell membranes, and still others slice up DNA inside target cells. Each toxin is paired with a matching “immunity” protein that protects the attacker from friendly fire. One DNA‑cutting toxin, now called TafE, can even kill fungi, hinting at cross‑kingdom skirmishes in places like the gut or lungs. Genomic studies suggest that Acinetobacter encodes many more, as‑yet‑uncharacterized toxins, often bundled in large Rhs proteins that act as modular carriers. Rather than relying on a single blow, these bacteria appear to use layered attacks that damage several essential structures at once.
Shaping infections, genes, and future treatments
Evidence from patient isolates and animal models suggests that an active T6SS often goes hand‑in‑hand with more severe disease, stronger inflammation, and better survival of the bacteria inside hosts. By killing neighboring microbes, the system also releases DNA that naturally competent Acinetobacter cells can absorb, speeding the spread of traits like antibiotic resistance. Yet keeping the weapon constantly active is costly, so many successful hospital strains carry plasmids or mutations that silence it once resistance is secure. Overall, the authors argue that this secretion system should be seen not as a simple virulence factor but as a flexible “fitness module” that Acinetobacter tunes up or down to balance aggression, persistence, and energy use. This new view points toward practical applications, from vaccines that target conserved T6SS parts to drugs that selectively jam the firing mechanism, helping doctors outsmart a dangerous and adaptable hospital foe.
Citation: Jie, J., Gu, S., Li, D. et al. The type VI secretion system of Acinetobacter: mechanisms, biology and therapeutic potential. Commun Biol 9, 327 (2026). https://doi.org/10.1038/s42003-026-09782-w
Keywords: Acinetobacter, type VI secretion system, antibiotic resistance, bacterial competition, hospital infections