Clear Sky Science · en
Involvement of the hypothalamus–raphe magnus–spinal defecation center axis in stress-induced defecation in rats
Why stress can upset your stomach
Most people have experienced a churning stomach, sudden urgency, or even diarrhea during stressful situations such as exams or public speaking. While this mind–gut connection is well recognized, the exact nerve pathways that turn psychological stress into a rush to the bathroom have remained surprisingly unclear. This study in rats uncovers a specific chain of brain and spinal circuits that links emotional stress to powerful contractions in the lower bowel, helping explain why stress so often shows up in the restroom.
A brain route from worry to the gut
The researchers focused on a brain region called the hypothalamus, a key control center for both emotion and automatic body functions. Earlier work had shown that stress activates the hypothalamus and brainstem, and that these areas can speed up movement in the colon. However, it was not known exactly how signals traveled from the brain to the distant lower colon and rectum, which are crucial for defecation. By using tracing viruses that move along nerve fibers, the team mapped neurons that connect the hypothalamus to a brainstem structure known as the raphe magnus, and from there down the spinal cord toward the pelvis.
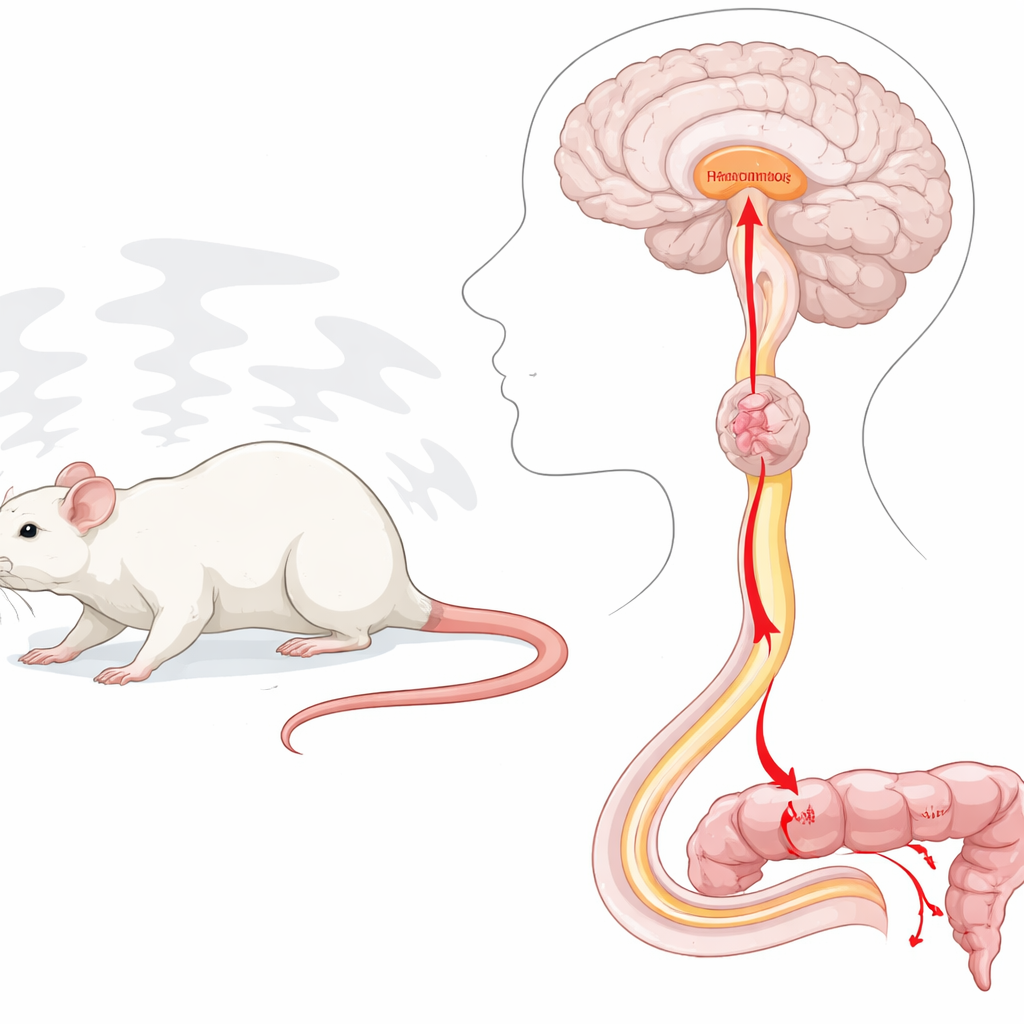
Stress lights up a specific brain–spinal–colon chain
To mimic psychological stress, rats were placed on a small platform surrounded by water, a classic “water avoidance” setup that reliably triggers anxiety and increased stool output. The scientists found that, under this stress, hypothalamic neurons that project to the raphe magnus became strongly activated. When they chemically excited the same hypothalamic zones—particularly parts called the paraventricular and dorsomedial regions—in anesthetized rats, the lower colon showed vigorous, propulsive contractions, and the animals’ blood pressure and heart rate rose. Blocking stress-related hormones called corticotropin-releasing factors did not stop these bowel contractions, suggesting that another signaling system was at work.
From brainstem to spinal cord to pelvic nerve
The next question was how activation in the brain reached the muscles of the bowel. The team showed that the raphe magnus sends long, downward fibers that release the chemical messenger serotonin in the lumbosacral spinal cord, where the defecation center resides. When the researchers applied drugs directly onto the spinal cord to block two key serotonin receptor types, the colon no longer responded to hypothalamic activation. Likewise, cutting the pelvic nerve—which carries parasympathetic signals from the spinal cord to the rectum and distal colon—abolished the enhanced motility. Together, these experiments reveal a continuous pathway: hypothalamus to raphe magnus, raphe to spinal defecation center, and spinal output through the pelvic nerve to the bowel.
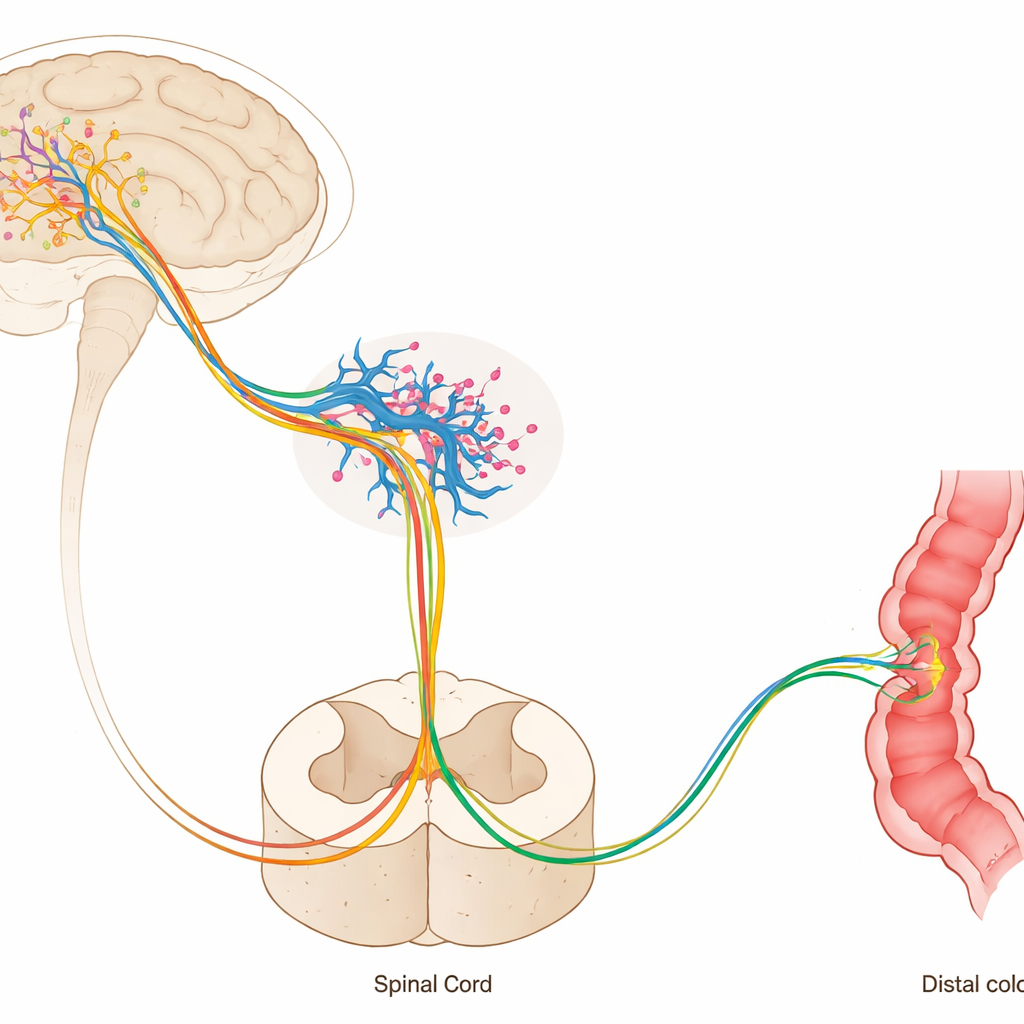
Oxytocin gives serotonergic neurons their marching orders
To identify what turns on the raphe magnus neurons in this chain, the researchers examined two candidates: glutamate, a common fast transmitter, and oxytocin, better known as a “bonding” hormone. By delivering targeted drugs into the raphe magnus while the hypothalamus was artificially stimulated, they discovered that blocking oxytocin receptors sharply reduced colon contractions, whereas blocking glutamate receptors did not. Microscopic imaging confirmed that oxytocin-containing fibers from the hypothalamus make close contact with serotonin-producing raphe neurons that project down to the spinal cord. In effect, oxytocin released in the raphe appears to flip on serotonergic cells, which then drive the spinal defecation center.
Turning the pathway off reduces stress-induced bowel movements
Finally, the team asked whether this pathway truly matters in awake animals under stress. Using a modern “chemogenetic” method, they engineered only those hypothalamic neurons that project to the raphe magnus to carry a special, drug-activated off switch. When this switch was triggered, hypothalamic stimulation no longer boosted colon motility, blood pressure, or heart rate in anesthetized rats. Most strikingly, in freely moving rats exposed to water-avoidance stress, silencing this pathway significantly reduced the number of fecal pellets they passed, even though the stressful situation was unchanged. This shows that the hypothalamus–raphe–spinal–pelvic nerve axis is not just present, but necessary for full-blown stress-induced defecation.
What this means for human gut troubles
In simple terms, the study identifies a concrete “worry-to-bowel” circuit: stress-sensitive cells in the hypothalamus activate oxytocin-sensitive serotonin neurons in the brainstem, which in turn stimulate spinal and pelvic nerves that make the lower colon squeeze and empty. Although the work was done in rats, similar wiring likely exists in humans and may help explain why some people develop diarrhea, and others constipation, when under pressure. By pinpointing specific brain and spinal hubs, this research offers new entry points for understanding and eventually treating stress-sensitive bowel disorders such as irritable bowel syndrome, where the brain–gut conversation goes awry.
Citation: Yuki, N., Sawamura, T., Mori, A. et al. Involvement of the hypothalamus–raphe magnus–spinal defecation center axis in stress-induced defecation in rats. Commun Biol 9, 411 (2026). https://doi.org/10.1038/s42003-026-09779-5
Keywords: stress and bowel function, brain gut connection, colon motility, oxytocin and serotonin, irritable bowel syndrome